Processo di ricerca
477 figure da ricerca revisionata da esperti

Gene expression profiles for inflammatory mediators in LPS-stimulated intestinal cells are compared across varying butyrate concentrations, revealing a clear anti-inflammatory dose response.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …

Cell viability assays confirm that butyrate concentrations used in the inflammation experiments are non-cytotoxic, validating the specificity of observed anti-inflammatory effects.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
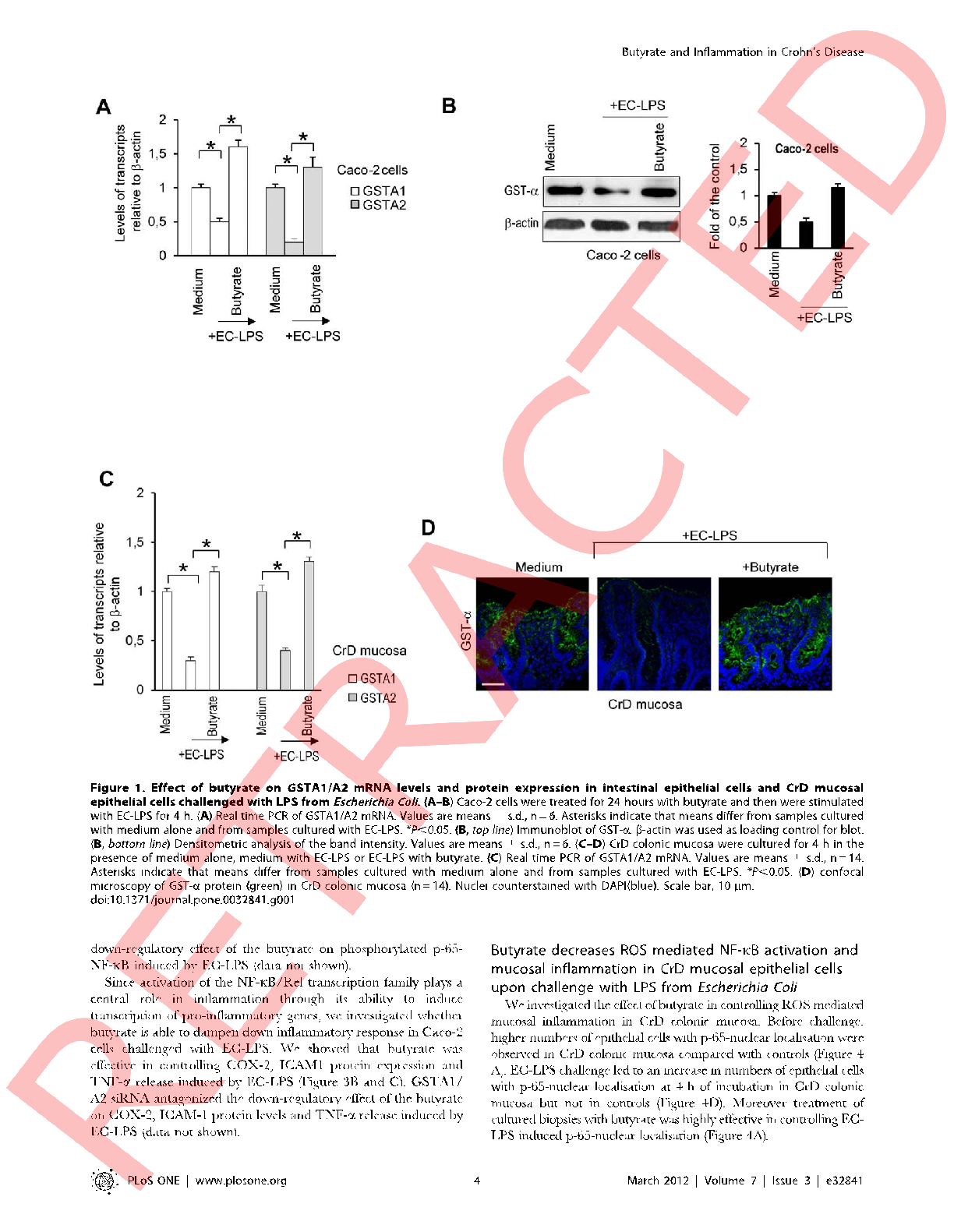
Transepithelial electrical resistance measurements across intestinal cell monolayers demonstrate butyrate's capacity to maintain epithelial integrity during inflammatory challenge.