अनुसंधान प्रक्रिया
477 सहकर्मी-समीक्षित शोध से आंकड़े
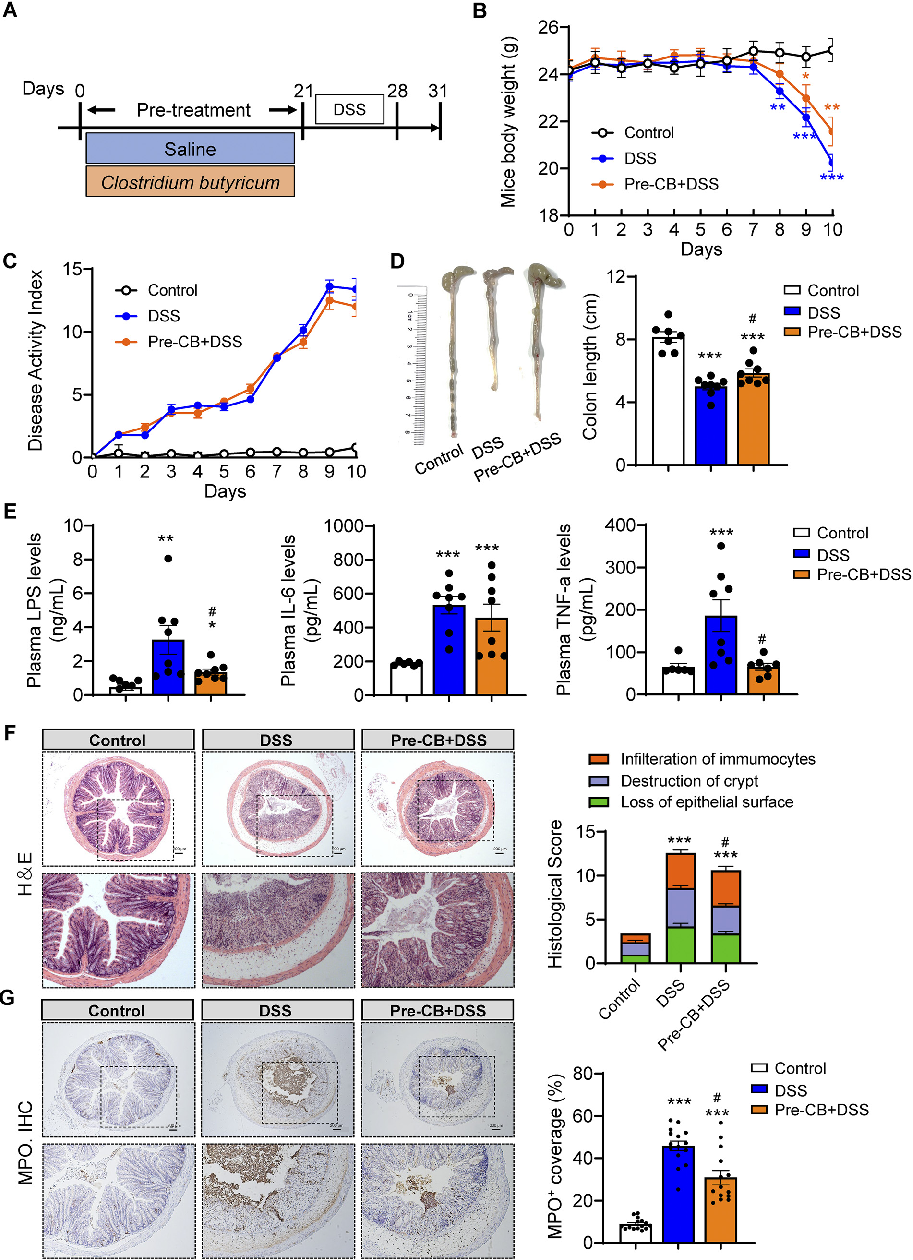
Histological analysis of colonic tissue reveals the extent of inflammatory infiltration and mucosal damage. This figure compares histopathological scores across treatment groups in the acute experimental colitis model.
Clostridium butyricum and Its Derived Extracellular Vesicles Modulate Gut Homeostasis and Ameliorate …
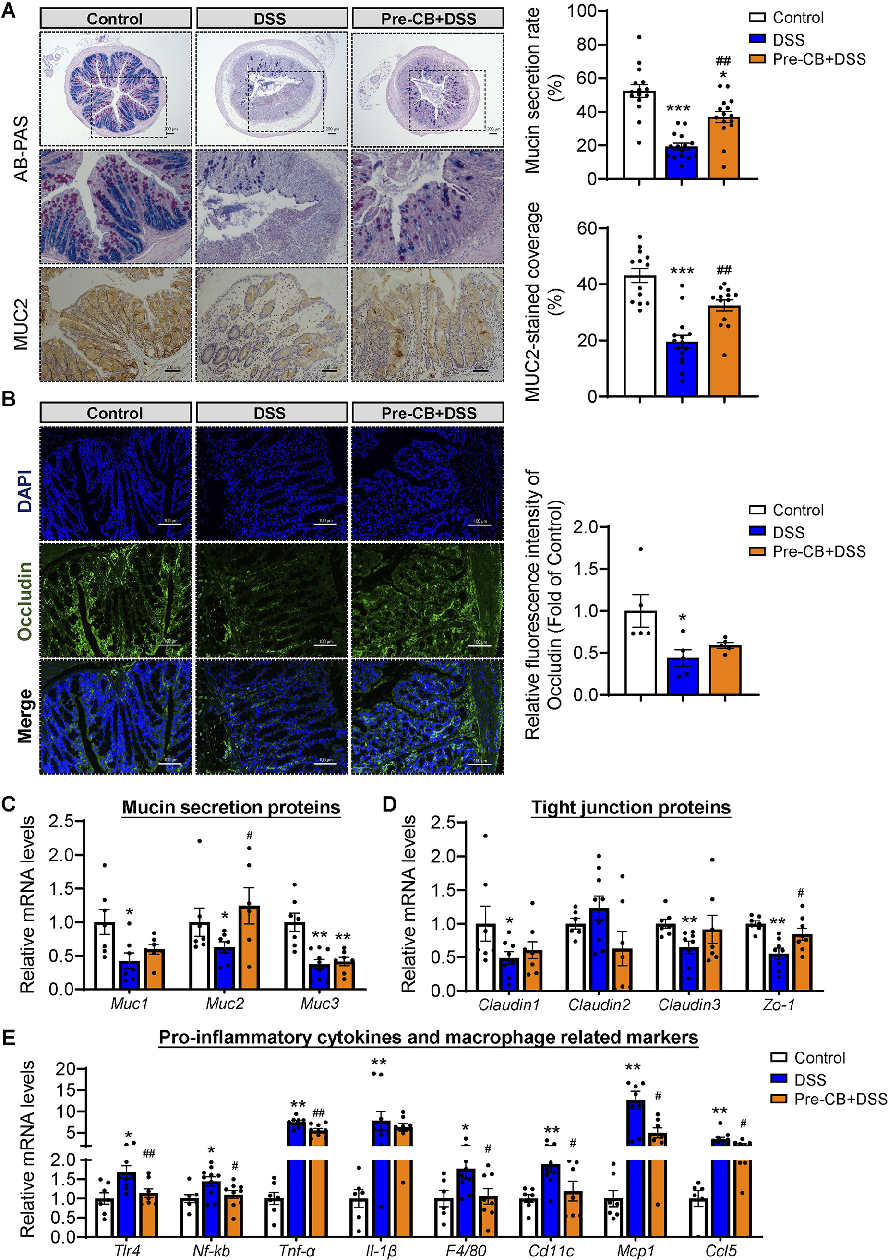
Pro-inflammatory cytokine levels in colonic tissue were quantified to assess the anti-inflammatory effects of C. butyricum treatment. This figure presents TNF-alpha, IL-6, and IL-1beta expression data across experimental groups.
