The effect of probiotics supplementation on Helicobacter pylori eradication rates and side effects during eradication therapy: a meta-analysis.
Study Design
- Loại nghiên cứu
- Meta-Analysis
- Cỡ mẫu
- 4459
- Đối tượng nghiên cứu
- H. pylori infected patients
- Can thiệp
- The effect of probiotics supplementation on Helicobacter pylori eradication rates and side effects during eradication therapy: a meta-analysis. None
- Đối chứng
- None
- Kết quả chính
- None
- Xu hướng hiệu quả
- Positive
- Nguy cơ sai lệch
- Low
Abstract
BACKGROUND: Previous meta-analyses reported that probiotics improve the effectiveness of Helicobacter pylori (H. pylori) eradication during antibiotic therapy, while results regarding a possible reduction of side effects remained inconclusive. Moreover, the effectiveness of different strains of probiotics has not been studied so far. It is further conceivable that probiotics will produce additional effects only if antibiotics are relatively ineffective. METHODS: This meta-analysis includes eligible randomized controlled trials examining effects of probiotics supplementation on eradication rates (ER) and side effects, published up to May 2014. Sub-group analysis was performed to compare different probiotic strains and antibiotic therapies with different effectiveness in controls (ER <80% vs.>80%). Publication bias was assessed with funnel plots and Harbord's test. The quality of the trials was assessed with the Cochrane risk of bias tool. RESULTS: Thirty-three RCTs involving a total of 4459 patients met the inclusion criteria in case of eradication rates of which 20 assessed total side effects in addition. Overall, the pooled eradication rate in probiotics supplementation groups was significantly higher than in controls (ITT analysis: RR 1.122, 95% CI 1.086-1.159, PP analysis: RR 1.114, 95% CI 1.070-1.159). Sub group-analysis could, however, confirm this finding only for four individual strains (Lactobacillus acidophilus, Lactobacillus casei DN-114001, Lactobacillus gasseri, and Bifidobacterium infantis 2036) and for relatively ineffective antibiotic therapies. There was a significant difference between groups in the overall incidence of side effects (RR 0.735, 95% CI 0.598-0.902). This result was, however, only confirmed for non-blinded trials. CONCLUSIONS: The pooled data suggest that supplementation with specific strains of probiotics compared with eradication therapy may be considered an option for increasing eradication rates, particularly when antibiotic therapies are relatively ineffective. The impact on side effects remains unclear and more high quality trials on specific probiotic strains and side effects are thus needed.
Tóm lược
The pooled data suggest that supplementation with specific strains of probiotics compared with eradication therapy may be considered an option for increasing eradication rates, particularly when antibiotic therapies are relatively ineffective.
Full Text
The Effect of Probiotics Supplementation on Helicobacter pyloriEradication Rates and Side Effects during Eradication Therapy: A Meta-Analysis
Yini Dang1, Jan D. Reinhardt2,3,4, Xiaoying Zhou1, Guoxin Zhang1*
1 Department of Gastroenterology, The First Affiliated Hospital of Nanjing Medical University, Nanjing, China, and First Clinical Medical College of Nanjing Medical University, Nanjing, China, 2 Institute for Disaster Management and Reconstruction, Sichuan University, Chengdu, China, and Hong Kong Polytechnical University, Hung Hom, Hong Kong, China, 3 Department of Health Sciences, University of Lucerne, Lucerne, Switzerland, 4 Swiss Paraplegic Research, Nottwil, Switzerland
Introduction
‘Helicobacter pylori (H. pylori) is a common bacterium infecting about half of the world’s population. It is causally associated with a diverse spectrum of gastrointestinal disorders’ [1]. Eradication of H. pylori is necessary for the management of H. pylori-related complications. The recommended first approach for H. pylori eradication is standard triple antibiotic therapy. Other choices include sequential therapy and quadruple therapy [2]. However, due to antibiotic resistance and patient non-compliance, several studies showed widespread failure of antibiotic therapy [3,4]. Driven by the growing necessity for alternative solutions to
eradication regimens, some studies have started to focus on probiotics [5], i.e. ‘live microorganisms which when administered in adequate amounts confer a health benefit on the host’ [6]. The most commonly used probiotic bacteria belong to the genera Lactobacillus and Bifidobacterium, and these also include several yeasts such as Saccharomyces boulardii [7]. It has been hypothesized that probiotics could improve H. pylori eradication and reduce side effects during therapy.