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
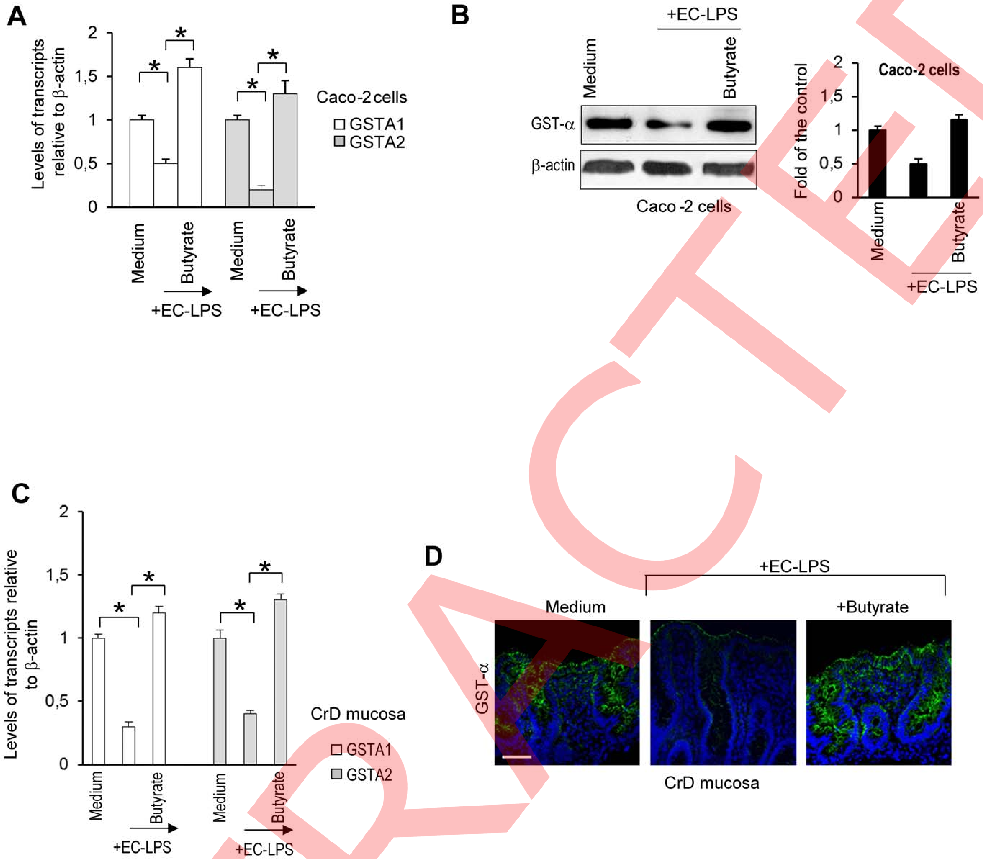
Western blot analysis of phosphorylated signaling proteins in LPS-treated intestinal cells shows reduced inflammatory cascade activation in the presence of butyrate.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
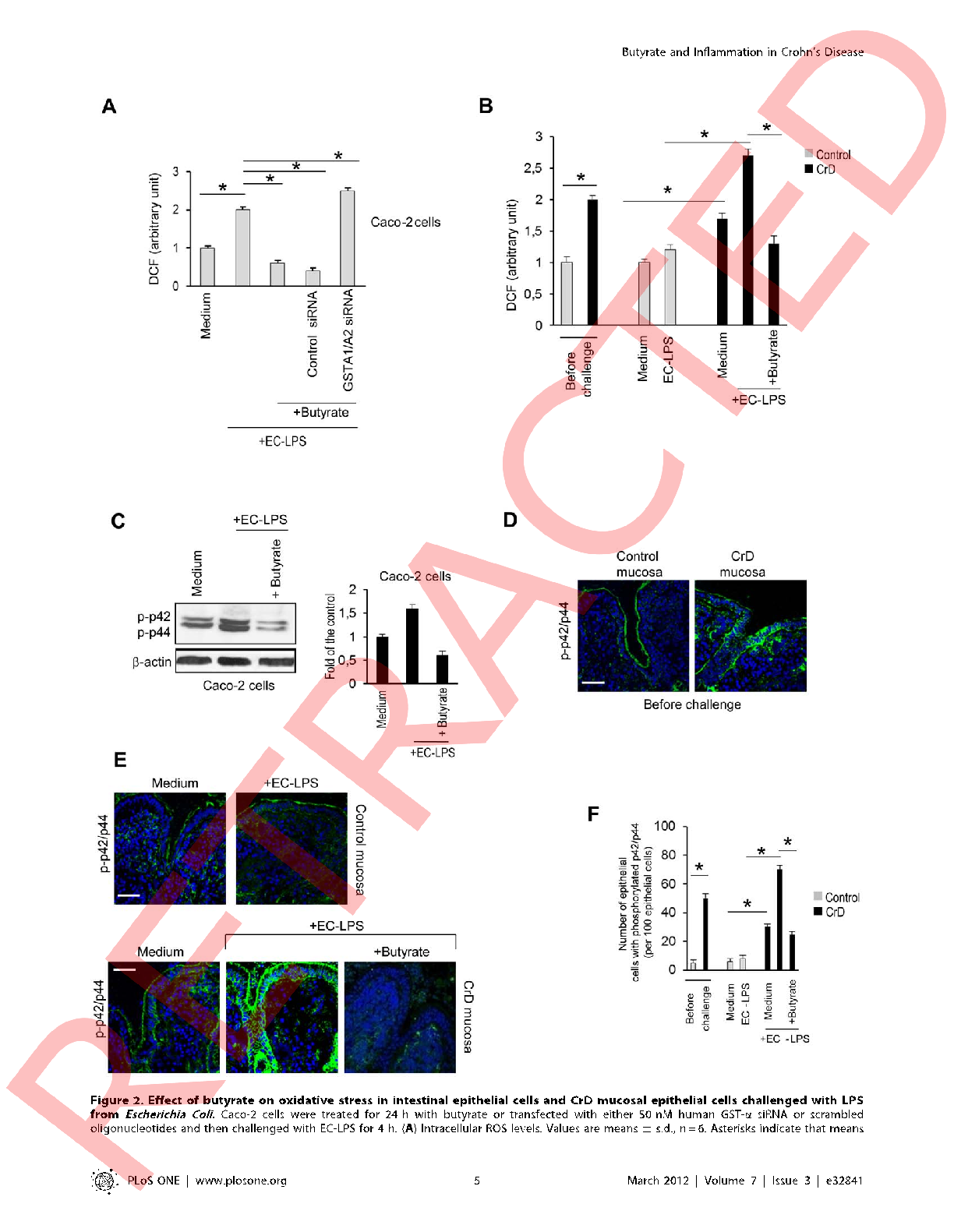
Cytokine secretion profiles including IL-6, IL-8, and TNF-α are quantified in cell culture supernatants, with butyrate-treated cells showing significantly attenuated inflammatory responses.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
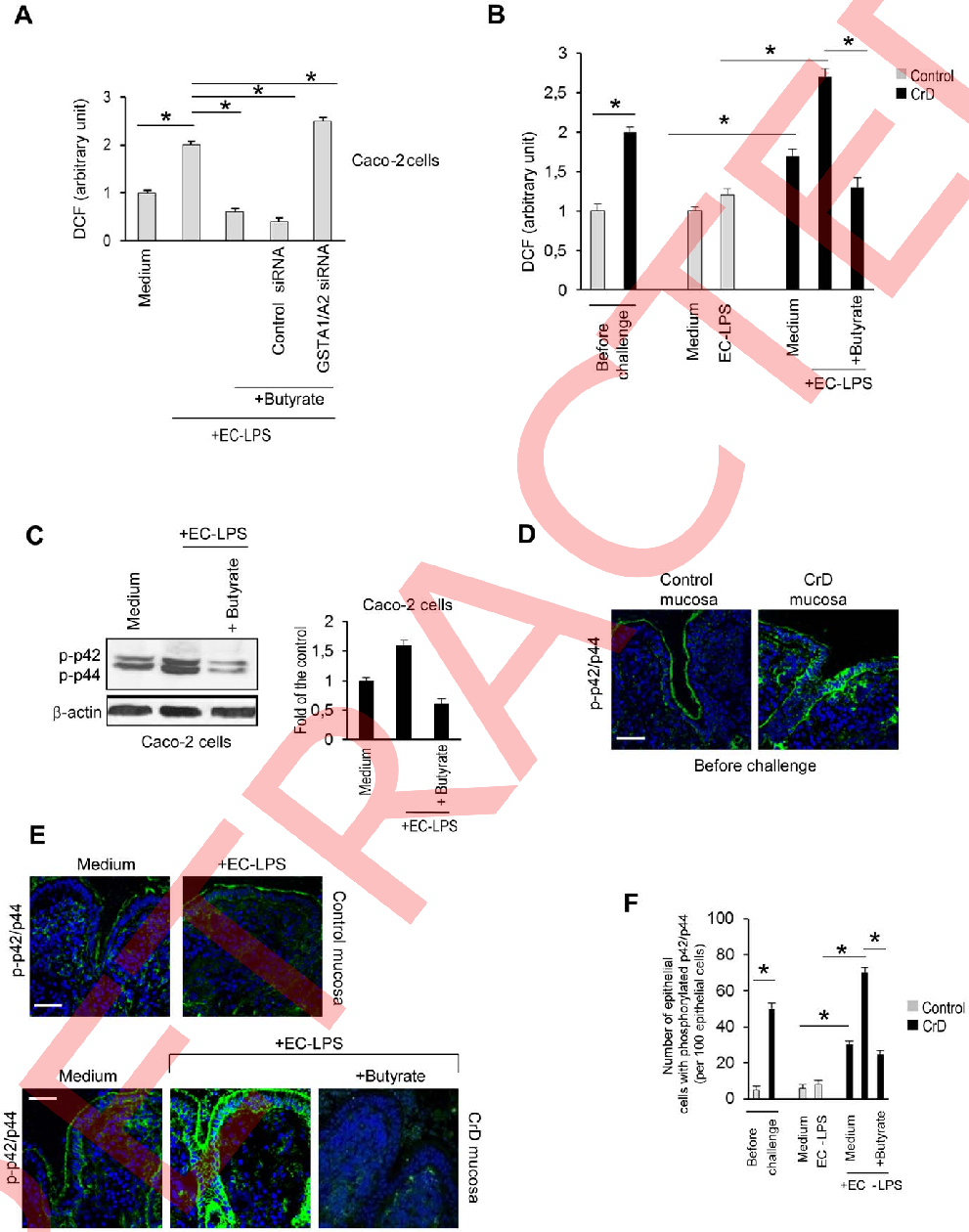