Clostridium butyricum and Its Derived Extracellular Vesicles Modulate Gut Homeostasis and Ameliorate …
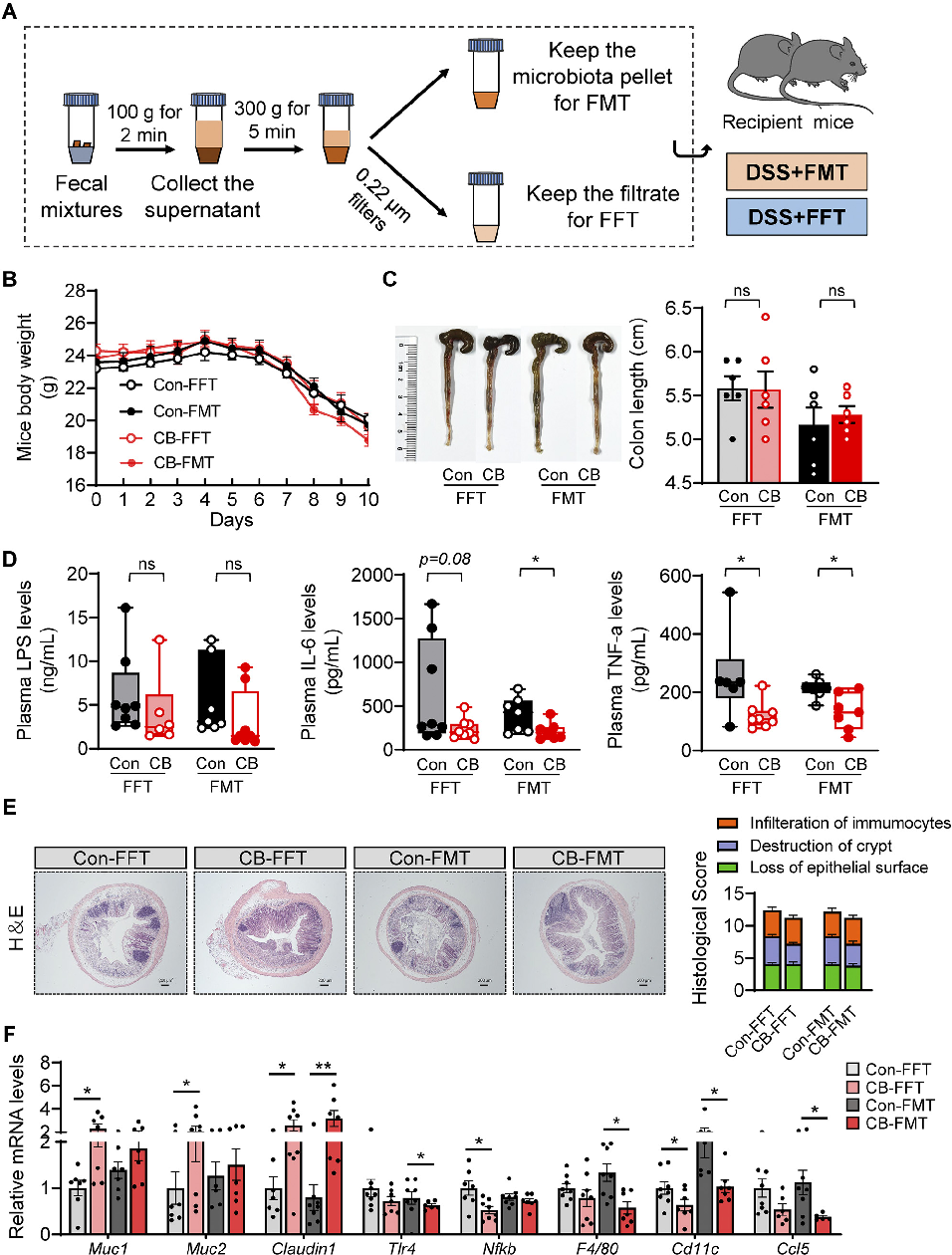
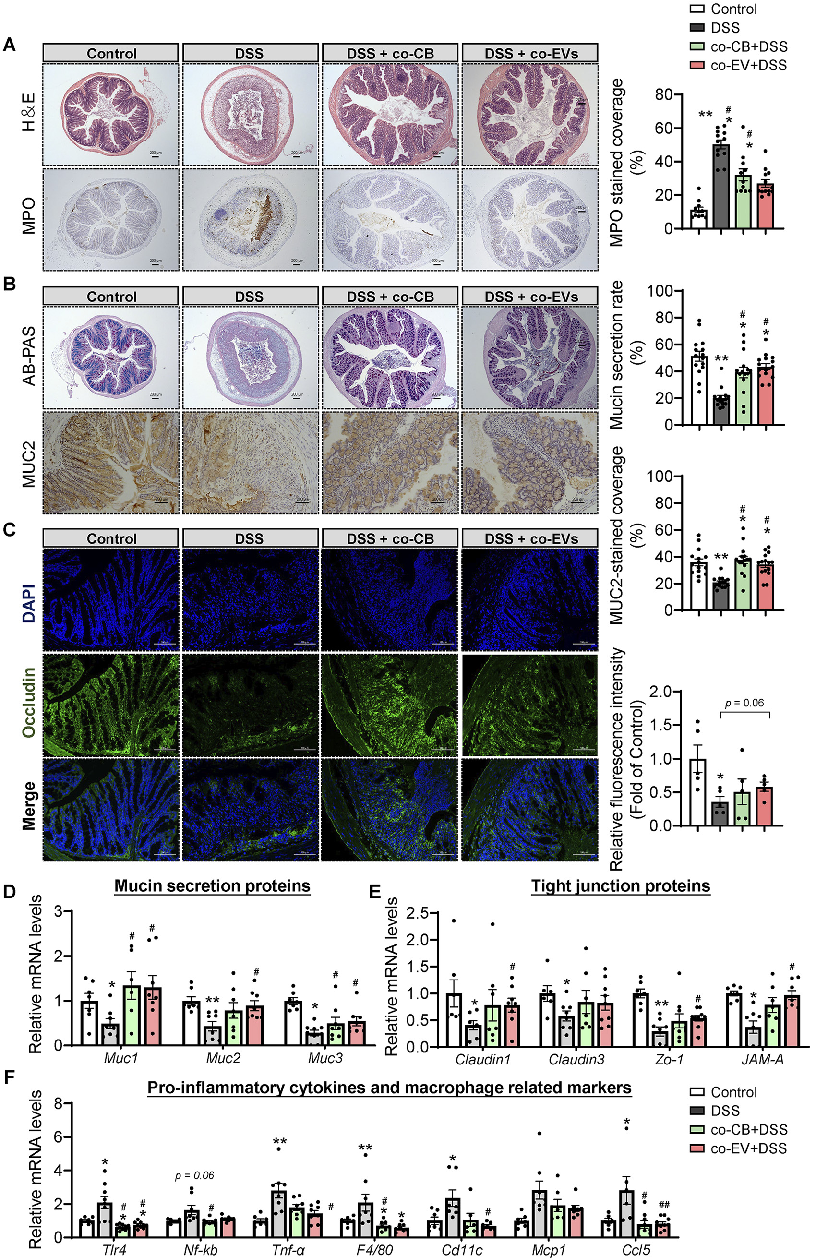
Tight junction protein expression indicates intestinal barrier integrity. This figure shows changes in occludin, claudin, and ZO-1 levels in colonic tissue following C. butyricum or vesicle treatment.