Although seven meta-analyses [8–14] on this topic have been conducted over the past seven years, we perform an additional meta-analysis based on the following considerations: 1) The number of articles included in six of the seven previously
- Figure 1. Flow of article selection.
- doi:10.1371/journal.pone.0111030.g001
conducted meta-analysis was ten or less [9–14]. In 2010–2014, many new studies were performed to evaluate the effectiveness of probiotics supplementation increasing the evidence base. 2) There are various genera of probiotics used in clinical practice and most meta-analyses [9,11,12,13] solely concentrated on one specific strain. 3) Opposite conclusions were drawn regarding side effects: Sachdeva [10], Zou [9], and Zheng [13] report no significant reduction of overall side effects in probiotics groups. However, Tong [8], Szajewska [11], Wang [12], and Li [14] arrive at opposite conclusions. Moreover, no sensitivity analysis regarding side effects has been conducted so far. 4) While all meta-analysis conducted so far reported increased eradication rates due to probiotics supplementation, no sensitivity analysis with regard to the effectiveness of the antibiotic therapy has been conducted, i.e. it is conceivable that probiotics supplementation will solely increase effectiveness in relatively ineffective antibiotic regimens.
This meta-analysis is thus designed to evaluate the current evidence regarding effects of probiotics supplementation compared with eradication triple therapy only on H. pylori eradication rates and side effects. Moreover, we aim to compare different strains of probiotics as well as differentially effective antibiotic therapies and to evaluate the quality of the trials conducted so far.
Methods Search Strategy
We performed a literature search in Pubmed and Web of Science, covering papers published up to May 2014. A combination of the following keywords was used: (probiotics OR prebiotics OR Bifidobacterium OR Lactobacillus OR Saccharomyces) AND (Helicobacter pylori OR H. pylori). The reference lists of the selected papers and the seven previous meta-analyses were also screened for other eligible articles that may have been missed in the initial search. Next we scanned the titles and abstracts of the trials identified in the computerized search to exclude studies that were obviously irrelevant and scrutinized the full-texts of the remaining studies.
Inclusion and Exclusion Criteria
The following criteria were used for the selection of relevant articles: 1) studies should be randomized controlled trials (RCTs); 2) study populations should never have been treated for H. pylori infection before; 3) studies should include at least two branches of treatment consisting of (a) studies should have patients in a control group who received antibiotic therapy, (b) there should be patients
(57.1% (12/20)
79.2% (19/24) 86.9% (20/23)
91.7% (22/24)
57.1% (12/20)
(24/30) 40.0% (12/30)
33.3% (10/30)
80.7% (46/57) 43.3% (26/60)
61.7% (37/60)
57.1% (12/20)
74.0% (37/50) 74.0% (40/54)
82.7% (43/50)
21.3% (10/47)
patients) (treatment/
80.0% (16/20) 23.8% (5/21)
71.2% (42/59) 10.0% (6/60)
10.0% (6/60)
80.0% (16/20) 14.3% (3/21)
80.0% (16/20) 13.6% (3/22)
79.2% (27/44) 23.1% (9/39)
control)
87.5% (63/72) /
% Rradication
of Control
(19/24) PP
(63/80) PP
(16/21) PP
(42/60) PP
(42/60) PP
(16/21) PP
(16/21) PP
(37/52) PP
(27/47) PP
91.3% (21/23) ITT 79.2%
94.8% (73/77) ITT 78.8%
85.7% (18/21) ITT 76.2%
89.7% (52/58) ITT 70.0%
(25/30) ITT 80.0%
80.0% (48/60) ITT 70.0%
76.2% (16/21) ITT 76.2%
85.0% (17/20) ITT 76.2%
78.0% (39/50) ITT 71.2%
91.7% (33/36) ITT 57.4%
(no. of
% Eradication
of Treatment
(21/23) PP
(73/80) PP
(18/21) PP
(52/60) PP
(48/60) PP
(16/21) PP
(17/22) PP
(39/54) PP
(33/39) PP
patients)
ITT 91.3%
Corp., Tainan, Taiwan), bid, 5 weeks ITT 91.3%
ITT 85.7%
per capsule, tid, 10 days ITT 86.7%
Euroterapici S.p.A, Milan, Italy), bid, 14 days ITT 83.3%
Euroterapici S.p.A, Milan, Italy), bid, 14 days ITT 80.0%
Euroterapici, Milan, Italy), bid, 14 days ITT 76.2%
Beecham, Milan, Italy), bid, 14 days ITT 77.3%
OTC, Milan, Italy), tid, 14 days ITT 72.2%
14 days ITT 84.6%
(no. of
Bb99andPropionibacterium freudenreichii ssp.
