연구 프로세스
59 동료 심사 연구의 그림
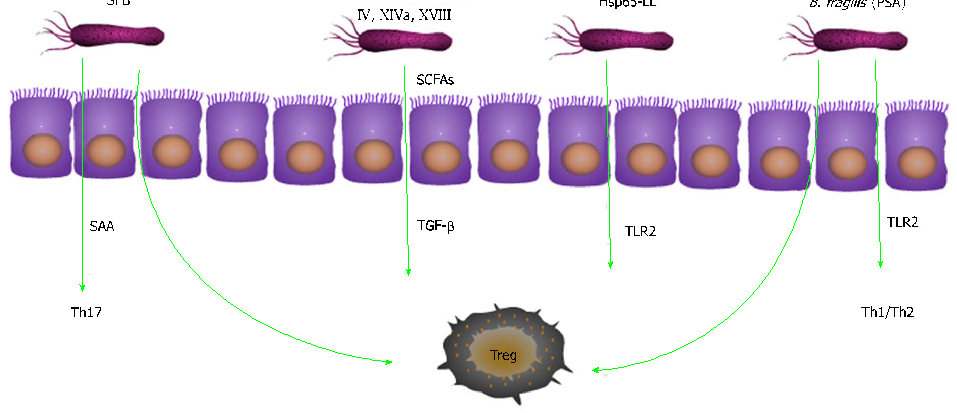
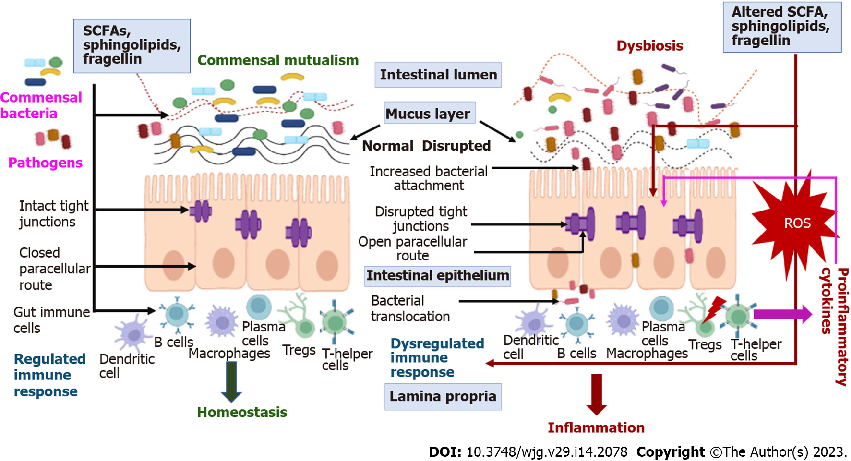
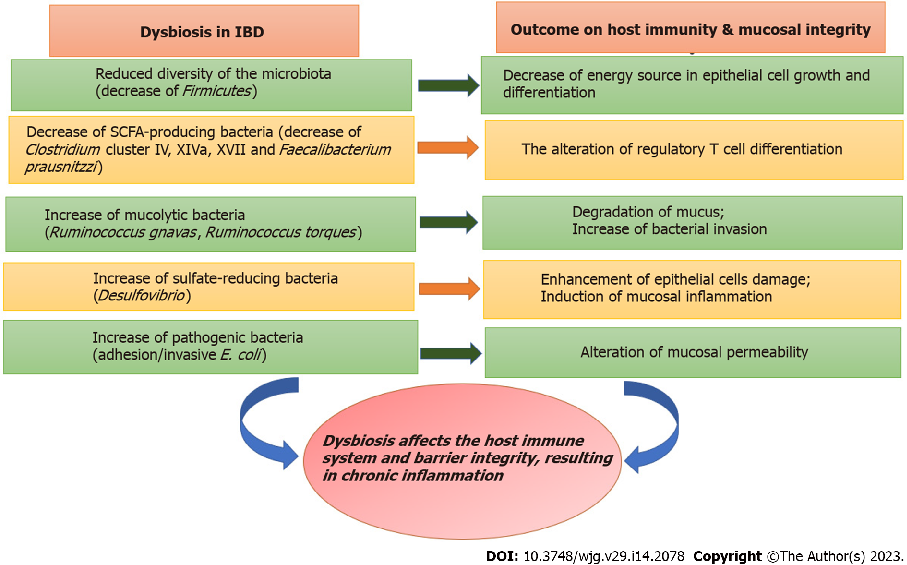
The intestinal microbiota in ulcerative colitis patients shows characteristic dysbiosis patterns, with reduced microbial diversity and altered composition compared to healthy individuals. Key bacterial phyla such as Firmicutes and Bacteroidetes are disproportionately affected.