Clostridium butyricum and Its Derived Extracellular Vesicles Modulate Gut Homeostasis and Ameliorate …
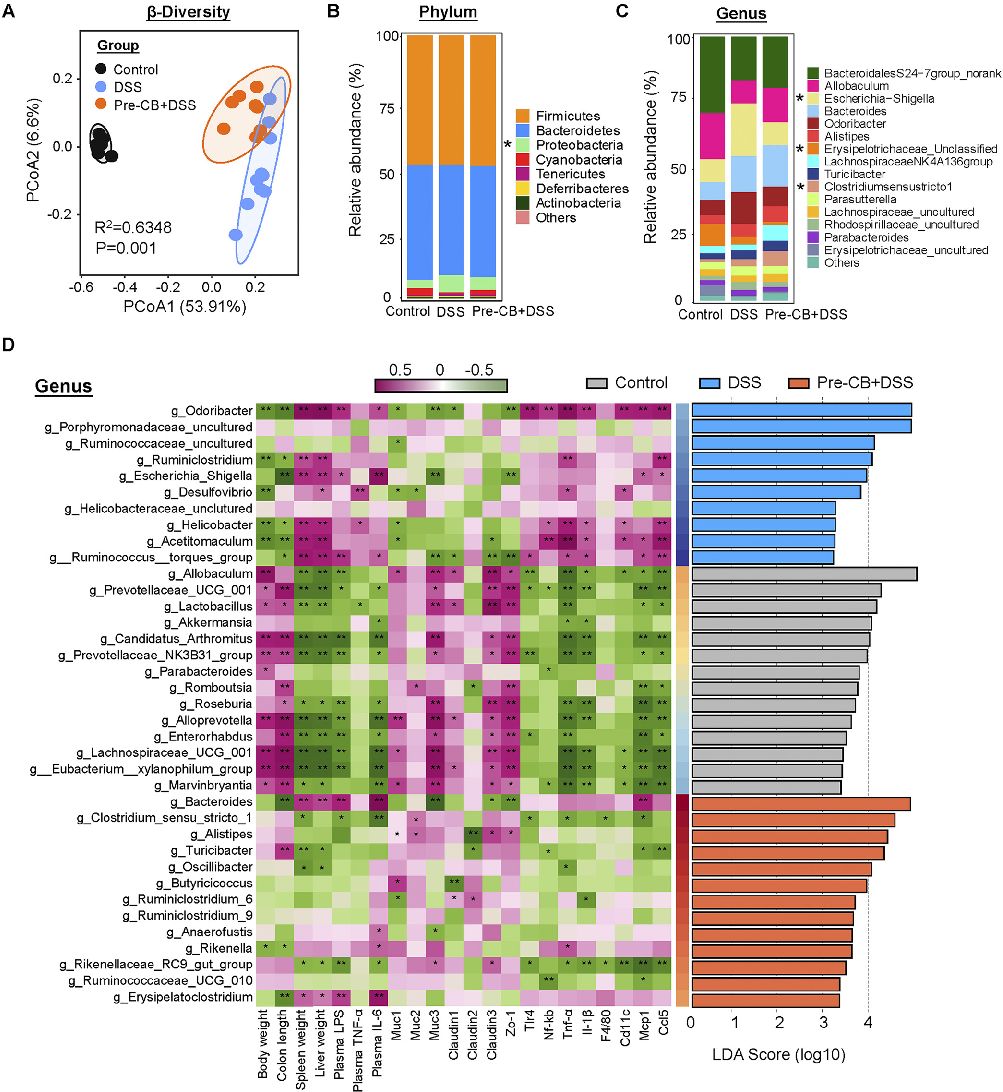
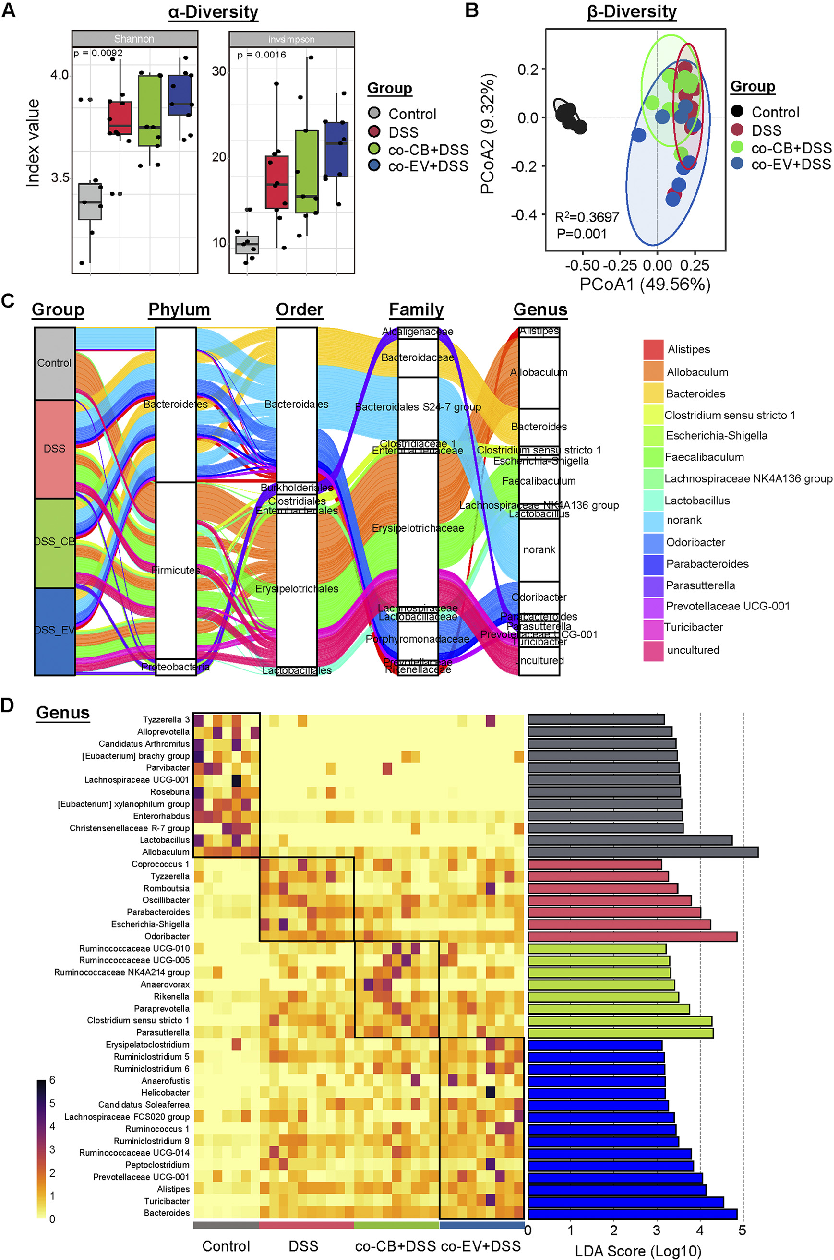
Gut microbiome composition was analyzed to determine how C. butyricum treatment modulates the intestinal microbial community. This figure presents 16S rRNA sequencing data showing microbial diversity and taxonomic abundance.