Histone deacetylase activity in intestinal epithelial cells is measured following butyrate exposure, linking epigenetic modulation to the observed anti-inflammatory phenotype.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
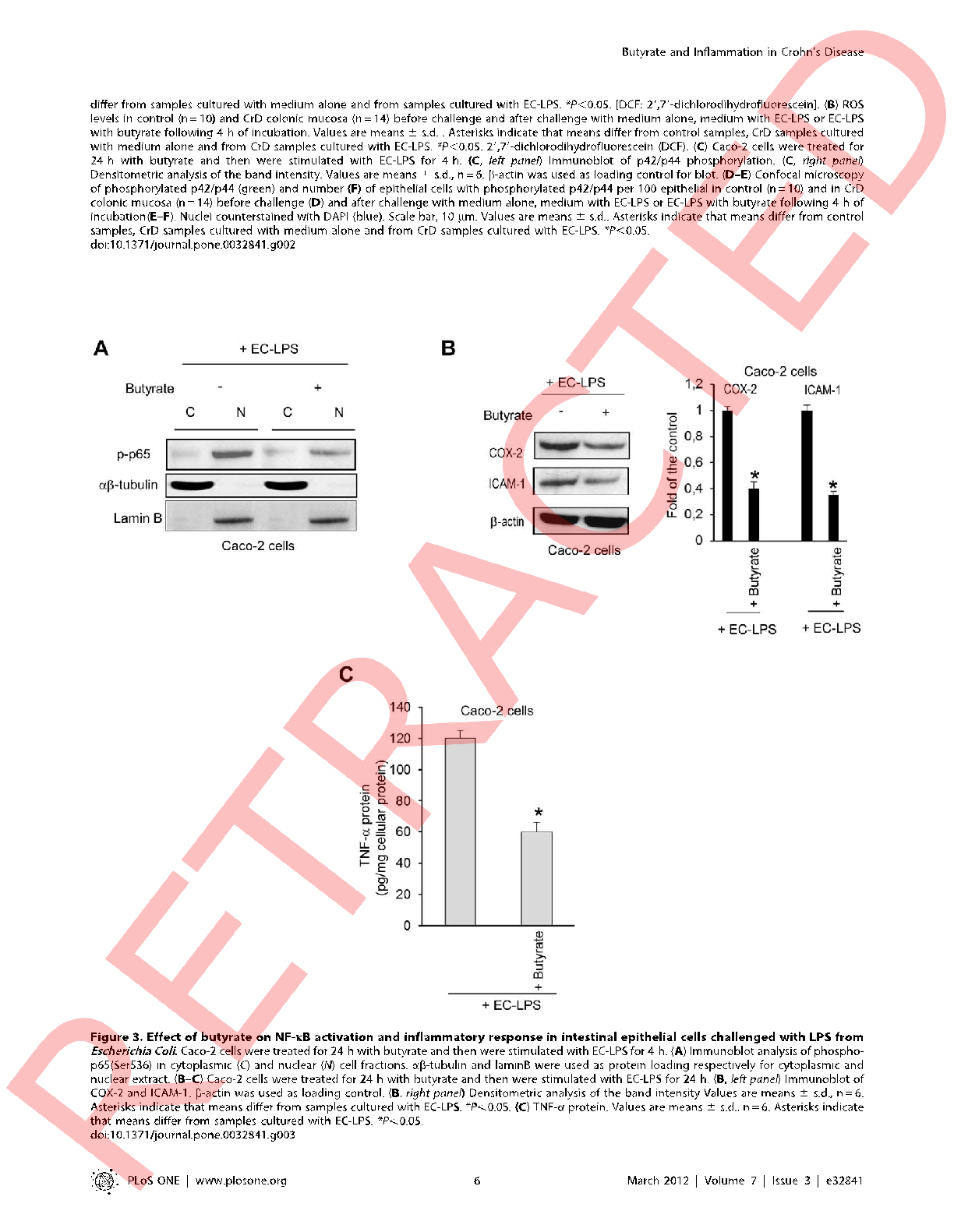
Intracellular reactive oxygen species levels are visualized in LPS-challenged intestinal cells with and without butyrate treatment, showing reduced oxidative stress in supplemented cultures.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
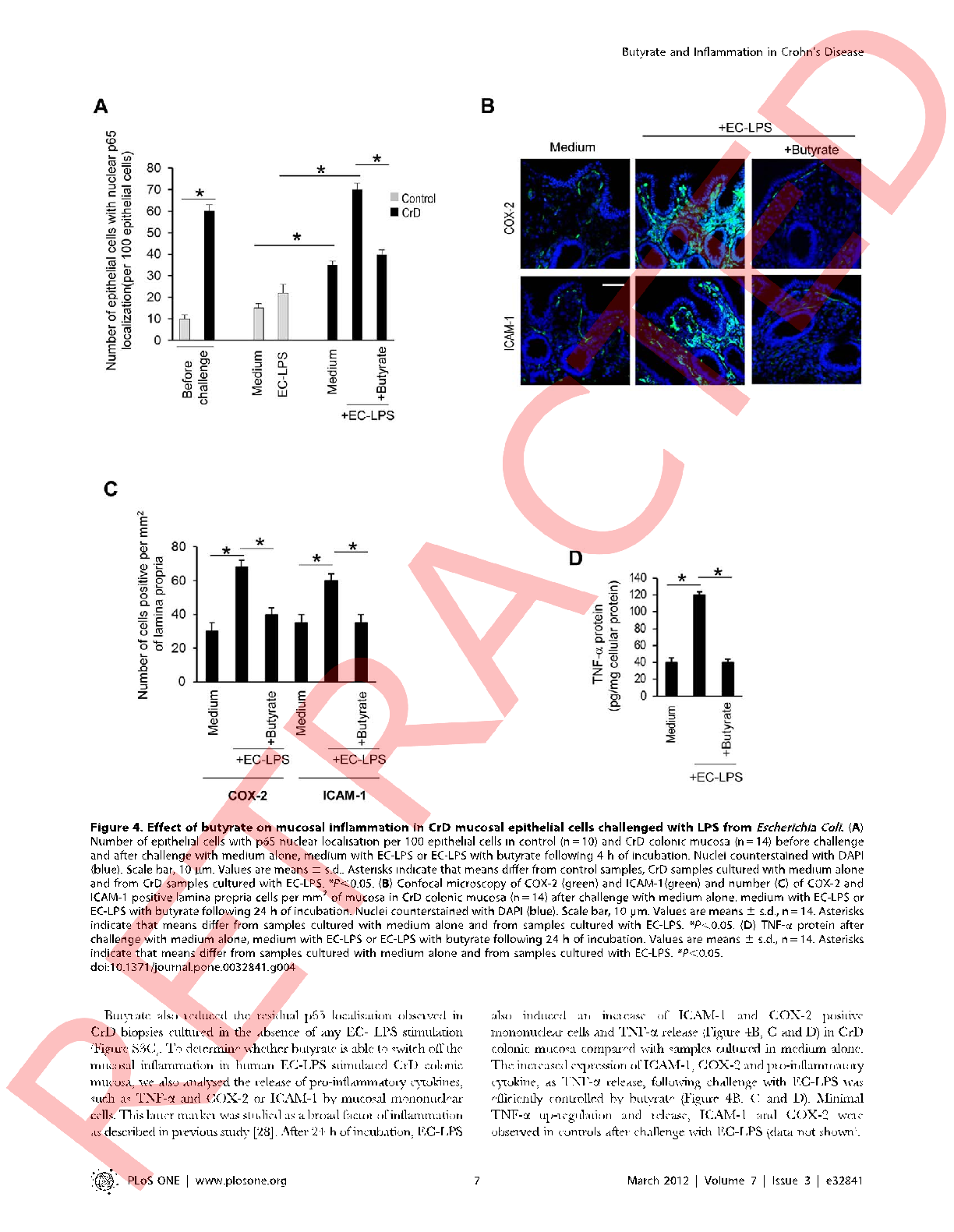
Prostaglandin E2 production by intestinal cells under inflammatory conditions is attenuated by butyrate, as measured by ELISA across multiple treatment concentrations.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