Relationship between intestinal microbiota and ulcerative colitis: Mechanisms and clinical application of …
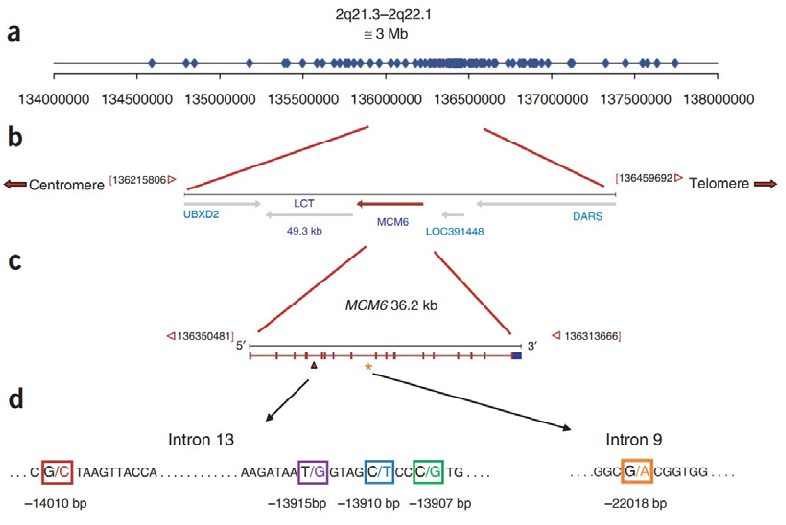
Genetic map of the lactase (LCT) and MCM6 gene region displays the locations of key single nucleotide polymorphisms associated with lactase persistence and non-persistence phenotypes across populations.
Lactose Intolerance in Adults: Biological Mechanism and Dietary Management.
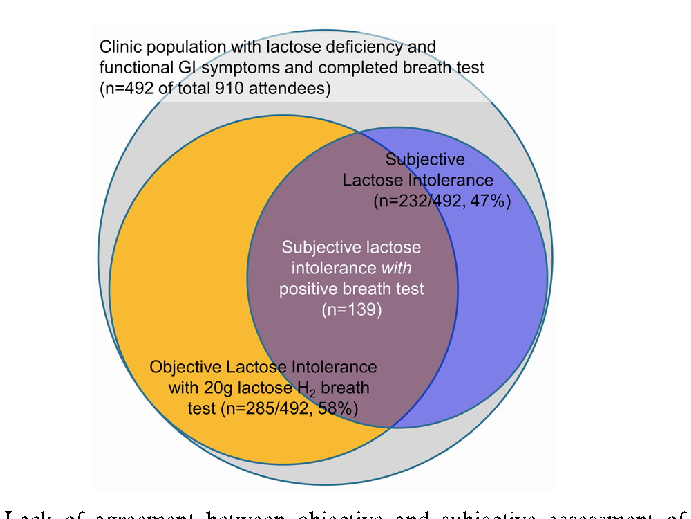
Dietary management strategies for lactose intolerance are summarized, including enzyme supplementation, dairy product selection, and gradual tolerance-building approaches.
Lactose Intolerance in Adults: Biological Mechanism and Dietary Management.
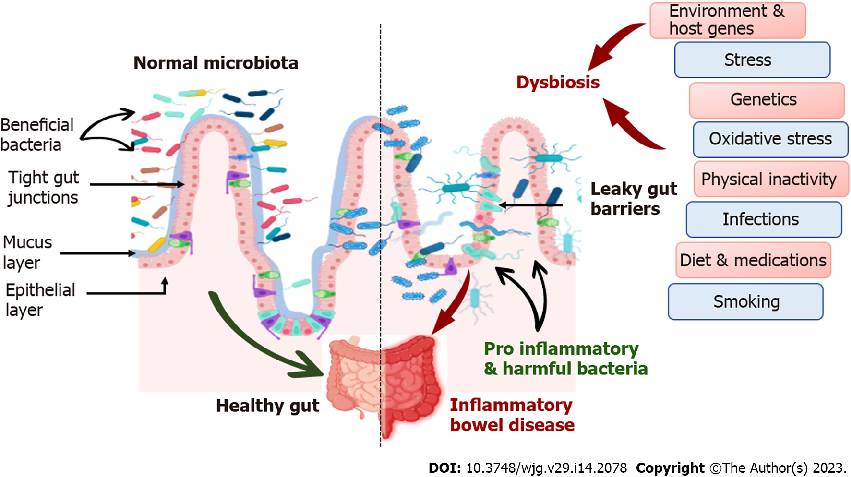
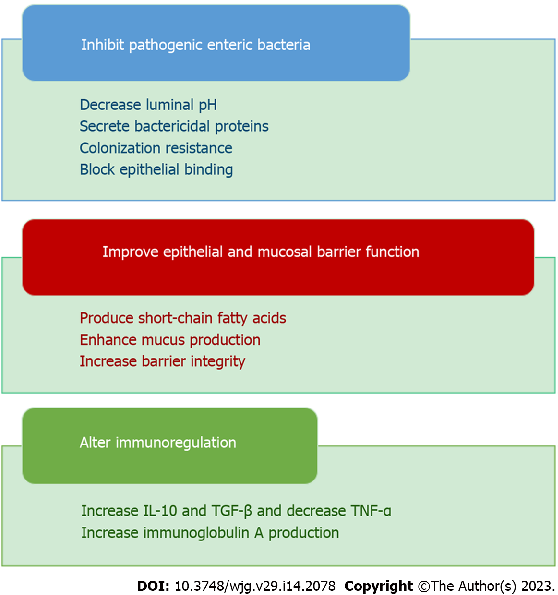
Inflammatory bowel disease pathogenesis involves interplay between genetic susceptibility, environmental factors, immune dysregulation, and gut microbiota alterations. Prebiotics, probiotics, and synbiotics target the microbial component of this multifactorial pathway.
