Bone health in patients with inflammatory bowel disease.
Study Design
- अध्ययन प्रकार
- Review
- जनसंख्या
- Aged individuals
- हस्तक्षेप
- Bone health in patients with inflammatory bowel disease. None
- तुलनित्र
- None
- प्राथमिक परिणाम
- Inflammatory markers
- प्रभाव की दिशा
- Neutral
- पूर्वाग्रह का जोखिम
- Unclear
Abstract
Patients with inflammatory bowel disease (IBD) are prone to reduced bone mineral density and elevated overall fracture risk. Osteopenia affects up to 40% of patients with IBD (high regional variability). Besides disease activity, IBD specialists must consider possible side effects of medication and the presence of associated diseases and extraintestinal manifestations. Osteopenia and osteoporosis remain frequent problems in patients with IBD and are often underestimated because of widely differing screening and treatment practices. Malnutrition, chronic intestinal inflammation and corticosteroid intake are the major pathophysiological factors contributing to osteoporosis. Patients with IBD are screened for osteoporosis using dual-energy X-ray absorptiometry (DXA), which is recommended for all patients with a prolonged disease course of more than three months, with repeated corticosteroid administration, aged >40 years with a high FRAX risk score or aged <40 years with multiple risk factors. From a therapeutic perspective, besides good disease control, vitamin D supplementation and glucocorticoid sparing, several specific osteological options are available: bisphosphonates, receptor activator of nuclear factor kappa-B ligand (RANKL) inhibitors (denosumab), parathyroid hormone (PTH) analogues and selective estrogen receptor modulators. This review provides an overview of the pathophysiology, diagnosis, prevention and treatment of IBD-associated bone loss.
संक्षेप में
The pathophysiology, diagnosis, prevention and treatment of IBD-associated bone loss is reviewed, including osteoporosis, which affects up to 40% of patients with IBD (high regional variability).
Full Text
Bone health in patients with inflammatory bowel disease
Andrea S. Kreienbuehla, Gerhard Roglera, Emanuel Burrib, Luc Biedermanna, Christian Meierc, Pascal Juilleratde, Sophie Restellinifgh, Petr Hruzi, Stephan R. Vavrickaa, Daniel Aeberlijk, Frank Seiboldlm, on behalf of the Swiss IBDnet (SIBDCS) investigators
- a Department of Gastroenterology and Hepatology, University Hospital Zurich, University of Zurich, Zurich, Switzerland
- b Gastroenterology and Hepatology, University Medical Clinic, Kantonsspital Baselland, Liestal, Switzerland
- c Division of Endocrinology, Diabetes and Metabolism, University Hospital Basel
- d Crohn's and Colitis Center, Gastroenterologie Beaulieu, Lausanne, Switzerland
- e Gastroenterology, Clinic for Visceral Surgery and Medicine, Inselspital, Bern University Hospital, University of Bern, Bern, Switzerland
- f Inflammatory Bowel Disease Center, Division of Gastroenterology, Hôpital de la Tour, Geneva, Switzerland
- g Faculty of Medicine, University of Geneva, Switzerland
- h McGill University Health Center, McGill University, Montréal, Québec, Canada
- i Clarunis, University Center for Gastrointestinal and Liver Diseases, Basel, Switzerland
- j Department of Rheumatology and Immunology, University Hospital Bern, Bern, Switzerland
- k Department of Internal Medicine, Spital Emmental, Burgdorf, Switzerland
- l Intesto, Gastroenterologische Praxis, Crohn-Colitis-Zentrum Bern, Bern, Switzerland
- m University of Fribourg, Switzerland
Dr. med. Andrea S. Kreienbuehl Department of Gastroenterology and Hepatology University Hospital Zurich Rämistrasse 100 CH-8091 Zurich andrea.kreienbuehl[at] usz.ch
Summary
Patients with inflammatory bowel disease (IBD) are prone to reduced bone mineral density and elevated overall fracture risk. Osteopenia affects up to 40% of patients with IBD (high regional variability). Besides disease activity, IBD specialists must consider possible side effects of medication and the presence of associated diseases and extraintestinal manifestations. Osteopenia and osteoporosis remain frequent problems in patients with IBD and are often underestimated because of widely differing screening and treatment practices.