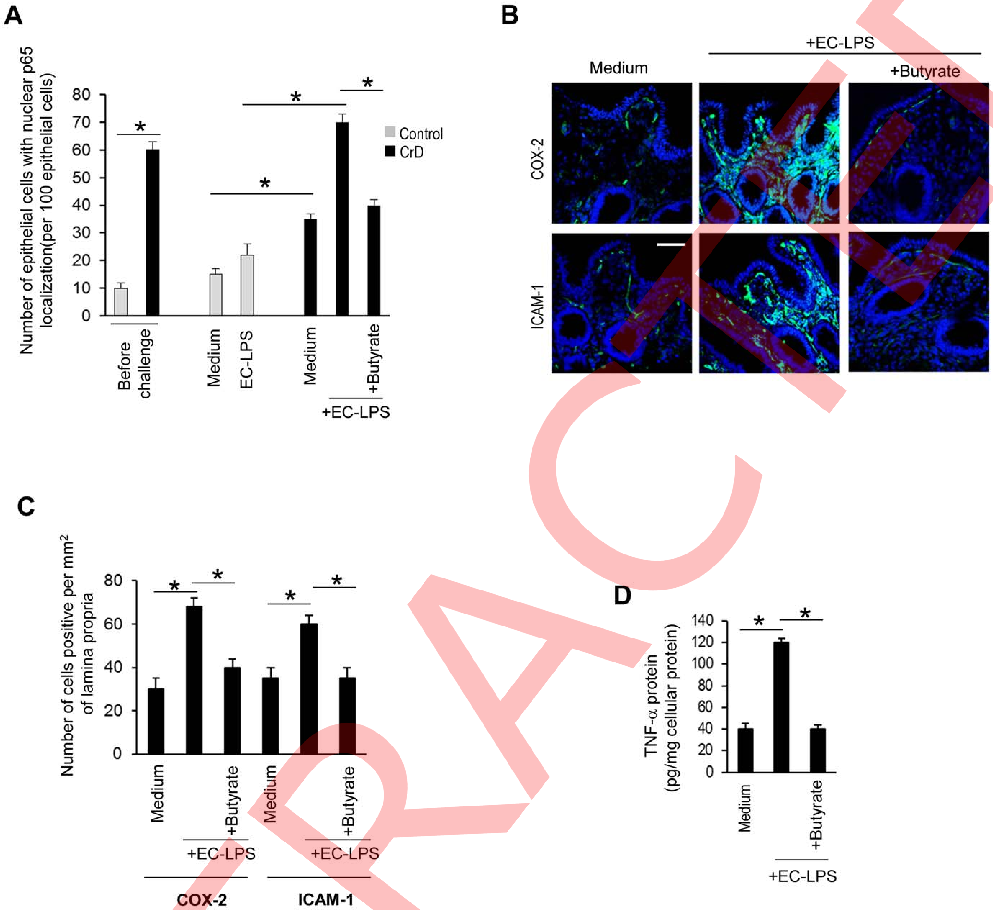
Apoptosis rates in intestinal epithelial cells exposed to LPS are compared between butyrate-treated and control conditions, indicating a cytoprotective effect of the short-chain fatty acid.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …

mRNA expression of tight junction components claudin-1 and occludin is upregulated in butyrate-treated intestinal cells, supporting enhanced paracellular barrier function.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …

Permeability assays using fluorescent dextran tracers demonstrate reduced paracellular flux in butyrate-pretreated intestinal monolayers challenged with LPS.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …

Butyrate's anti-inflammatory effects on intestinal epithelial cells stimulated with lipopolysaccharide are quantified, showing dose-dependent suppression of pro-inflammatory cytokine release.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
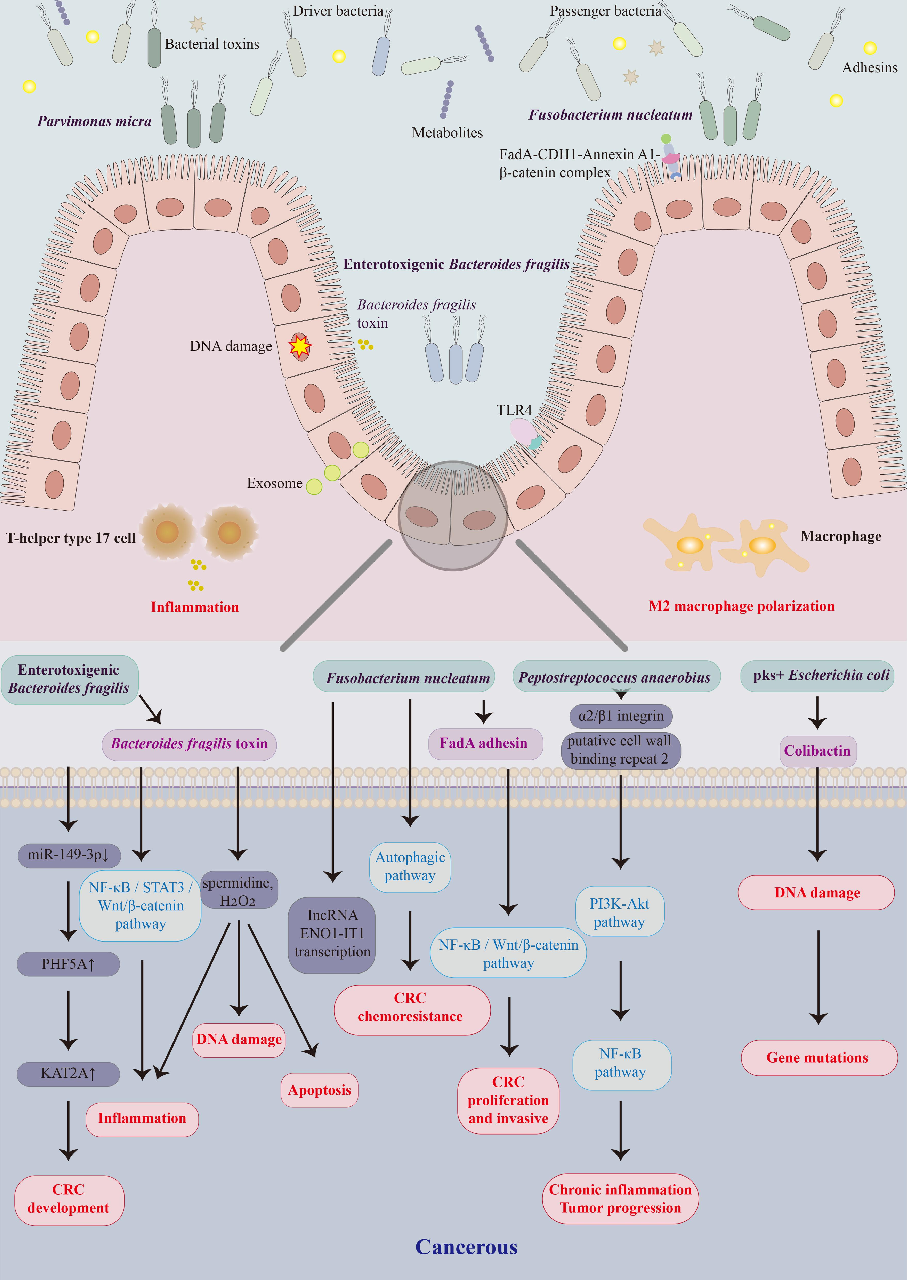
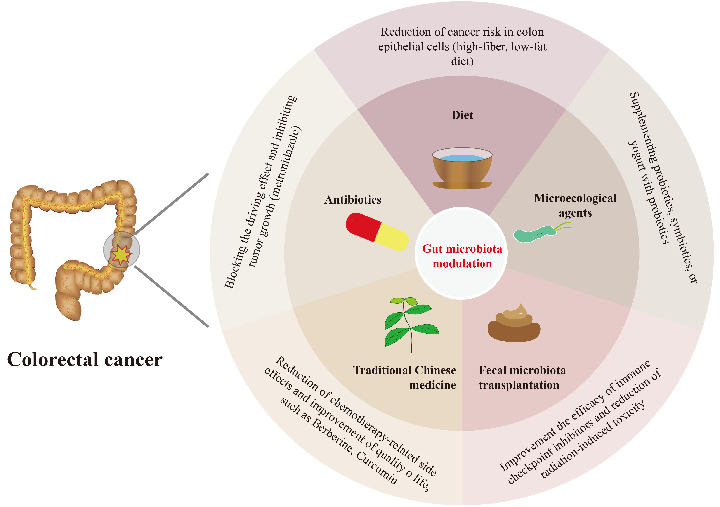
Gut microbiota alterations associated with colorectal cancer in Chinese patient cohorts are characterized, identifying specific bacterial taxa enriched or depleted in tumor-bearing individuals.
Colorectal cancer and gut microbiota studies in China.