Role of prebiotics, probiotics, and synbiotics in management of inflammatory bowel disease: …
Prebiotics serve as selective substrates for beneficial gut bacteria, promoting the growth of health-associated species such as Bifidobacterium and Lactobacillus. Common prebiotics include inulin, fructooligosaccharides, and galactooligosaccharides.
Role of prebiotics, probiotics, and synbiotics in management of inflammatory bowel disease: …
Probiotic mechanisms of action in IBD include strengthening the intestinal epithelial barrier, competing with pathogenic bacteria for adhesion sites, and modulating both innate and adaptive immune responses.
Role of prebiotics, probiotics, and synbiotics in management of inflammatory bowel disease: …
Synbiotics combine probiotics with prebiotics to achieve synergistic effects on gut health. The prebiotic component selectively nourishes the co-administered probiotic strain, potentially enhancing its colonization and metabolic activity.
Role of prebiotics, probiotics, and synbiotics in management of inflammatory bowel disease: …
Mechanisms of probiotic action in poultry include competitive exclusion of pathogens, production of bacteriocins, enhancement of intestinal barrier integrity, and stimulation of mucosal immune responses.
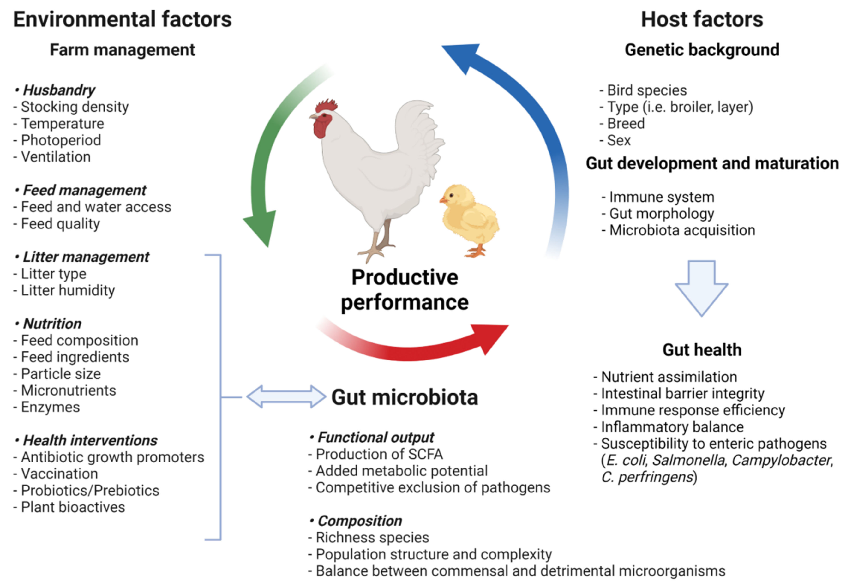
Probiotics, Prebiotics, and Phytogenic Substances for Optimizing Gut Health in Poultry.