shermanii JS(Valio Ltd, Helsinki, Finland), bid
during the eradication treatment and qd for
B. clausii(Enterogermina, Sanofi–Synthelabo
69 heat-killedL. acidophiluscontaining 510
Lacteol Fort (Bruschettini s.r.l., Genoa, Italy),
LactobacillusandBifidobacterium(President
Saccharomyces boulardii(Codex, SmithKline
‘6a probiotic preparation containing 6109
‘6a probiotic preparation containing 6109
‘69a probiotic preparation containing 610
‘69a probiotic preparation containing 510
‘69a probiotic preparation containing 210
‘6a combination of 5109 liveLactobacillus
65 mL milk-based fruit drink containing
rhamnosus LC705,Bifidobacterium breve
acidophilusandBiphidobacterium lactis
(Ferzym, Specchiasol, Milan, Italy), bid,
‘6200 mL AB-Yogurt containing 5109
‘69 cfu/mL ofLactobacillus GG,L.110
100 mL of fermented milk containing
‘10 cfu/mLL. casei DN-114001, qd,10
Lactobacillus GG(Giflorex, Errekappa
Lactobacillus GG(Giflorex, Errekappa
Lactobacillus GG(Giflorex, Errekappa
MethodsEradication TherapyProbiotics Supplementation
the following 3 weeks
14 days
(30 kg) bid, clarithromycin
7.5 mg/kg bid, amoxicillin
(6 weeks later) pantoprazole 40 mg bid,
ELISA (6 weeks later) lansoprazole 30 mg bid,
(8 weeks later) lansoprazole 30 mg bid,
bid, amoxicillin 1 g bid,
(6 weeks later) rabeprazole 20 mg bid,
(6 weeks later) rabeprazole 20 mg bid,
(6 weeks later) rabeprazole 20 mg bid,
(5–7 weeks later) rabeprazole 20 mg bid,
(5–7 weeks later) rabeprazole 20 mg bid,
(5–7 weeks later) rabeprazole 20 mg bid,
bid, amoxicillin 1 g tid,
bid, amoxicillin 1 g tid,
tid, amoxicillin 500 mg
bid, tinidazole 500 mg
bid, tinidazole 500 mg
bid, tinidazole 500 mg
bid, tinidazole 500 mg
bid, tinidazole 500 mg
clarithromycin 250 mg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 500 mg
25 mg/kg bid, 7 days
(15–30 kg) or 20 mg
(4 weeks later) omeprazole 10 mg
bid, 7 days
bid, 7 days
bid, 7 days
bid, 7 days
bid, 7 days
tid, 7 days
7 days
7 days
7 days
+HistologyRUT/UBT
+HpSA/UBTHpSA
++UBTELISA/UBT
++HistologyRUT
Table 1.Characteristica of studies included in the meta-analysis.