Clostridium butyricum and Its Derived Extracellular Vesicles Modulate Gut Homeostasis and Ameliorate …
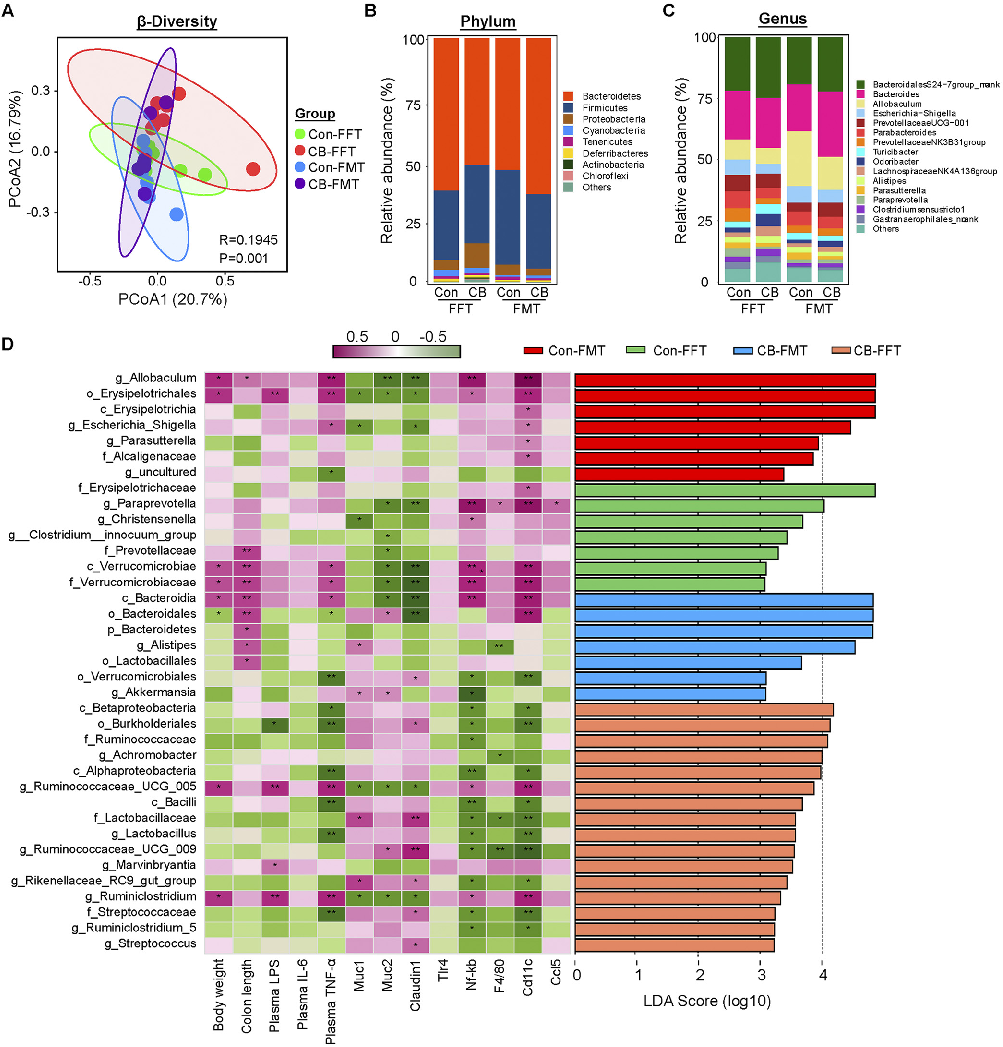
Alpha and beta diversity metrics reveal shifts in gut microbial community structure following probiotic intervention. This figure compares microbiome diversity indices between C. butyricum-treated and control groups.
Clostridium butyricum and Its Derived Extracellular Vesicles Modulate Gut Homeostasis and Ameliorate …
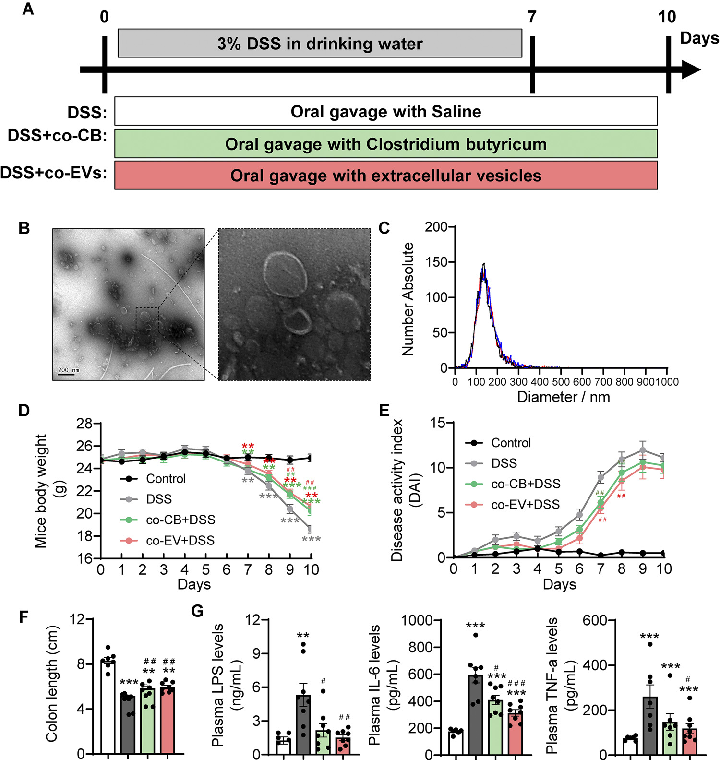
Specific bacterial taxa were differentially abundant in C. butyricum-treated mice compared to controls. This figure highlights the key microbial shifts associated with amelioration of colitis symptoms.