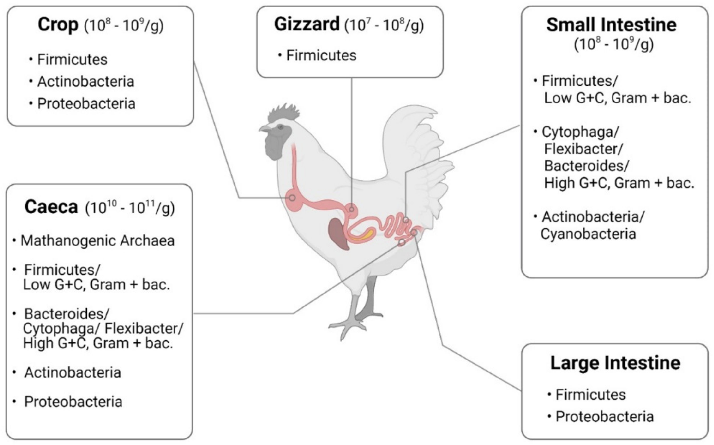
Prebiotic supplementation in poultry diets selectively promotes beneficial cecal microorganisms, particularly Bifidobacterium and Lactobacillus, leading to increased short-chain fatty acid production and improved gut barrier function.
Probiotics, Prebiotics, and Phytogenic Substances for Optimizing Gut Health in Poultry.
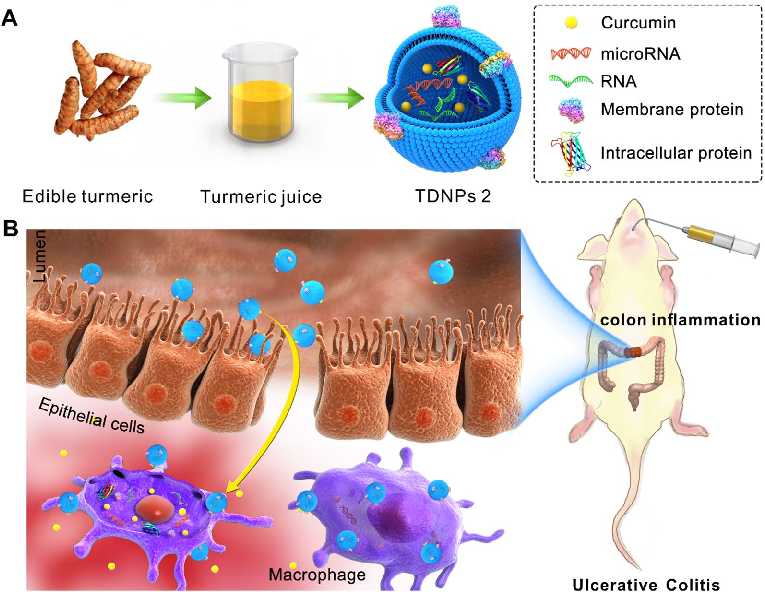
Turmeric-derived nanoparticles (TDNPs 2) are isolated through sucrose gradient ultracentrifugation and administered orally to target inflamed colonic tissue in a murine colitis model. The schematic outlines the isolation workflow from edible turmeric to purified nanovesicles.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
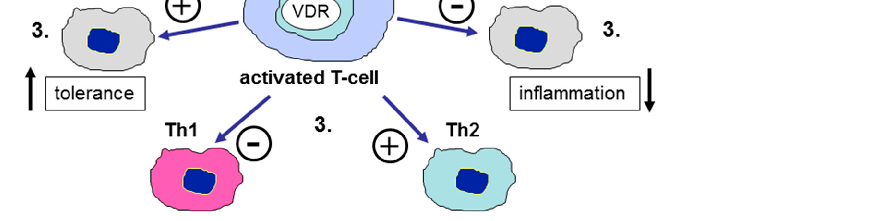
Vitamin D receptor expression and vitamin D-activating enzyme CYP27B1 are present in human colonic epithelial cells, enabling local production of active calcitriol. This paracrine signaling enhances tight junction integrity and antimicrobial defense at the mucosal surface.
The Role of Vitamin D in Inflammatory Bowel Disease: Mechanism to Management.
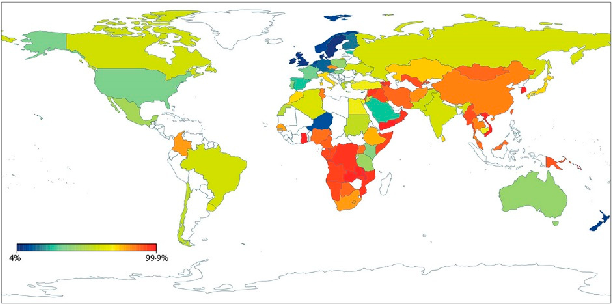
Geographic distribution of lactase non-persistence frequencies reveals wide variation across world populations, with highest persistence rates in Northern European and certain African and Middle Eastern pastoralist populations. East Asian and Native American populations show near-universal non-persistence.
Lactose Intolerance, Dairy Avoidance, and Treatment Options.