+UBTELISA/UBT
+UBTELISA/UBT
+UBTHistology/
UBT-Histology
control)Patients Diagnositic
adults UBT/UBT
adults UBT/UBT
adults UBT/UBT
adults UBT/UBT
LB[18] 2002Italy42 (21/21)asymptomatic
SM[18] 2002Italy42 (22/21)asymptomatic
MS[18] 2002Italy42 (21/21)asymptomatic
Nista [20]2004Italy106 (54/52)asymptomatic
Myllyuom [21]2005Finland47 (23/24)asymptomatic
Armuzzi a[16]2001Italy60 (30/30)asymptomatic
(Taiwan) 160 (80/80)symptomatic
Sykora[22]2005UK86 (39/47)symptomatic
Canducci [15]2000Italy120 (60/60)symptomatic
Armuzzi b 17]2001Italy120 (60/60)symptomatic
children
adults
adults
adults
adults
adults
(references)Year Area Case Number
(treatment/
Sheu [19]2002China
Cremonini
Cremonini
Cremonini
Authors
40.6% (41/101)
41.1% (69/168)
26.3% (47/179)
59.7% (37/62) 22.6% (14/62)
59.7% (37/62)
(73/96) 9.5% (10/105)
patients) (treatment/
25.0% (4/16)
66.7% (10/15) 5.9% (1/17)
control)
80.0% (16/20) /
/
84.2% (16/19) /
37.5% (12/32) /
% Rradication
(93/101) ITT 72.3% (73/
101) PP 76.0%
(165/192) PP
(129/179) PP
of Control
(16/20) PP
(10/16) PP
(37/62) PP
(16/20) PP
(12/32) PP
85.0% (17/20) ITT 80.0%
52.9% (9/17) ITT 62.5%
71.0% (44/62) ITT 59.7%
96.2% (51/53) ITT 85.9%
ITT 72.1%
89.5% (17/19) ITT 80.0%
45.5% (15/33) ITT 37.5%
(165/192)
(129/164)
(no. of
85.9%
78.6%
% Eradication
of Treatment
ITT 88.6% (93/
105) PP 92.1%
(133/168) PP
(17/20) PP
(44/62) PP
(51/53) PP
(17/20) PP
(15/33) PP
patients)
55730 (Reuterin, No´os), qd, 4 weeks ITT 85.0%
55730 (Reuterin, No´os), bid, 7 days ITT 52.9%
Turkey), bid, 14 days ITT 71.0%
andLactobacillus rhamnosus, bid, 10 days ITT 96.2%
ITT 79.2%
qd, 20 days ITT 85.0%
andLactobacillus casei ITT 45.5%
(133/152)
(9/17) PP
(no. of
87.5%
.5 cfu/mL),B. longumHY 8001 (106 cfu/10
‘..5 cfu/mL),L. caseiHY 2743 (HY 2177 (10
‘.mL), andS. thermophilusB-1 (108 cfu/mL)
150 mL Will yogurt containingL. acidophilus
Lacidofil containingLactobacillus acidophilus
(Reflor, Sanofi-Synthelabo Ilac A.S., Istanbul,
(Korea Yakult Company Limited; Chunan-Si,
‘each tablet containing 108L. reuteriATCC
‘8L. reuteriATCCeach tablet containing 10
250 mg sachets ofSaccharomyces boulardii
‘‘669 L.salivarius, 1109L. acidophilus,110
‘6Lactobacillus plantarum, 2109 L. reuterii,
8 cfu ofL. reuterieach pill containing 10
‘‘669L. caseisubsp.rhamnosus, 2109210
ATCC 55730(SD2112) (BioGaia, Sweden),
‘69Streptococcus termophilus, and510
250 mL commercial yogurt containing
69bLf 20 mg bid; Pb containing 510
Bifidobacterium infantisandB.longum,
7 cfu/mLBifidobacterium animalis10
Chungnam, S. Korea), qd, 3 weeks
‘61109 L.sporogenes, bid, 7 days
MethodsEradication TherapyProbiotics Supplementation
15 mg/kg qd, amoxicillin
(4–6 weeks later) lansoprazole 30 mg bid,
omeprazole 1 mg/kg/d,
bid, amoxicillin 1 g bid,
bid, amoxicillin 1 g bid,
500 mg bid, amoxicillin
500 mg bid, amoxicillin
500 mg bid, amoxicillin
50 mg/kg/d for 5 days;
later) omeprazole 1 mg/kg/d
plus amoxicillin 1 g, bid