Clostridium butyricum and Its Derived Extracellular Vesicles Modulate Gut Homeostasis and Ameliorate …
Short-chain fatty acid production in the gut is modulated by C. butyricum colonization. This figure presents metabolomic data on butyrate and other SCFA concentrations in the colonic lumen.
Clostridium butyricum and Its Derived Extracellular Vesicles Modulate Gut Homeostasis and Ameliorate …
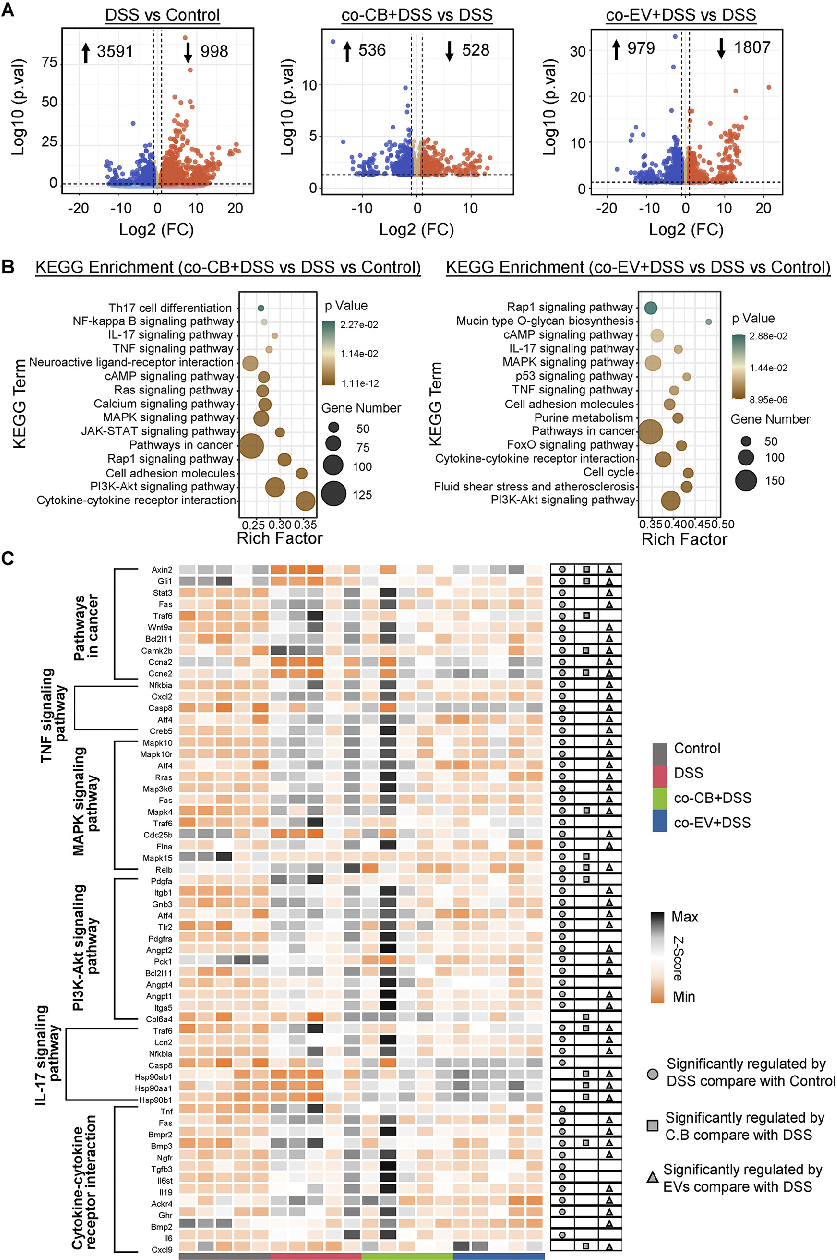
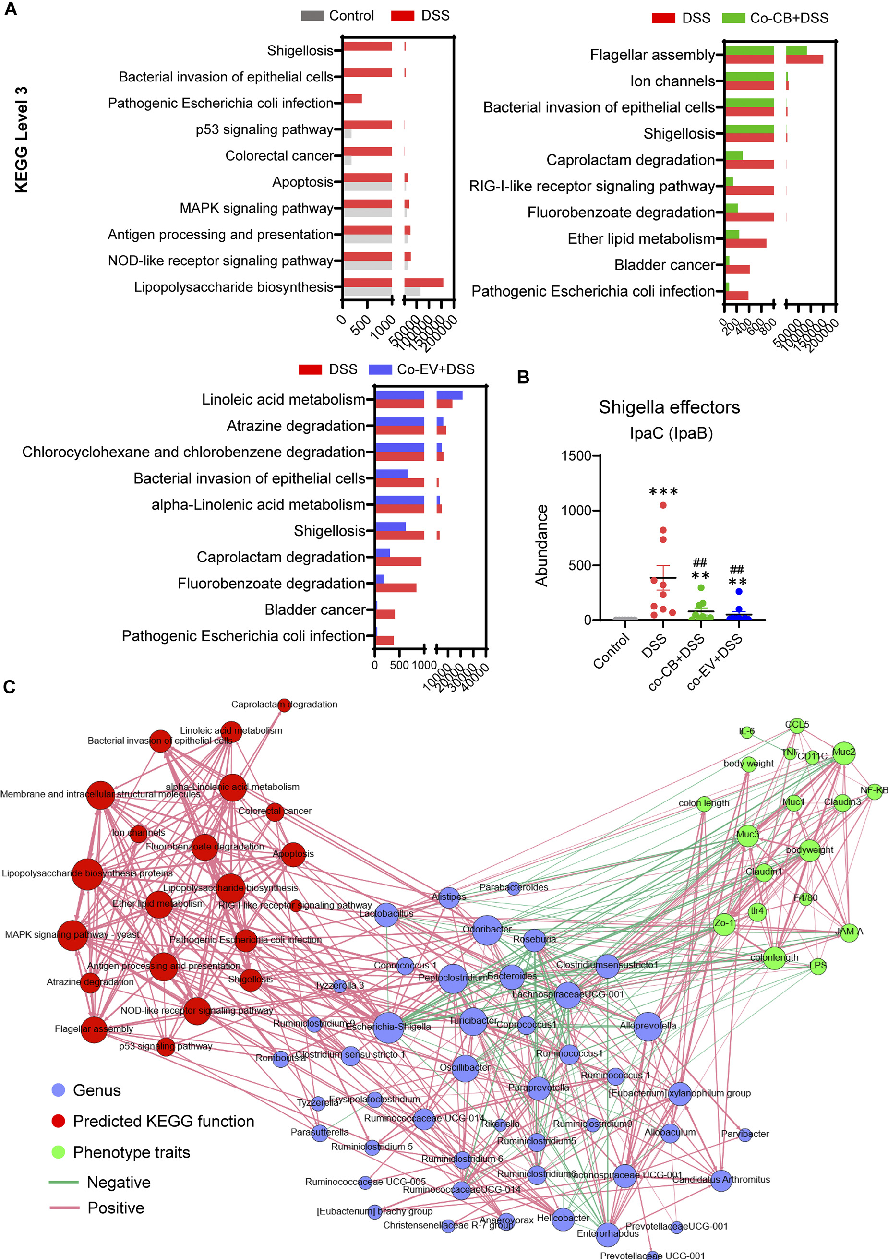
Signaling pathway analysis reveals molecular mechanisms underlying C. butyricum's protective effects. This figure presents Western blot or gene expression data for key inflammatory and barrier-related signaling pathways.