Malnutrition, chronic intestinal inflammation and corticosteroid intake are the major pathophysiological factors contributing to osteoporosis. Patients with IBD are screened for osteoporosis using dual-energy X-ray absorptiometry (DXA), which is recommended for all patients with a prolonged disease course of more than three months, with repeated corticosteroid administration, aged >40 years with a high FRAX risk score or aged <40 years with multiple risk factors. From a therapeutic perspective, besides good disease control, vitamin D supplementation and glucocorticoid sparing, several specific osteological options are available: bisphosphonates, receptor activator of nuclear factor kappa-B ligand (RANKL) inhibitors (denosumab), parathyroid hormone (PTH) analogues and selective estrogen receptor modulators.
This review provides an overview of the pathophysiology, diagnosis, prevention and treatment of IBD-associated bone loss.
Introduction
Patients with inflammatory bowel disease (IBD) are prone to reduced bone mineral density and have a >38% higher
overall risk of spine fractures than the general population [1, 2]. Besides disease activity, IBD specialists must consider possible side effects of medication and the presence of associated diseases and extraintestinal manifestations.
Osteopenia and osteoporosis are complications of IBD that may not receive sufficient attention from the treating physicians. Malnutrition, chronic intestinal inflammation and corticosteroid intake are the major pathophysiological factors contributing to osteoporosis. The primary mechanism involved is an imbalance of the receptor activator of nuclear factor kappa-B ligand (RANKL)/osteoprotegerin (OPG) ratio at the molecular level or bone anabolic and catabolic activity at the cellular level.
The established standard diagnostic procedure to detect IBD-associated bone deterioration is dual-energy X-ray absorptiometry (DXA). From a therapeutic perspective, besides good disease control, vitamin D supplementation and glucocorticoid sparing, several specific osteological options are available: bisphosphonates, RANKL inhibitors (denosumab), parathyroid hormone (PTH) analogues and selective estrogen receptor modulators (SERMs).
Pathophysiology of inflammatory bowel disease-associated bone deterioration
The pathophysiology of low bone mineral density in patients with IBD is multifactorial. It involves malabsorption, corticosteroid treatment and the systemic inflammatory process itself, all of which may adversely affect the adequate formation of peak bone mass (up to the age of about 30 years), age-related loss of bone mass or both. Malnutrition might occur in patients with IBD from reduced dietary intake, malabsorption due to intestinal inflammation, short bowel syndrome after intestinal resection or increased energy requirements due to hypercatabolism [3]. In particu-
lar, calcium and vitamin D deficiencies are detrimental to bone health and mineralisation, especially in combination with physical inactivity and decreased exposure to sunshine in patients with active disease. In patients with IBD, bone mineral density correlates more with disease severity than disease location and extent [4]. Chronic gut inflammation is an independent risk factor promoting loss of bone mineral density [5].
From an osteoimmunological perspective, the high prevalence of reduced bone mineral density and osteoporosis in patients with IBD can be explained by pro-inflammatory and pro-apoptotic mechanisms. Much of the evidence regarding the effects of pro-inflammatory mechanisms on bone loss originates from studies on rheumatoid arthritis [6–8]. Because rheumatoid arthritis causes bone loss not only at sites prone to inflammation, such as the joints of the hands and fingers, but also systemically throughout the skeleton, a comparison of rheumatoid arthritis and IBD is helpful and valid.
At the cellular level, the key effector cells at the interface between inflammation and bone are the osteoclasts, boneforming osteoblasts, regulatory osteocytes, and T cells. At the cytokine level, secreted cytokine levels are elevated systemically in both rheumatoid arthritis and IBD: TNFα, IL-1β, IL-6, IL-8, IL-12/IL-23, IL-17, IL-18, IL-32, and interferon-gamma (IFN-γ) [9].