clarithromycin 500 mg
clarithromycin 500 mg
tinidazole 20 mg/kg/d
(4 weeks later) rabeprazole 20 mg bid
for 5 days; rabeprazole
clarithromycin 500 mg
(8 weeks later) esomeprazole 20 mg
(4 weeks later) omeprazole 1 mg/kg
50 mg/kg qd, 7 days
(6 weeks later) pantoprazole 40 mg
(6 weeks later) lansoprazole 30 mg
for the next 5 days
bid, clarithromycin
bid, clarithromycin
bid, clarithromycin
500 mg bid for the
qd, clarithromycin
bid and tinidazole
(4 weeks later) standard PPI bid,
15 mg/kg/d and
1 g bid, 14 days
1 g bid, 10 days
plus amoxycillin
1 g bid, 7 days
clarithromycin
next 5 days
20 mg bid,
7 days
7 days
+HistologyUBT/UBT
UBT/UBT (20 days
++HistologyHpSA
++HistologyRUT
+UBT/HpSAUBT
+HpSAUBT/UBT
adults Histology/UBT
adults Histology/UBT
control)Patients Diagnositic
children UBT/UBT
adults UBT/UBT
Goldman[23]2006Argentina 65 (33/32)symptomatic
Zieminak[25]2006Poland245 (53/192)symptomatic
[28] 2008Italy40 (20/20)symptomatic
LB[30] 2008Italy33 (17/16)symptomatic
[27] 2007Italy206 (105/101)symptomatic
Kim[29]2008Korea347 (168/179)symptomatic
Lionetti[24]2006Italy40 (20/20)symptomatic
Cindoruk[26]2007Turkey124 (62/62)symptomatic
children
adults
adults
adults
(references)Year Area Case Number
(treatment/
Table 1.Cont.
De Bortoli N
Scaccianoce
Francavill
Authors
30.9% (13/42)
68.8% (22/32) 52.9% (18/34)
40.6% (13/32)
patients) (treatment/
66.7% (10/15) 20.0% (3/15)
25.0% (4/16)
71.2% (79/111) 5.2% (6/115)
3.5% (4/114)
75.6% (34/42) 8.3% (4/48)
control)
/
/
77.4% (24/31) /
/
89.1% (82/92) /
% Rradication
(237/331) PP
of Control
(79/114) PP
(18/36) PP
(20/38) PP
(24/31) PP
(34/42) PP
(10/16) PP
(22/32) PP
(82/95) PP
ITT 50.0%
ITT 52.6%
ITT 77.4%
ITT 71.6%
100% (45/45) ITT 80.9%
53.3% (8/15) ITT 62.5%
67.6% (23/34) ITT 68.8%
ITT 69.3%
77.2% (71/92) ITT 86.3%
(237/296)
(no. of
(18/36)
(20/38)
50.0%
52.6%
80.0%
% Eradication
of Treatment
(264/330) PP
(95/115) PP
(36/46) PP
(25/38) PP
(26/31) PP
(45/48) PP
(23/34) PP
(71/98) PP
patients)
BifidobacteriumDN-173 010, qd, 14 d ITT 65.8%
bid, 8 days ITT 83.9%
Korea), tid, 4 weeks ITT 80.0%
4 weeks ITT 93.8%
ITT 53.3%
.(10 cfu/g), bid, 4 weeks ITT 82.6%
boulardii, qd, 14 days ITT 72.4%
9 cfu/mLLactobacillus GG, bid, 7 daysITT 67.6%10
250 mL kefir, bid, 141dITT 78.3%
(264/309)
(8/15) PP
(95/106)
(no. of
(36/46)
(25/38)
(26/31)
78.3%
65.8%
83.9%
85.4%
89.6%
‘‘669),Bifidobacterium infantis(2109),(210
yogurt containing 112 gL. gasseriOLL2716
capsules containingsaccharomyces250 mg
Saccharomyces boulardii(Enterol; Biocodex,
6capsule containing 5109L. acidophilus,
Gentilly Cedex, France), 250 mg/day, bid,
Lacteol (BioSau´de laboratories, Portugal),
‘69), andStreptococcus termophilus(510
669),L. reuteri(2109),plantarum(510
‘63109 cfu/gSaccharomyces boulardii
‘10 cfu/g125 ml yogurt containing 10
a probiotic mixture withLactobacillus
Lactobacillus casei subsp.Rhamnosus
(Bioflor250, Kuhnil Pharmacy, Seoul,
69),Lactobacillus acidophilus(110