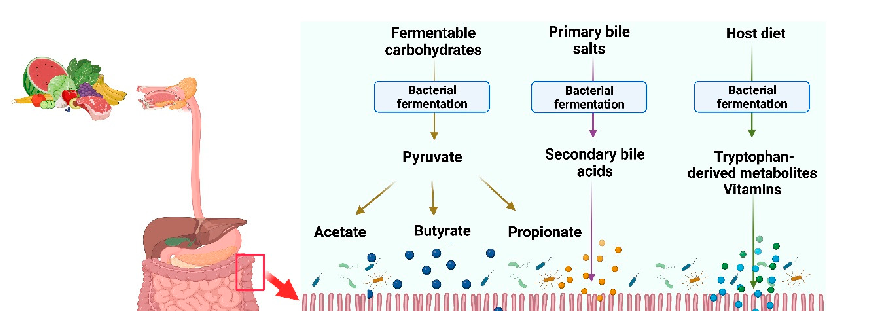
Butyrate modulates immune cell function in the gut mucosa, suppressing pro-inflammatory macrophage and dendritic cell activation while promoting regulatory T-cell differentiation. These immunomodulatory effects contribute to intestinal homeostasis.
Gut Microbial Metabolite Butyrate and Its Therapeutic Role in Inflammatory Bowel Disease: …
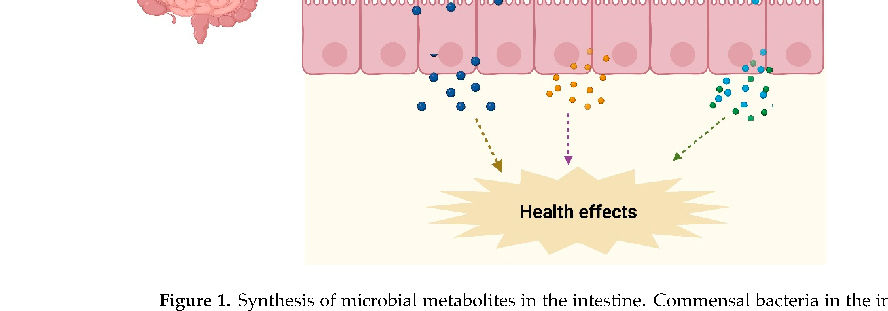
Dietary fiber fermentation pathways in the colon illustrate how complex carbohydrates are sequentially broken down by microbial consortia, ultimately yielding butyrate, propionate, and acetate as primary end products.
Gut Microbial Metabolite Butyrate and Its Therapeutic Role in Inflammatory Bowel Disease: …
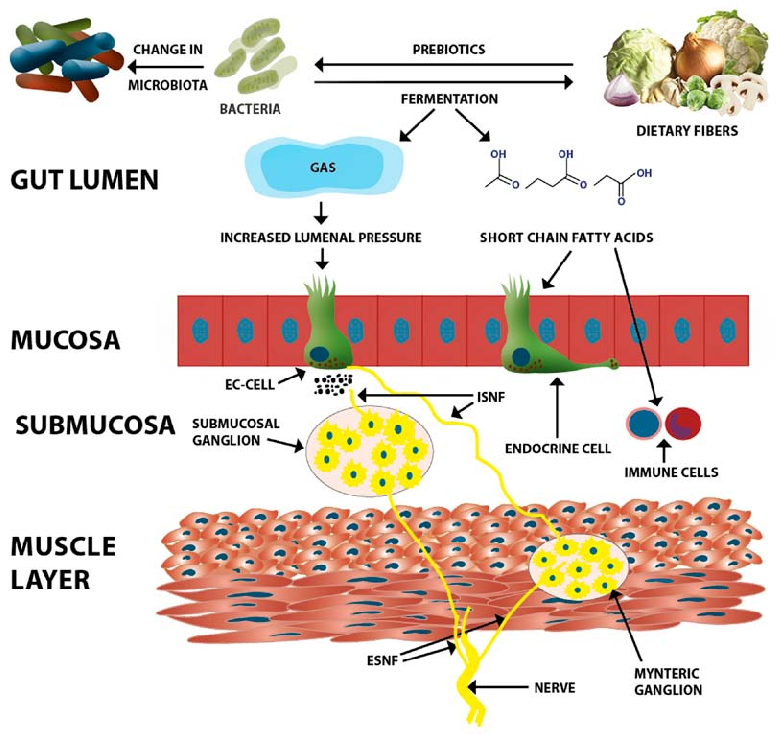
Dietary fiber acts as a prebiotic for intestinal microbiota, promoting growth of beneficial bacteria and production of short-chain fatty acids. This diagram illustrates multiple mechanisms through which fiber affects gastrointestinal function, including modulation of gut microbiome composition and intestinal barrier integrity.