Clostridium butyricum and Its Derived Extracellular Vesicles Modulate Gut Homeostasis and Ameliorate …
Extracellular vesicles from C. butyricum may directly modulate intestinal epithelial cell responses. This figure presents in vitro data on vesicle uptake and downstream cellular effects in colonic epithelial cultures.
Clostridium butyricum and Its Derived Extracellular Vesicles Modulate Gut Homeostasis and Ameliorate …
Comprehensive mechanistic model summarizing how C. butyricum and its extracellular vesicles modulate gut homeostasis. This figure integrates findings on barrier function, immune modulation, and microbiome changes in experimental colitis.
Clostridium butyricum and Its Derived Extracellular Vesicles Modulate Gut Homeostasis and Ameliorate …
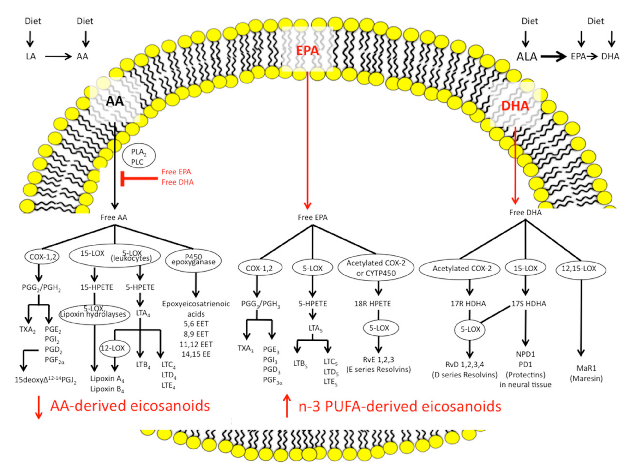
N-3 and n-6 polyunsaturated fatty acids follow distinct biosynthetic pathways to produce eicosanoids and specialized pro-resolving mediators. This diagram outlines the synthesis routes from alpha-linolenic acid and arachidonic acid through COX and cytochrome P450 enzymes to their bioactive lipid products.
The Imbalance between n-6/n-3 Polyunsaturated Fatty Acids and Inflammatory Bowel Disease: A …
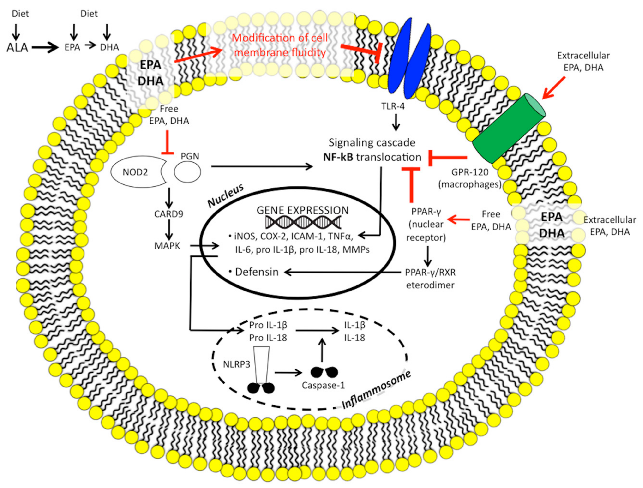
N-3 polyunsaturated fatty acids modulate intestinal innate immunity through multiple receptor and signaling targets. This diagram maps the interactions of DHA and EPA with G-protein coupled receptors, COX enzymes, and caspase recognition domains in intestinal immune cells.
The Imbalance between n-6/n-3 Polyunsaturated Fatty Acids and Inflammatory Bowel Disease: A …
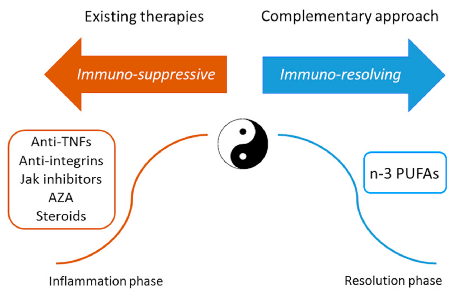
Current IBD treatments employ both immuno-suppressive and immuno-resolving strategies. This diagram contrasts conventional therapies (azathioprine, anti-TNF agents) with emerging PUFA-based approaches that promote active resolution of intestinal inflammation.