‘6Lactobacillus sporogenes(1109);
69),Bifidobacterium longum(210
6Lactobacillus salivarius(1109),
MethodsEradication TherapyProbiotics Supplementation
5 g/dose, bid, 7 days
10 mg/kg bid, amoxicillin
(4 weeks later) pantoprazole 40 mg bid,
esomeprazole 0.5 mg/kg
(4–6 weeks later) lansoprazole 30 mg bid,
later) lansoprazole 30 mg bid,
(5 weeks later) lansoprazole 30 mg bid,
bid, amoxicillin 1 g bid,
bid, amoxicillin 1 g bid,
bid, amoxicillin 1 g bid,
bid, amoxicillin 1 g bid,
bid, amoxicillin 1 g bid,
bid, amoxicillin 750 mg
500 mg bid, amoxicillin
(4 weeks later) omeprazole 20 mg bid,
later) rabeprazole 10 mg bid,
UBT (4–6 weeks later) omeprozole 0.5 mg/kg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 200 mg
25 mg/kg bid, 7 days
$UBT (6 weeks later) esomeprazole 20 mg
amoxicillin 25 mg/kg
bid, clarothromycin
bid, clarithromycin
bid, clarithromycin
1 g bid, 8 days
(4–6 weeks later) omeprozole or
7.5 mg/kg bid,
bid, 7–10 days
bid, 7 days
14 days
14 days
14 days
7 days
7 days
adults Histology-RUT-culture/
++HistologyRUTUBT/
+UBTHpSA (8 weeks
+Histologyculture/
adults UBT/UBT (45 days
adults Histology/HpSA
+HistologyRUT/
+HistologyRUT
adults Histology/UBT
children Histology/UBT
control)Patients Diagnositic
adults UBT/UBT
Szajewsk[32]2009Poland66 (34/32)asymptomatic
Madeiros[36]2011France62 (31/31)asymptomatic
Hurduc[31]2009Romania90 (48/42)symptomatic
MS[30] 2008Italy31 (15/16)symptomatic
Bekar[35]2011Turkey82 (46/36)symptomatic
Ozdil[37]2011Turkey193 (98/95)symptomatic
Yasar[34]2010Turkey76 (38/38)symptomatic
Song[33]2010Korea661 (330/331)symptomatic
Deguchi[38]2012Japan229 (115/114)symptomatic
children
children
adults
(references)Year Area Case Number
(treatment/
Table 1.Cont.
Scaccianoce
Authors
18.9% (17/90)
16.7% (15/90)
64.5% (20/31) 64.7% (22/34)
67.6% (23/34)
92.8% (65/70) 39.7% (29/73)
65.8% (50/76)
58.2% (32/55)
71.2% (37/52)
patients) (treatment/
61.5% (48/78) 1.3% (1/78)
0.0% (0/79)
control)
76.5% (26/34) /
/
69.7% (23/33) /
% Rradication
of Control
(73/106) PP
(26/34) PP
(23/33) PP
(48/79) PP
(73/90) PP
(20/34) PP
(65/76) PP
(44/52) PP
88.2% (30/34) ITT 76.5%
83% (83/100) ITT 68.9%
90.9% (30/33) ITT 69.7%
81.6% (62/76) ITT 60.7%
82.1% (69/84) ITT 81.1%
61.3% (19/31) ITT 58.8%
94.4% (67/71) ITT 85.5%
96.1% (49/51) ITT 84.6%
(73/106)
(no. of
(73/86)
(44/49)
68.9%
84.9%
89.8%
% Eradication
of Treatment
(83/100) PP
(30/34) PP
(30/33) PP
(62/78) PP
(69/90) PP
(19/34) PP
(67/73) PP
(49/55) PP
patients)
ITT 88.2%
ITT 90.9%
Lactobacillus acidophilus, qd, 14 d ITT 79.5%
ITT 76.7%
ITT 91.8%
ITT 89.1%
150 mg probiotic yogurt, bid, 7 daysITT 55.9%
‘69B. infantis2036, bid, 7 daysITT 83%310
(no. of
bulgaricus),Bifidobacteriumstrains (B. breveand
‘8 CFU/percapsule, bid,total viable count is 10
Balance capsue containingLactobacillusstrains
68Bifidobacterium bifidum,acidophilus, 510
‘69 CFUsStreptococcus faecium,and 1.2510
(L. casei, L. rhamnosus, L. acidophilus, andL.