Microbial community composition differences between colorectal cancer patients and healthy controls in Chinese studies are visualized, highlighting potential biomarker species.
Colorectal cancer and gut microbiota studies in China.
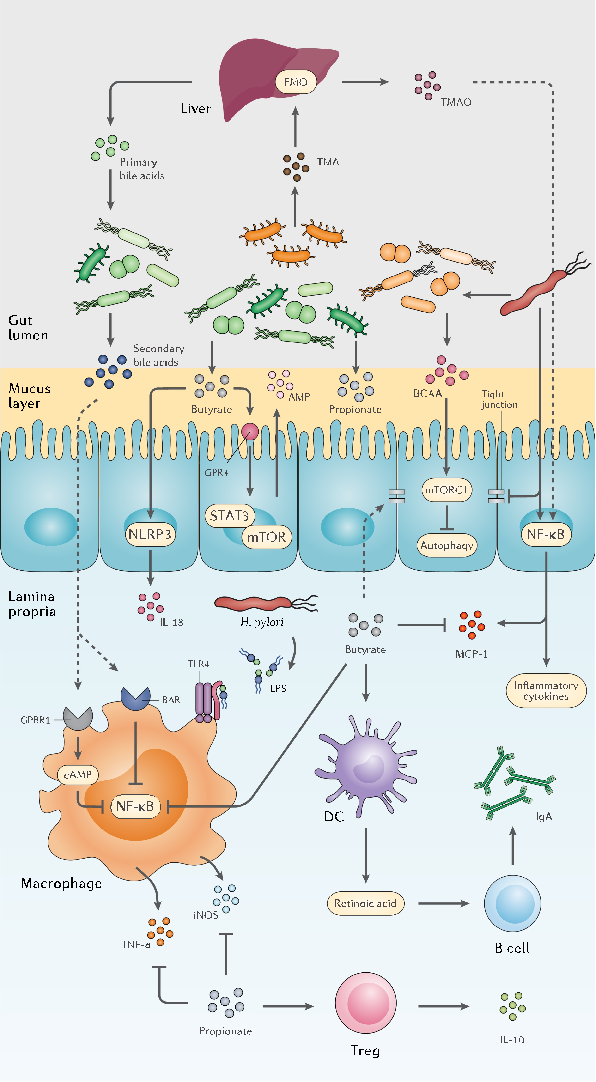
Specific bacterial taxa responsive to probiotic treatment are identified, showing differential abundance patterns that suggest strain-specific effects on gut ecosystem structure.
The double-edged sword of probiotic supplementation on gut microbiota structure in Helicobacter …
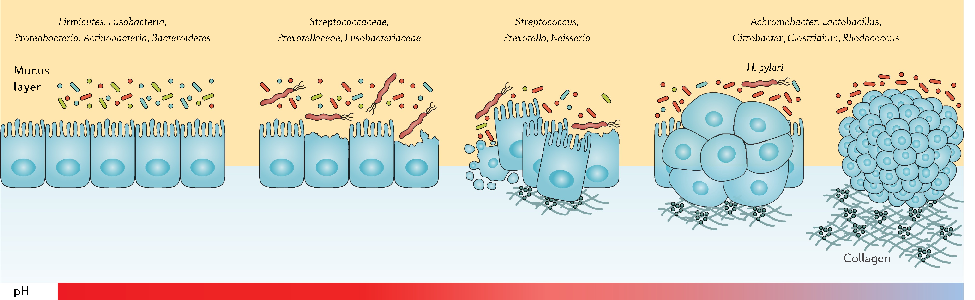
The dual nature of probiotic effects on established gut communities is depicted, with some participants showing enhanced diversity while others experience transient dysbiosis.