The Imbalance between n-6/n-3 Polyunsaturated Fatty Acids and Inflammatory Bowel Disease: A …
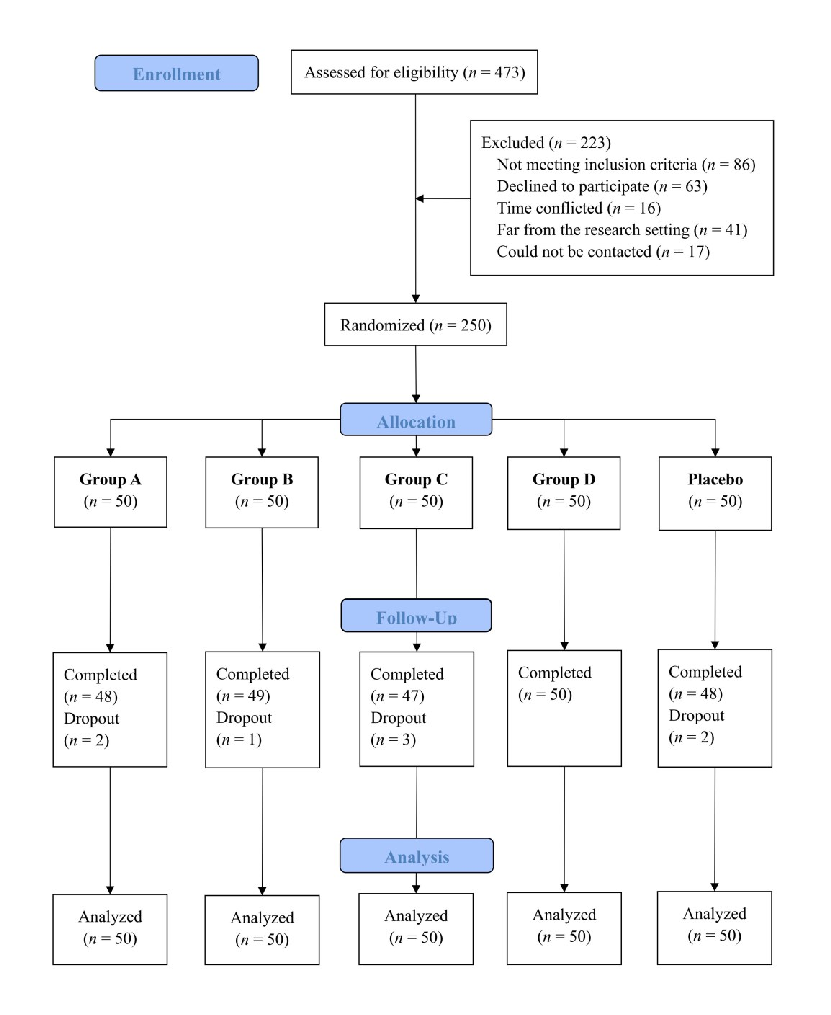
A double-blinded randomized placebo trial evaluated dietary fiber and probiotic formulas for functional constipation. This figure presents the study design, participant flow, or clinical outcomes showing how fiber and probiotic interventions modulated constipation symptoms and gut microbiota.
Effects of dietary fibers or probiotics on functional constipation symptoms and roles …
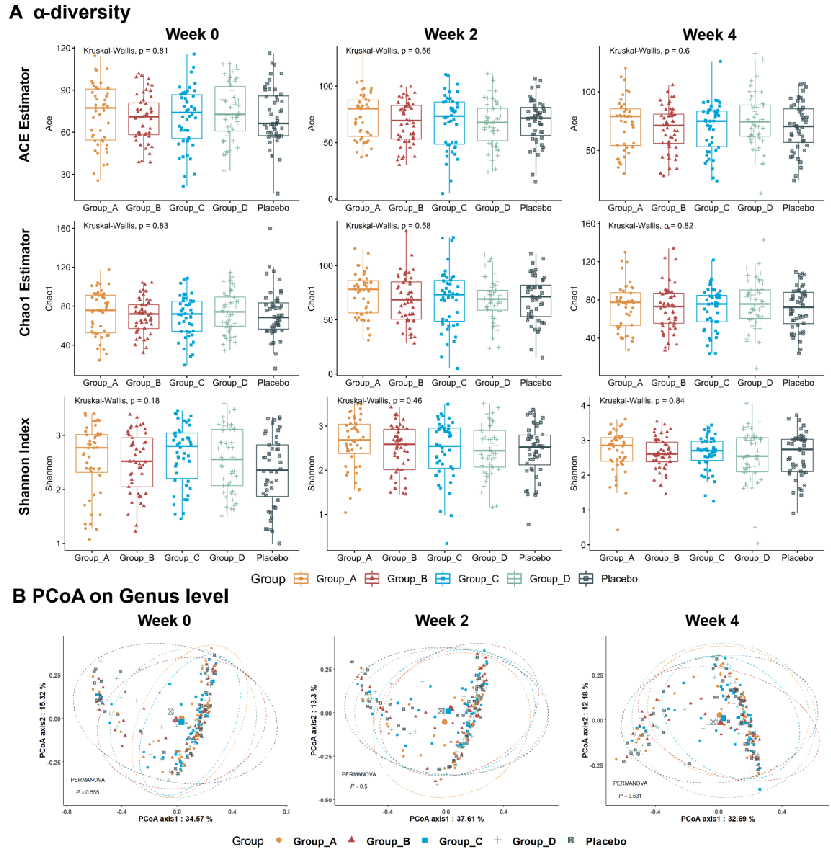
Gut microbiota composition changes were associated with symptom improvement in constipated patients receiving fiber or probiotic supplementation. This figure presents microbiome analysis data identifying bacterial taxa of relevance to constipation relief.
Effects of dietary fibers or probiotics on functional constipation symptoms and roles …
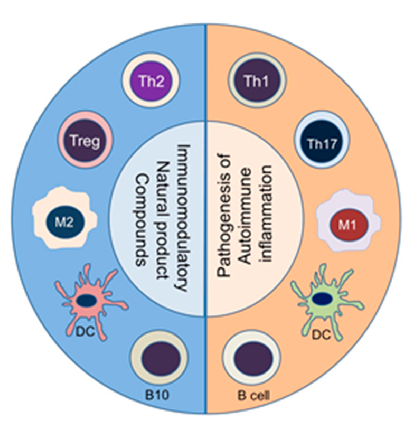
Natural product compounds shift the balance between pathogenic pro-inflammatory immune cells (right panel) and protective anti-inflammatory/immunomodulatory cell subsets (left panel). This diagram illustrates how diverse natural products can rebalance immune cell populations in autoimmune disease.
The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation.
In vivo models of autoimmune diseases have demonstrated therapeutic efficacy of various natural compounds. This figure presents preclinical evidence from animal models of rheumatoid arthritis, multiple sclerosis, or inflammatory bowel disease treated with natural products.
The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation.
Clinical translation of natural anti-inflammatory products requires understanding of bioavailability and dose-response relationships. This figure discusses the therapeutic potential and limitations of natural product-based interventions for autoimmune conditions.
The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation.
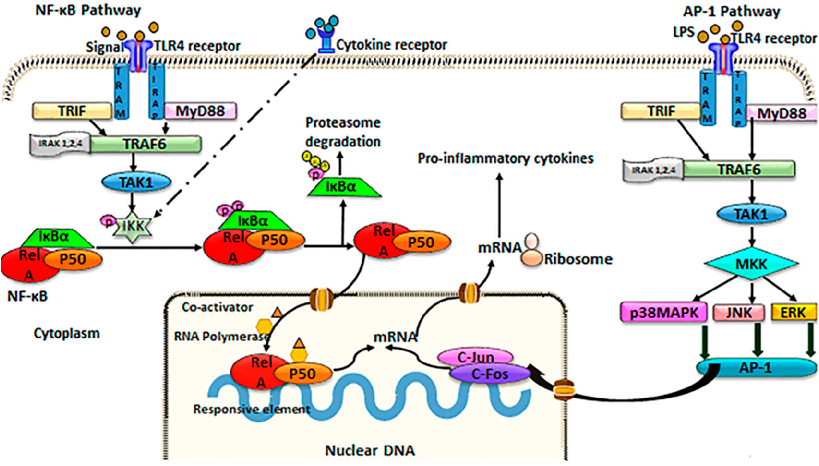
TLR4/NF-kB/AP-1 signaling is a key inflammatory pathway in intestinal disease. This schematic illustrates the cascade from TLR4 receptor activation through NF-kB and AP-1 transcription factors to pro-inflammatory gene expression, and indicates where curcumin may intervene.