Dietary fiber in irritable bowel syndrome (Review).
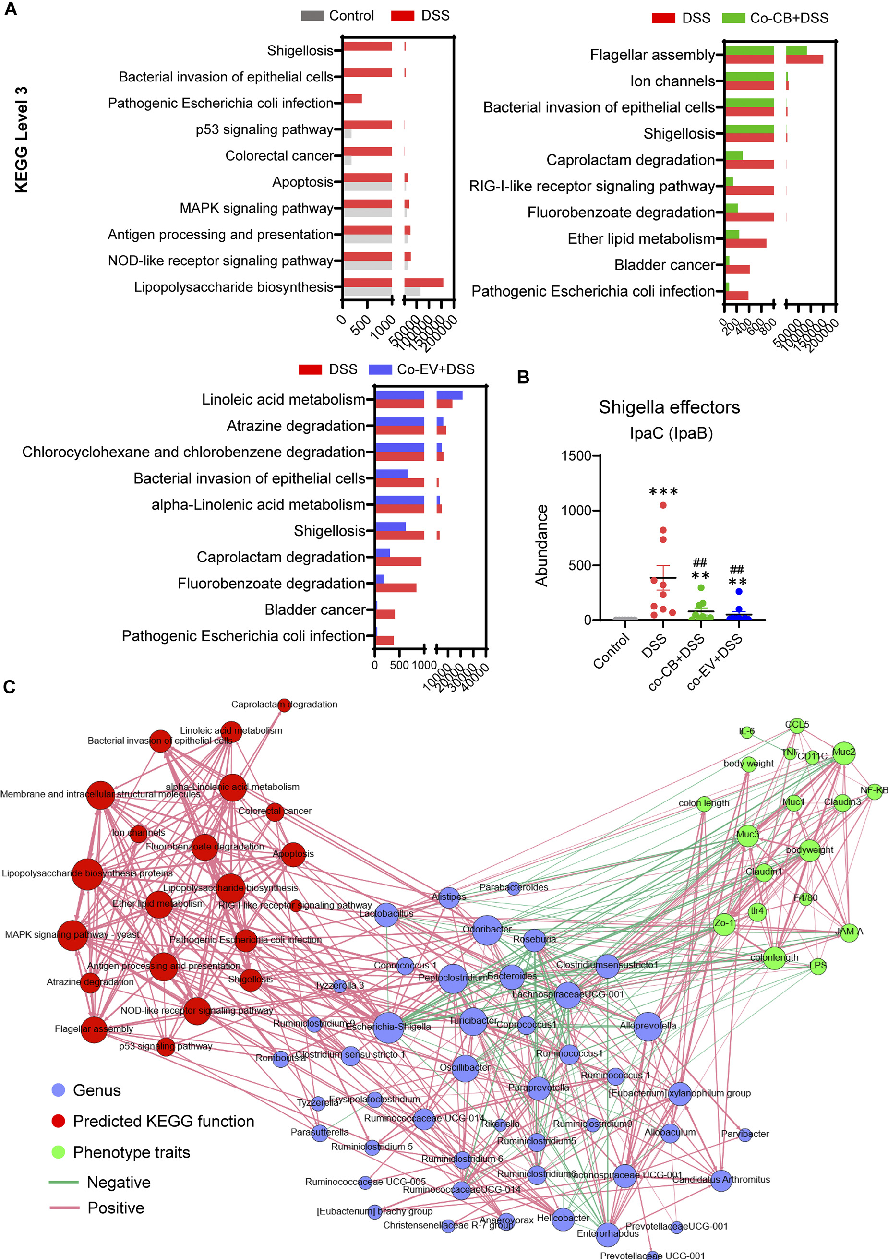
Comprehensive mechanistic model summarizing how C. butyricum and its extracellular vesicles modulate gut homeostasis. This figure integrates findings on barrier function, immune modulation, and microbiome changes in experimental colitis.
Clostridium butyricum and Its Derived Extracellular Vesicles Modulate Gut Homeostasis and Ameliorate …
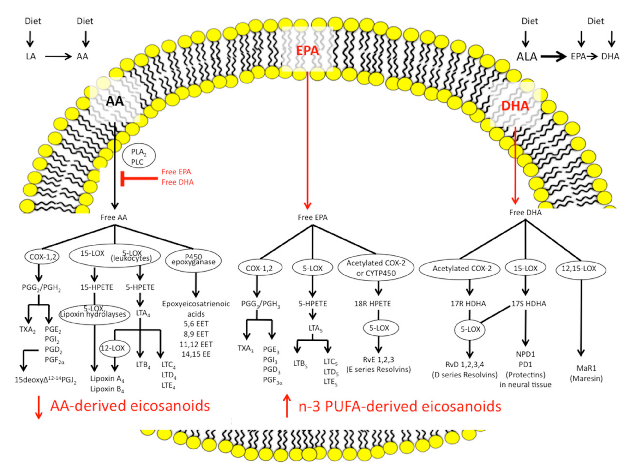
N-3 and n-6 polyunsaturated fatty acids follow distinct biosynthetic pathways to produce eicosanoids and specialized pro-resolving mediators. This diagram outlines the synthesis routes from alpha-linolenic acid and arachidonic acid through COX and cytochrome P450 enzymes to their bioactive lipid products.