The double-edged sword of probiotic supplementation on gut microbiota structure in Helicobacter …
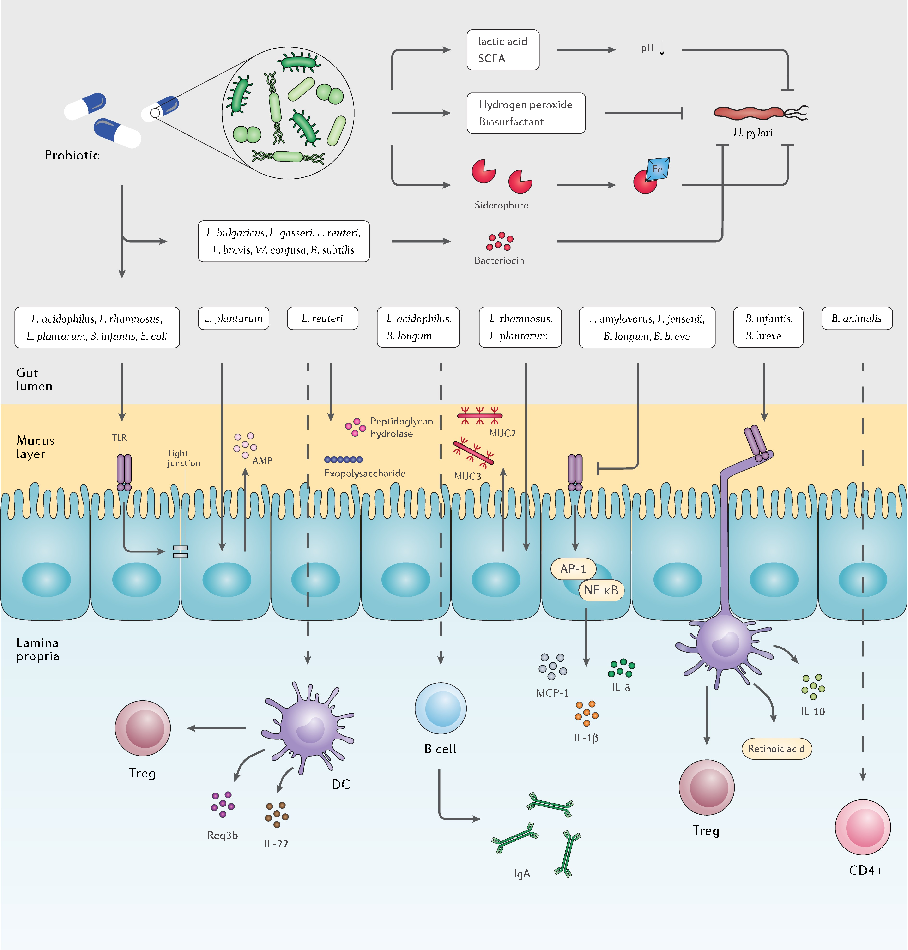
Gut microbiota structural changes following probiotic supplementation are characterized, revealing both beneficial shifts and potential disruptions depending on the host's baseline microbial composition.
The double-edged sword of probiotic supplementation on gut microbiota structure in Helicobacter …
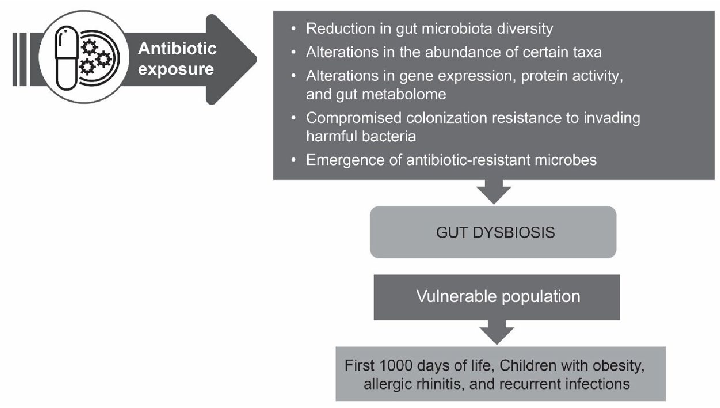
Approaches for managing antibiotic-associated dysbiosis are summarized, including strategies to restore gut microbial diversity after antibiotic treatment. The figure outlines interventions such as probiotics and microbiota-targeted therapies that may help mitigate dysbiosis in populations predisposed to gut disruption.
Current understanding of antibiotic-associated dysbiosis and approaches for its management.
Illustration of Saccharomyces boulardii CNCM I-745 mechanisms of action in the intestinal lumen, showing direct antimicrobial effects and pathogen binding at the gut epithelial surface.
Diversity of Saccharomyces boulardii CNCM I-745 mechanisms of action against intestinal infections.
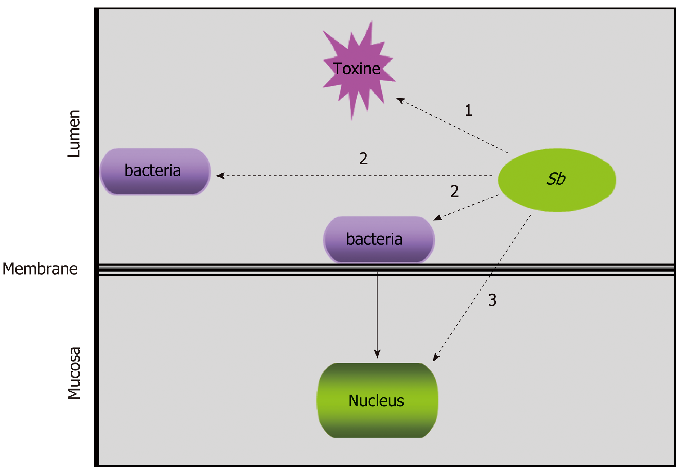
Schematic depicting how S. boulardii CNCM I-745 modulates intestinal epithelial barrier function, including tight junction protein preservation and mucus secretion enhancement.
Diversity of Saccharomyces boulardii CNCM I-745 mechanisms of action against intestinal infections.
Schematic depicting how S. boulardii CNCM I-745 modulates intestinal epithelial barrier function, including tight junction protein preservation and mucus secretion enhancement.