The release of these inflammatory cytokines, particularly TNF-α, IL-1, IL-6 and IFN-y, modulates the receptor activator of nuclear factor kappa-B (RANK)/RANKL/OPG pathway, inducing bone loss [9]. The RANK and its ligand (RANKL) and decoy receptor OPG regulate bone-resorbing cells (osteoclasts) and lead to bone resorption [10]. RANK is a transmembrane protein belonging to the TNF receptor family and is the primary activator of osteoclastogenesis [11]. Osteoclast maturation is necessary to initiate the bone resorption process. RANKL secreted by osteoblasts binds to RANK on the surface of pre-osteoclasts and mature osteoclasts, inducing their proliferation, activity, and survival. Osteoprotegerin is produced by osteoblasts and competes with RANKL to bind to the RANK receptor, thus blocking osteoclastogenesis.
In addition, increased levels of IL-2, IL-17, IFN-y and TNF-α have been documented in Crohn’s disease. In contrast, ulcerative colitis is associated with cytokines of the Th2 profile, such as IL-4, IL-5, and IL-13 [12]. IL-12, a Tcell mediator, is also an important inflammatory cytokine found in IBD and interacts with TNF-α to inhibit osteoclastogenesis [13–15].
Moreover, there is sufficient evidence that TNF-α plays an important role in bone loss in Crohn’s disease. TNF-α blockade improves bone formation and reduces bone resorption by suppressing T cells [16, 17]. Infliximab maintenance therapy also leads to about a 14% increase in lumbar bone density [18, 19]. The Swiss IBD cohort study (SIBDCS), launched in 2006 with the aim of enrolling and following patients with IBD throughout Switzerland [20], demonstrated that early administration of anti-TNF-α was associated with a lower osteoporosis risk.
TNF-α inhibition appears to have a beneficial effect on bone turnover, especially when administered in combination with bisphosphonates [21]. The fact that this was ob-
served in infliximab responders and non-responders and during the maintenance phase in patients with or without corticosteroid co-administration suggests an independent mode of action [22, 23]. Of course, this effect might also be secondary to reducing active inflammation and decreased glucocorticoid exposure.
While some cytokines directly act through their receptors (IL-1, IL-6, IL-17 and TNF-α), others act indirectly through the OPG/RANKL/RANK pathway via T cells or fibroblasts [24]. Importantly, the upregulated cytokines are not only bone destructive (e.g. IL-6, TNF-α and IL-1β) but also bone protective and anti-osteoclastogenic (e.g. IL-12/ IL-23, IL-17, IL-18, IL-4, IL-10 and IFN-y) [25]. This observation is of interest since the therapeutic effects of anti-IL12/23 inhibitors were recently found to lower both the need for glucocorticoids and direct bone-specific harm [26]. However, reliable data for vedolizumab or ustekinumab are currently lacking.
Regarding the apoptotic mechanisms leading to bone loss, the mode of action of systemic glucocorticoids (GCs) must be elucidated. Glucocorticoids decrease the ability of osteoblasts to rebuild eroded bone and compensate for bone loss by dose-dependently activating caspases and the Wingless-related integration site (WNT) pathway in osteoblasts [27]. In addition, dying osteoblasts and systemic glucocorticoids activate bone resorption via the OPG/ RANKL/RANK pathway, resulting in a double negative bone balance [28]. In addition, apoptosis of osteocytes also leads to a reduced load-bearing capacity of the bone, since on one hand, microfractures are no longer recognized and repaired, and on the other hand, adaptations no longer occur.
Corticosteroids also increase the expression of RANKL and decrease OPG receptors in stem cells and osteoblastic cells, thus inhibiting stem cell differentiation into osteoblasts, inducing osteoblast apoptosis, and favouring the differentiation of precursors into osteoclasts [29]. This mechanism will decrease the secretion of the osteoid matrix and enhances bone reabsorption. However, the increase in RANKL is only transient, and the failure of bone formation seems to be more important than bone resorption [30].
In summary, an imbalance of the RANKL/OPG ratio plays an essential role in developing low bone mineral density in patients with IBD.