The Imbalance between n-6/n-3 Polyunsaturated Fatty Acids and Inflammatory Bowel Disease: A …
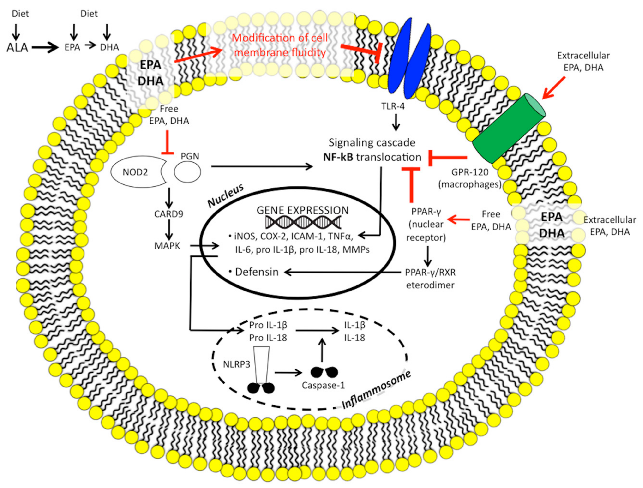
N-3 polyunsaturated fatty acids modulate intestinal innate immunity through multiple receptor and signaling targets. This diagram maps the interactions of DHA and EPA with G-protein coupled receptors, COX enzymes, and caspase recognition domains in intestinal immune cells.
The Imbalance between n-6/n-3 Polyunsaturated Fatty Acids and Inflammatory Bowel Disease: A …
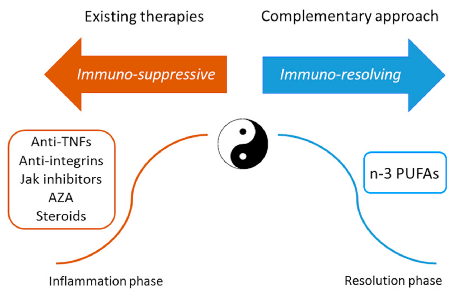
Current IBD treatments employ both immuno-suppressive and immuno-resolving strategies. This diagram contrasts conventional therapies (azathioprine, anti-TNF agents) with emerging PUFA-based approaches that promote active resolution of intestinal inflammation.
The Imbalance between n-6/n-3 Polyunsaturated Fatty Acids and Inflammatory Bowel Disease: A …
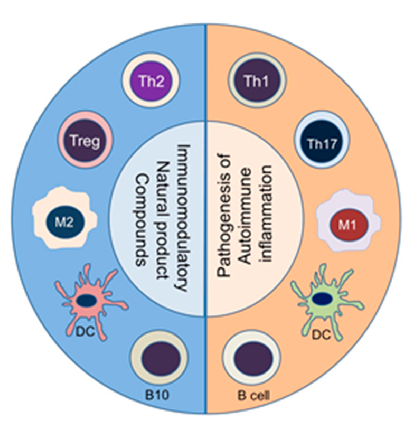
Natural product compounds shift the balance between pathogenic pro-inflammatory immune cells (right panel) and protective anti-inflammatory/immunomodulatory cell subsets (left panel). This diagram illustrates how diverse natural products can rebalance immune cell populations in autoimmune disease.
The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation.
Clinical translation of natural anti-inflammatory products requires understanding of bioavailability and dose-response relationships. This figure discusses the therapeutic potential and limitations of natural product-based interventions for autoimmune conditions.
The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation.