Diversity of Saccharomyces boulardii CNCM I-745 mechanisms of action against intestinal infections.
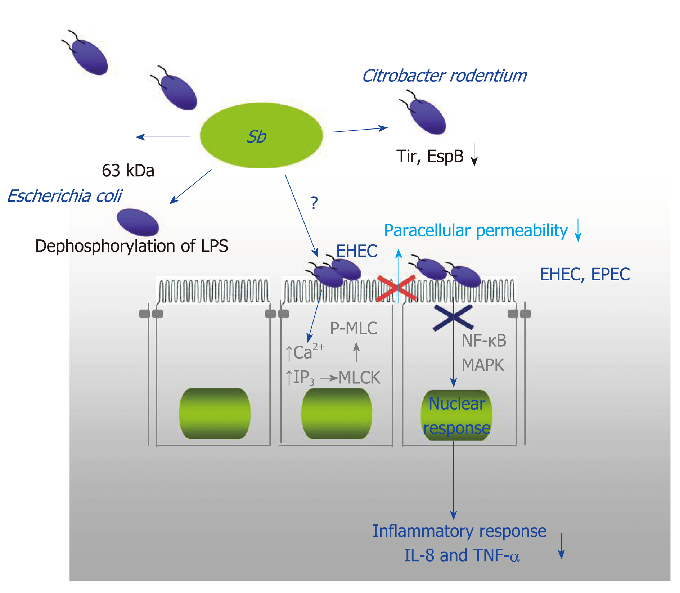
Diagram of S. boulardii's immunomodulatory actions in the gut, illustrating effects on secretory IgA production, anti-inflammatory cytokine release, and immune cell regulation.
Diversity of Saccharomyces boulardii CNCM I-745 mechanisms of action against intestinal infections.
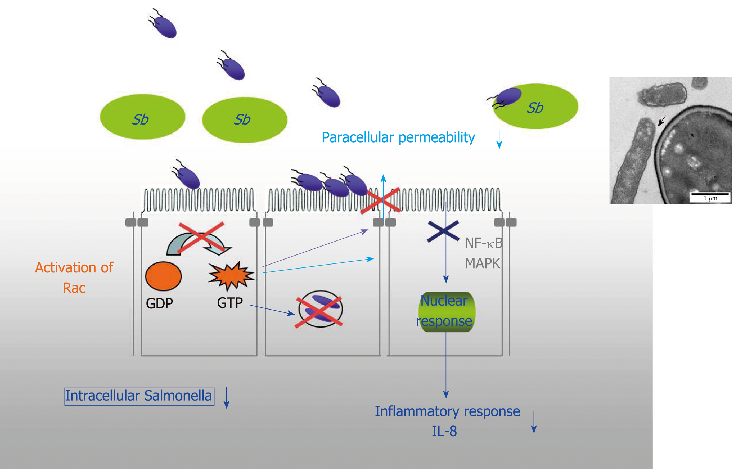
Diagram of S. boulardii's immunomodulatory actions in the gut, illustrating effects on secretory IgA production, anti-inflammatory cytokine release, and immune cell regulation.
Diversity of Saccharomyces boulardii CNCM I-745 mechanisms of action against intestinal infections.
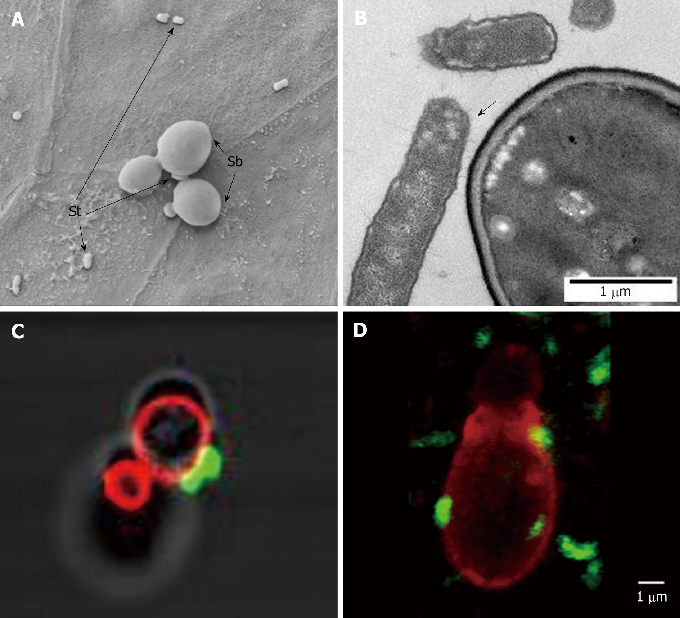
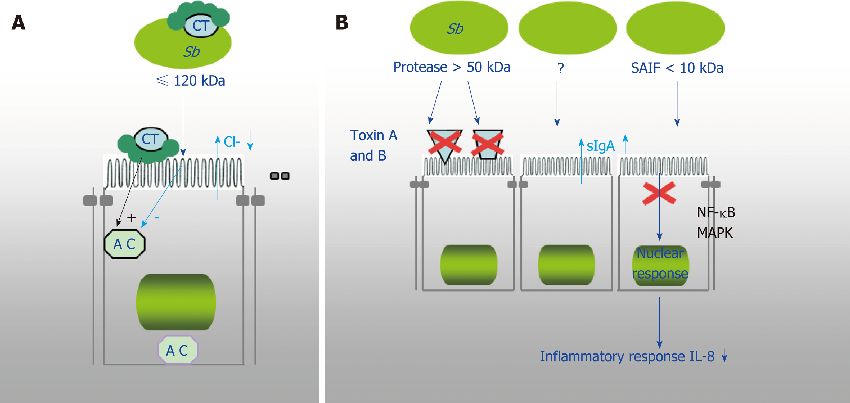
Evidence summary for S. boulardii CNCM I-745 efficacy against specific intestinal pathogens including Clostridium difficile, showing toxin neutralization and receptor binding inhibition mechanisms.
Diversity of Saccharomyces boulardii CNCM I-745 mechanisms of action against intestinal infections.
Pagina 6 di 20