Endocrine manifestations are frequent in IBD including metabolic bone disease with bone loss, growth failure and hypogonadism with pubertal delay in patients with childhood onset of disease. The pathogenesis of growth failure is multifactorial and include disease-related inflammation, malnutrition, glucocorticoid use, and hypogonadism [31, 32] and is related to an impairment of the GH-IGF1 axis with relative growth hormone resistance [33]. In parallel to growth failure, the presence of delayed puberty, weight loss and glucocorticoid treatment impair an increase of bone mass in adolescence. Therefore, patients with childhood onset of IBD may present with low peak bone mass in early adulthood, an important predisposing factor for low bone mass and increased fracture risk later in life.
One of the key hormones involved in bone metabolism is vitamin D. Vitamin D levels are lower in adult and pae-
diatric patients with IBD as compared to healthy controls [34], which is due to impaired intestinal absorption (specifically in patients with small-bowel resection), reduced dietary or supplemental intake, low sun exposure and vitamin D loss in the presence of protein-losing enteropathies. As malabsorptive conditions severely impair vitamin D status with hypocalcaemia, impaired bone mineralisation and increased risk of fractures, biochemical assessment and supplementation should be routinely considered in all patients with IBD. Furthermore, recent data suggest that poor vitamin D status may negatively impact the clinical course of IBD due to its immune-modulating effect [35].
Adipokines are cytokines secreted by adipose tissue and have been implicated in the pathogenesis of IBD. Recent research has suggested that adipokines (such as leptin, adiponectin, apelin, and chemerin) may be involved in the development of IBD-induced bone loss. Specifically visceral adipose tissue exhibits pro-inflammatory, immunoregulatory, and endocrine activity [36, 37]. While the exact mechanisms regarding the crosstalk between fat and bone tissues are still unclear, dysregulation of adipokine levels has been shown to play an important role in the regulation of osteoblastic and osteoclastic cell functions.
In summary, the chronic inflammation and malnutrition associated with IBD can disrupt the normal function of several hormones involved in bone metabolism resulting in decreased bone density and increased risk of fractures. In clinical practice, preventive measures to ascertain bone accrual in adolescence and prevention of accelerated bone loss in adulthood include assessment of calcium/vitamin D status as well as exclusion of sex hormone deficiencies, and consider replacement therapies if needed.
Another, maybe underestimated mode of action associated with bone deterioration in IBD patients is the iron-induced hypophosphataemia, an emerging complication first been reported in 1982 in a patient receiving repeated intravenous therapy with saccharated iron oxide [38], iron polymaltose [39] or ferric carboxymaltose [40]. The application of iron isomaltose is not associated with hypophosphataemia [41] and therefore an option in patients with low bone density or osteoporosis.
Epidemiology of bone diseases in inflammatory bowel disease
A recent systematic review including 3661 IBD patients and 12,789 healthy controls identified a prevalence of osteoporosis of 2–16% in the former population (7–15% for Crohn’s disease and 2–9% for ulcerative colitis) and of 3–10% in the later. These results are consistent with observations made in a 10 years follow-up population-based inception Danish cohort, where the prevalence of osteoporosis in 513 IBD patients was 14%, which was also significantly increased compared to controls [42, 43].
Because of widely differing screening and treatment practices and a lack of awareness, osteopenia and osteoporosis are often underestimated, particularly in patients with IBD. In the Swiss IBD Cohort Study (SIBDCS), 19% of patients had low bone mineral density [44]. Other studies estimate that osteopenia and osteoporosis affect 35–40% and up to 15% of patients with IBD, respectively [45–47], with a high regional variability.
The lack of awareness in patients with IBD might be explained by young age and the lack of a direct burden from musculoskeletal pain.
Assessment of bone health
Bone mineral density is a surrogate for bone health and a predictor of fracture risk. It is measured by dual-energy Xray absorptiometry (DXA) [48] in the femur (femoral neck or total hip) and/or lumbar spine [49]. In postmenopausal women and men aged ≥50 years, osteoporosis is defined as a bone mineral density value at least 2.5 standard deviations (SDs) below the peak bone mass of healthy adults (T-score ≤−2.5), and osteopenia, a precursor of osteoporosis, is defined as a bone mineral density value between 1.0 and 2.5 SDs below normal [2] (table 1). In individuals aged <50 years, low bone mineral density (osteoporosis) is defined as a bone mineral density value “below the expected range for age” (Z-score <–2 SDs) [50].