Curcumin and Intestinal Inflammatory Diseases: Molecular Mechanisms of Protection.
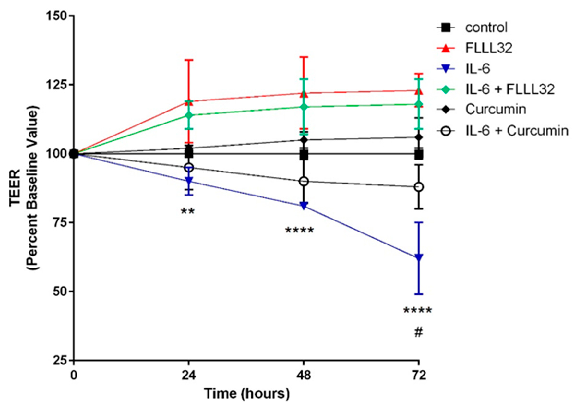
FLLL32 (a curcumin analogue) and curcumin both protect against IL-6-induced reduction of transepithelial electrical resistance (TEER) in T84 cell monolayers. This graph shows that curcumin preserves intestinal barrier integrity by counteracting cytokine-mediated tight junction disruption over 72 hours.
Curcumin and Intestinal Inflammatory Diseases: Molecular Mechanisms of Protection.
Curcumin's effects on intestinal tight junction proteins have been demonstrated in multiple experimental models. This figure presents protein expression or immunofluorescence data showing curcumin-mediated preservation of barrier function.
Curcumin and Intestinal Inflammatory Diseases: Molecular Mechanisms of Protection.
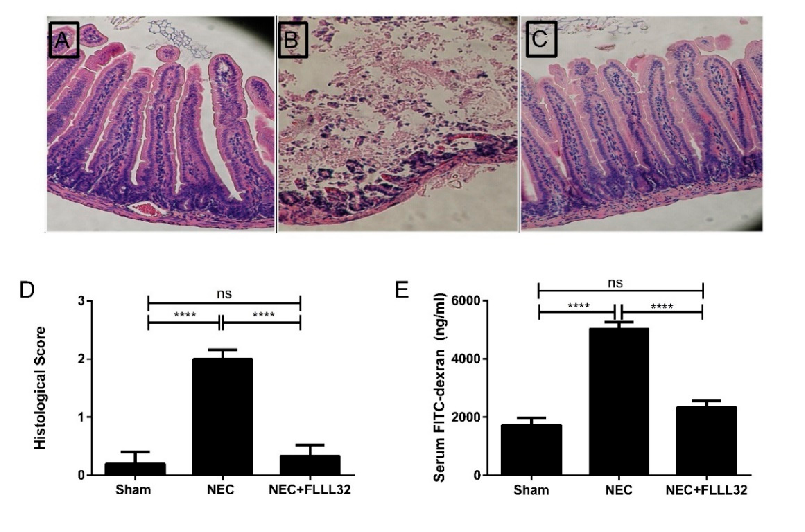
Animal models of necrotizing enterocolitis and colitis respond favorably to curcumin treatment. This figure presents in vivo data on curcumin's protective effects in experimental intestinal inflammation.
Curcumin and Intestinal Inflammatory Diseases: Molecular Mechanisms of Protection.
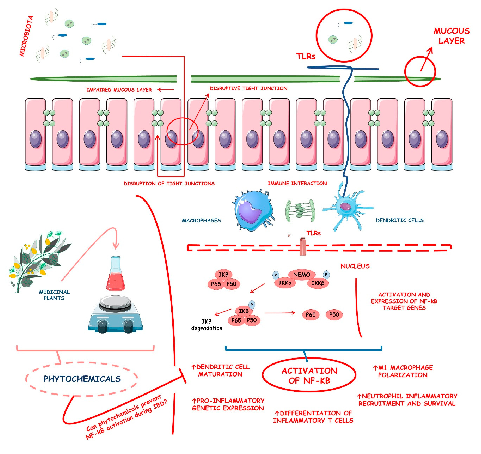
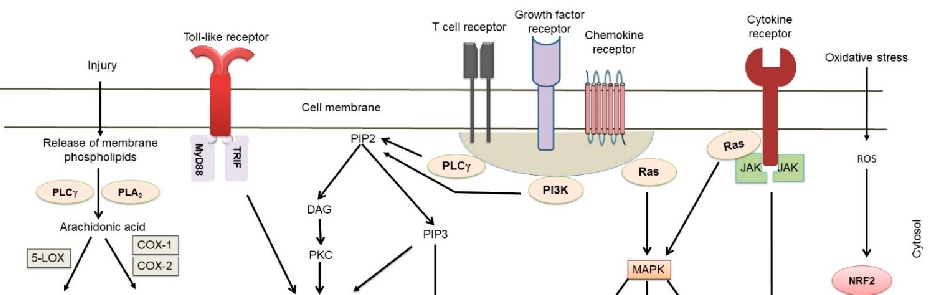
Inflammatory bowel diseases are associated with activation of the NF-kB signaling pathway. This diagram illustrates how various phytochemicals may counteract NF-kB pathway activation through inhibition of Toll-like receptors, IKK complex, and downstream transcriptional targets in IBD.
Phytochemicals and Regulation of NF-kB in Inflammatory Bowel Diseases: An Overview of …
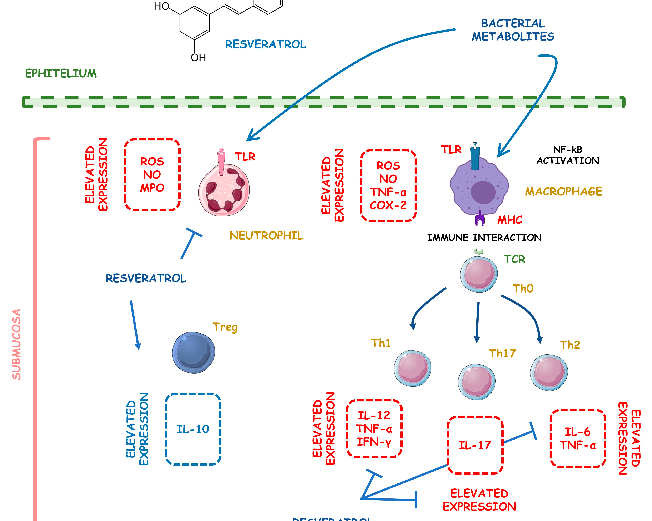
Supplementary figure from a comprehensive review of phytochemical effects on NF-kB regulation in inflammatory bowel disease. The review examines multiple plant-derived compounds with potential to attenuate chronic intestinal inflammation through modulation of key signaling pathways.
Phytochemicals and Regulation of NF-kB in Inflammatory Bowel Diseases: An Overview of …
पृष्ठ 4 / 20