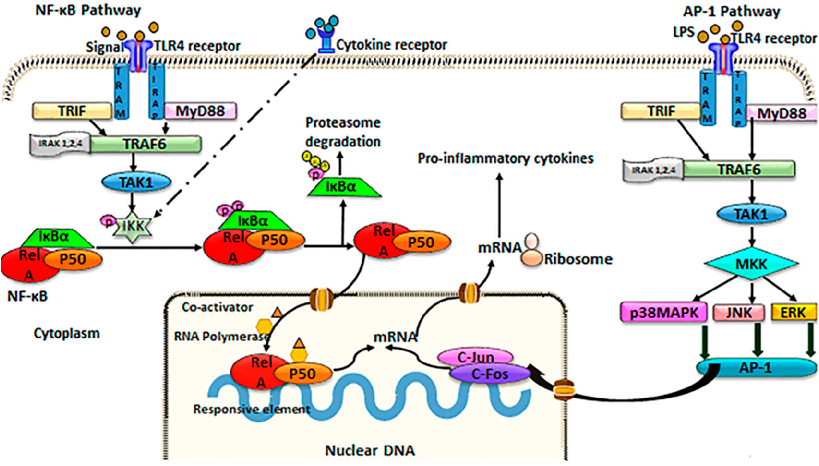
TLR4/NF-kB/AP-1 signaling is a key inflammatory pathway in intestinal disease. This schematic illustrates the cascade from TLR4 receptor activation through NF-kB and AP-1 transcription factors to pro-inflammatory gene expression, and indicates where curcumin may intervene.
Curcumin and Intestinal Inflammatory Diseases: Molecular Mechanisms of Protection.
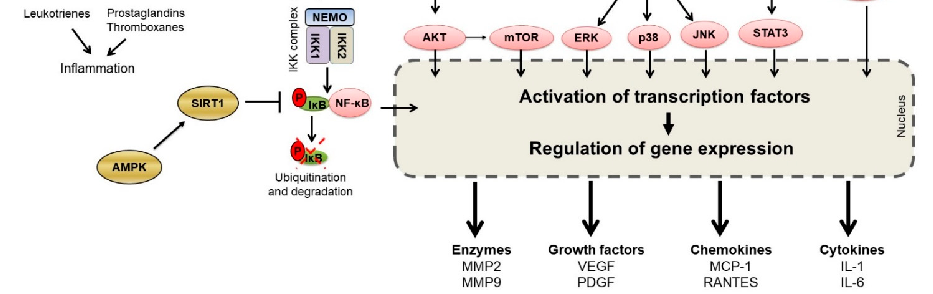
Inflammatory bowel diseases are associated with activation of the NF-kB signaling pathway. This diagram illustrates how various phytochemicals may counteract NF-kB pathway activation through inhibition of Toll-like receptors, IKK complex, and downstream transcriptional targets in IBD.
Phytochemicals and Regulation of NF-kB in Inflammatory Bowel Diseases: An Overview of …
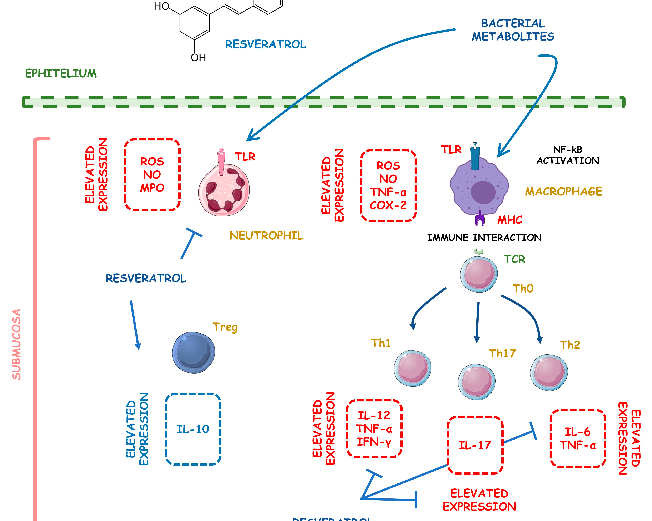
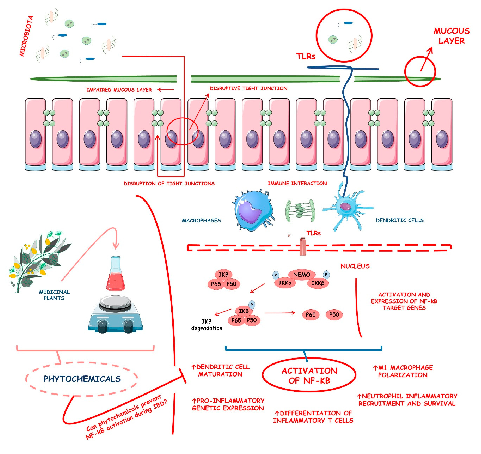
Supplementary figure from a comprehensive review of phytochemical effects on NF-kB regulation in inflammatory bowel disease. The review examines multiple plant-derived compounds with potential to attenuate chronic intestinal inflammation through modulation of key signaling pathways.
Phytochemicals and Regulation of NF-kB in Inflammatory Bowel Diseases: An Overview of …
3페이지 중 1페이지