Indications for osteoporosis screening in patients with inflammatory bowel disease
Patients aged >40 years without a previous history of prolonged corticosteroid therapy should be assessed using the fracture risk assessment tool (FRAX; https://frax.shef.ac.uk/FRAX/tool.aspx?country=15),
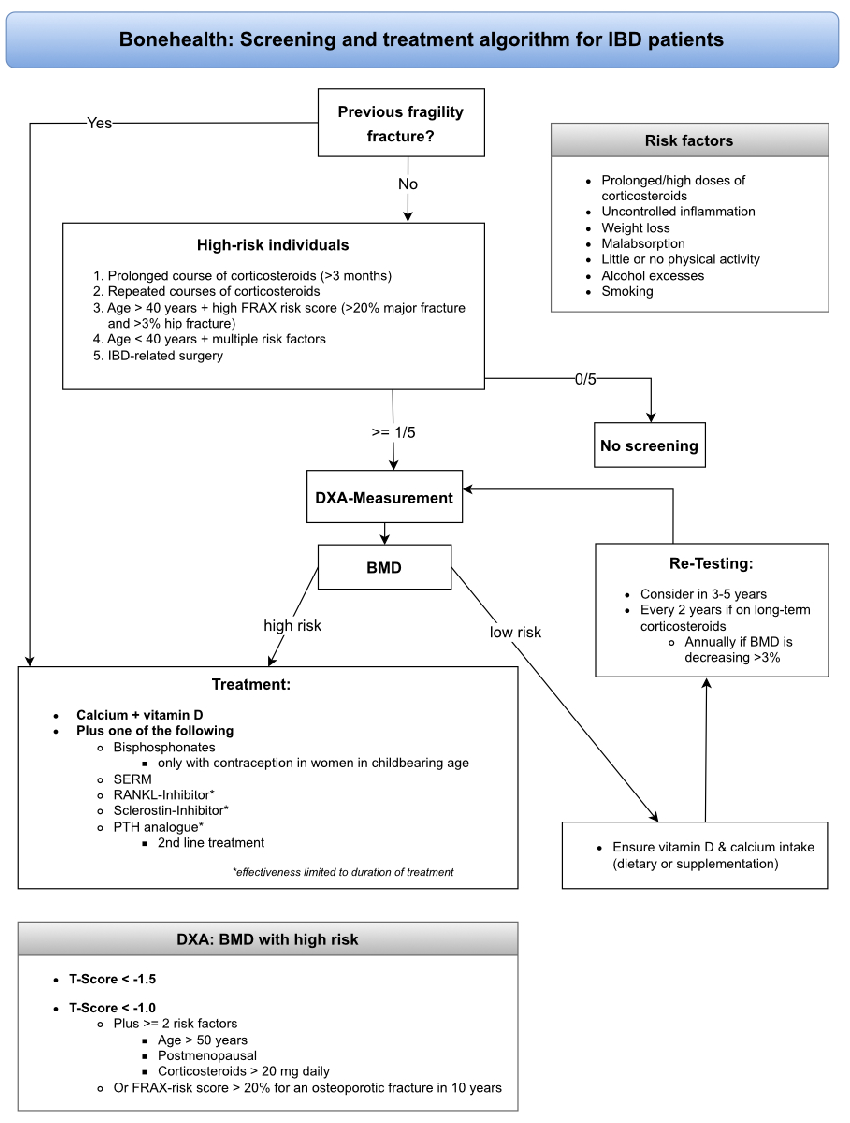
which assesses the 10-year probability of a major osteoporotic fracture (MOF). FRAX is a proven tool but does not discriminate past versus current corticosteroid use or stratify risk according to corticosteroid doses >7.5 mg [52]. According to current guidelines (British Society of Gastroenterology, European Crohn’s and Colitis Organisation and American Gastroenterological Association), we suggest screening for osteopenia or osteoporosis using DXA in high-risk individuals and after IBD-related surgery [26, 50, 53]. At-risk individuals are patients with IBD with a prolonged course (>3 months) or repeated corticosteroid courses, those aged >40 years with a high FRAX risk score (>20% major fracture and >3% hip fracture), and those <40 years with multiple risk factors (prolonged/high doses corticosteroids, uncontrolled inflammation, weight loss, malabsorption, little or no physical activity, alcohol excesses and smoking; figure 1 and table 2).
An antiresorptive treatment is recommended for patients with a previous fragility fracture, and DXA measurements may be used for treatment monitoring.
There is currently no evidence that budesonide causes reduced bone mineral density in patients with IBD. Studies on microscopic colitis – where budesonide is the stateof-the-art treatment – did not show significant differences in the incidence of osteopenia/osteoporosis [54]. Nonethe-
Table 1 : The World Health Organization’s definition of osteoporosis based on DXA measurement [51].
less, these two clinical conditions differ in particular regarding the severity of secondary malabsorption and consequential deficiencies. Therefore, it cannot be excluded that budesonide may have more adverse effects in patients with IBD.
Smoking is a major risk factor for patients with Crohn’s disease. It is associated with relapses, more frequent postoperative complications due to impaired microcirculation and reduced bone mineral density with an increased frac-
ture risk. Therefore, patients should make every effort to stop smoking immediately [26, 55, 56].
DXA measurement is still the standard diagnostic modality for assessing bone mineral density/bone health. However, since the two-dimensional nature of DXA imposes limitations, other imaging modalities (currently mainly used in research settings) should be mentioned to complete the diagnostic spectrum [57]:
- – Quantitative computed tomography (QCT) and highresolution peripheral QCT (HR-pQCT) provide threedimensional volumetric bone mineral density measurements and can differentiate between the cortical and trabecular bone.
- – Magnetic resonance imaging can evaluate bone microarchitecture while avoiding radiation exposure compared to DXA and identify common vertebral fractures.
Trabecular bone score based on texture analysis of the DXA lumbar spine image.
Only a few studies have examined adults with IBD using HR-pQCT, demonstrating decreased trabecular and volumetric bone mineral density and possibly a decreased cortical bone area [58]. In 2017, Pepe et al. evaluated the SWISS IBD Cohort using HR-pQCT (Xtreme I; Scanco, Switzerland). They demonstrated a predominantly altered trabecular bone microstructure compared to healthy controls, which is associated with fractures during childhood/adolescence and may predispose to fragility fractures throughout life [43]. Further prospective studies with HRpQCT are needed to elucidate its clinical impact and possible advantages compared to DXA.
DXA measurement and therapeutical consequences
Patients on long-term corticosteroids should undergo bone densitometry every second year. However, yearly DXA measurements are recommended for those showing a >3% decrease in bone mineral density annually. However, in Switzerland, health insurance currently does not cover repeated DXA measurements after one year, and corresponding cost reimbursement requests must be submitted.
A patient is considered high-risk, and treatment to prevent osteoporosis should be initiated, if their bone mineral densitometry indicates a T-score <−1.5, a T-Score <−1.0 and they have at least two risk factors (age >50 years, postmenopausal or corticosteroids >20 mg daily) or a FRAX score with a >20% risk for an osteoporotic fracture within 10 years [59].
Additionally, patients with past fragility fractures, aged >69 years, taking corticosteroids (>7.5 mg/day) or with a high-risk FRAX score should be evaluated for bone protective therapy before the start of corticosteroid treatment [60]. Newer guidelines from the American College of
Rheumatology strongly recommend an initial clinical fracture risk assessment for all adults initiating or continuing corticosteroid therapy ≥2.5 mg/day for >3 months, those who have never had fracture risk assessment, and those previously treated with osteoporotic therapy. For those taking glucocorticoids at >7.5 mg/day, the FRAX score should be multiplied by 1.15 to determine the 10-year risk of major osteoporotic fracture and by 1.2 to determine the hip fracture risk [61].
Treatment
Adequate medical treatment can partially reverse bone deterioration and prevent osteoporotic fractures even in highrisk individuals such as postmenopausal women, older men with osteoporosis and patients taking glucocorticoids [49,62–64]. Risk factors for increased bone loss should be limited, and adequate dietary intake or supplementation of calcium or vitamin D should be maintained.
More than 50% of patients with ulcerative colitis and Crohn’s disease in Northern Europe and the United Kingdom suffer from vitamin D deficiency (25-hydroxyvitamin D [25OHD] <50 nmol/l) [65, 66]. Therefore, we recommend quantifying/measuring vitamin D levels (25OHD) in patients with IBD at least once per year. However, patients should be informed that the health insurance company only reimburses vitamin D supplementation for a severe deficiency (25OHD <25 nmol/l).
High-risk patients with IBD and osteopenia (not meeting the criteria for pharmacological treatment) and all patients starting corticosteroid therapy should be adequately supplemented with vitamin D (800–1000 IU/day) and calcium (500–1000 mg/day, dietary or supplement). We suggest determining the patient’s daily calcium intake to assess their supplementation needs (https://www.rheumaliga.ch/calciumrechner). Patients should be prevented from ingesting >1400 mg of calcium daily since this may increase their all-cause mortality [67]. British guidelines recommend that those with vitamin D deficiency receive an initial treatment of 50,000 IU weekly for six weeks [26].
In addition to vitamin D and/or calcium supplementation, individuals with osteoporosis or increased fracture risk should receive a specific osteological treatment with antiresorptives (bisphosphonates, denosumab or selective estrogen receptor modulators [SERMs]), an osteoanabolic agent (teriparatide) or a dual effect agent (romosozumab; table 3).
The treatment duration of antiresorptive drugs depends on their mode of action and long-term efficacy and safety. Data on reducing fracture risk in patients with IBD are primarily available for bisphosphonates (alendronate, risedronate and zoledronate). Intravenous zoledronate may be preferred due to the potential gastrointestinal side effects of oral bisphosphonates. For women of childbearing age, bisphosphonates should only be prescribed if they are taking effective contraception. Because of an elevated risk, osteonecrosis of the jaw or atypical femoral fractures should be ruled out before starting bisphosphonate therapy. Unlike bisphosphonates, which are deposited on the mineralised bone surface and taken up by the mature osteoclasts, denosumab, a RANKL inhibitor, inhibits the proliferation and differentiation of osteoclast precursor cells and impairs
the action of mature osteoclasts. Notably, the effectiveness of denosumab is limited to the duration of treatment, unlike bisphosphonates, which are known for their residual anti-resorptive efficacy after treatment is discontinued. Sequential bisphosphonate treatment is mandatory after discontinuing denosumab to prevent accelerated bone loss and vertebral fractures [68, 69]. Romoszumab can increase bone density via another mode of action. By inhibiting sclerostin, romosozumab strongly stimulates bone formation and, to a lesser extent, inhibits bone resorption. Like with denosumab, sequential therapy with an antiresorptive agent is mandatory after discontinuing romosozumab.
Antiresorptive, second-line therapy with teriparatide is suggested in cases of severe osteoporosis (bone mineral density at the spine <–3.5 SDs) or treatment failure (incident vertebral fractures during bisphosphonate treatment). Teriparatide (a PTH analogue) stimulates osteoblastic bone formation, resulting in bone accrual, specifically in the trabecular bone. After treatment with teriparatide (usually for 24 months), sequential antiresorptive therapy is necessary to support the secondary mineralisation of the newly formed bone and maintain the bone mineral content [70,71].
Since most studies on treating and preventing osteoporosis have been conducted in postmenopausal women, evidence for treating young patients with IBD is limited. In these patients, the primary focus should be on controlling the risk factors.
Of note are new guidelines by the American College of Rheumatology on preventing and treating glucocorticoidinduced osteoporosis. They suggest treating adults aged ≥40 years receiving high-dose glucocorticoids (initial dose ≥30 mg/day for 30 days or cumulative dose ≥5 gm in one year) with oral/intravenous bisphosphonate, PTH analogue or RANKL inhibitor [61].
Sequential treatments are recommended when initial osteoporotic therapy and glucocorticoids are discontinued and at low (10-year risk of MOF <10%, hip ≤1%) or moderate (10-year risk of MOF 10–20%, hip 1–3%) FRAX risk.
No subsequent osteoprotective therapy is needed if the initial osteoporotic therapy consists of oral/intravenous bisphosphonate or SERM. In cases initially treated with PTH or romosozumab, subsequent treatment with oral/intravenous bisphosphonate or denosumab followed by bisphosphonate is mandatory. Additionally, in cases given denosumab therapy, an oral/intravenous bisphosphonate treatment is indicated for 1–2 years until the rebound after denosumab is finished (table 4) [61].
When initial osteoporotic therapy and glucocorticoids are discontinued, and the patient remains at high risk, the continuation of current therapy or a switch to intravenous bisphosphonate, denosumab, PTH, SERM or romosozumab is indicated [61].
Acknowledgments We thank Prof. Dr. med. Hans Jörg Häuselmann for his support. We also thank Sandoz Pharmaceuticals AG for providing an initial platform for the scientific exchange.
Financial disclosure The authors received no financial support for writing this review.
Potential competing interests
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. The following relationships or activities outside of the submitted manuscript have been reported: L. Biedermann: consulting fees, honoraria or travel support from Abbvie, Amgen, Bristol Myers Squibb, Esocap, Falk, Janssen, Lilly, Pfizer, Sanofi and Takeda. P. Hruz: participation on a data safety board or advisory board for Abbvie, Bristol Myers Squibb, Falk, iQone, Lilly, MSD, Sandoz and Takeda. P. Juillerat: consulting fees or honoraria from AbbVie, Arena, Amgen, Bristol Myers Squibb, Ferring, Gilead, Janssen, Lilly, MSD, Pfizer, Pierre Fabre, Roche, Sandoz, Takeda, Tillots and UCB Pharma. A. Kreienbühl: honoraria or travel support from Amgen, JanssenCilag and Takeda and participation on a data safety board or advisory board for Takeda. S. Restellini: consulting fees, honoraria or travel support from AbbVie, Bristol Myers Squibb, Falk, iQone, Janssen, Sandoz, Takeda, UCB and Vifor. G. Rogler: grants, consulting fees or honoraria from Abbvie, Ardeypharm, Arena, Augurix, Bristol Myers Squibb, Boehringer, Calypso, Celgene, Falk, Ferring, Fisher, Flamentera, Genentech, Gilead, Janssen, Lilly, MSD, Novartis, Pfizer, Phadia, Roche, UCB, Takeda, Tillots, Vifor, Vital Solutions, USB and Zeller. F. W. Seibold: consulting fees from Abbvie, Amgen, Bristol
- Table 3 : An overview of osteoprotective medication.
- Table 4 : The sequential treatments recommended when initial osteoporotic and glucocorticoid therapies are discontinued and patients are at low or moderate risk.
Myers Squibb, Falk, Lilly, Takeda S. Vavricka: Participation on a Data Safety Monitoring Board or Advisory Board for Abbvie, Bristol Myers Squibb, Falk, iQone, Lilly, MSD, Sandoz and Takeda.
Figures
Tables
Table 1
Table 2
Table 2 : Etiologic factors associated with altered bone health in inflammatory bowel disease [48].
Table 3
Table 4
Used In Evidence Reviews
Similar Papers
Journal of inflammation research · 2014
Vitamin D and inflammatory diseases.
Gastroenterology · 2017
Diet as a Trigger or Therapy for Inflammatory Bowel Diseases.
The journal of headache and pain · 2020
Gut-brain Axis and migraine headache: a comprehensive review.
Joint bone spine · 2010
Vitamin D and inflammation.
The Australian and New Zealand journal of psychiatry · 2017
A double-blind, randomized, placebo-controlled trial of Lactobacillus helveticus and Bifidobacterium longum for the symptoms of depression.
World journal of gastroenterology · 2016