B. longum), andStreptococcus thermophiles,
‘9LactobacillusMilano, Italy), containing 10
‘669L. salivarius, 1109L. acidophilus,110
‘6Lactobacillus plantarum, 2109L. reuterii,
‘669L.casei subsp.rhamnosus, 2109210
‘61.25109 CFUsLactobacillus acidophilus,
‘61.25109 CFUsLactobacillus rhamnosus,
thermophilus,Bifidobacterium infantisand
‘probiotic combination (restore, 109 cfu/
each sachet, Protexin Co, UK) containing
‘69 CFUsBifidobacterium bifidum1.2510
Lactogermine plus (Humana Italia s.p.a.,
‘6the PB supplement containing 5109
‘69Streptococcus termophilus, and510
Bifidobacterium infantisandB. longum,
109Streptococcus thermophilus, 109
Lactobacillus acidophilus,Lactobacillus
the probiotic compound containing
rhamnosus,Lactobacillus bulgaricus,
‘69L. sporogenes, qd, 7 days110
Bifidobacterium breve, qd, 7 days
Lactobacillus casei,Streptococcus
MethodsEradication TherapyProbiotics Supplementation
67probiotics, containing 310
Lactobacillus bulgaricus
bid, 30 days
14 days
bid, furazolidone 200 mg
20 mg bid, clarithromycin
(4 weeks later) omeprazole 1 mg/kg qd,
bid, tetracycline 500 mg
later) pantoprazole 40 mg qd,
(8–10 weeks later) esomeprazole 20 mg bid
for 5 days; esomeprazole
bid, amoxicillin 1 g bid,
bid, amoxicillin 1 g bid,
bid, amoxicillin 1 g bid,
240 mg bid, amoxicillin
UBT (4 weeks later) omeprazole 20 mg bid,
(4 weeks later) omeprazole 20 mg bid,
later) omeprazole 20 mg bid,
plus amoxicillin 1 g, bid
(4–8 weeks later) omeprazole 0.5 mg/kg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 500 mg
clarithromycin 500 mg
furazolidone 3 mg/kg
15 mg/kg bid, 7 days
tinidazole 500 mg bid
amoxicillin 50 mg/kg
UBT (8 weeks later) lansoprazole 30 mg
bismuth subcitrate
bid, clarithromycin
for the next 5 days
500 mg bid and
bid, amoxicillin
25 mg/kg bid,
1000 mg bid,
bid, 14 days
bid, 7 days
bid, 7 days
7 days
7 days
7 days
+HistologyHpSA/HpSA
HpSA/UBT (6–8 weeks
+++HistologyRUTUBT
+HistologyRUT/HpSA
++HistologyRUTUBT/
++HistologyRUTUBT/
+HistologyUBT/UBT
+HistologyRUT/UBT
adults UBT/UBT (4 weeks
control)Patients Diagnositic
Du YQ[39]2012China157 (78/79)symptomatic
Emirates 206 (100/106)symptomatic
Ahmad[43]2013Iran66 (33/33)symptomatic
Navarro[44]2013Brazil107 (55/52)symptomatic
Tolone[42]2012Italy68 (34/34)symptomatic
Manfredi[40]2012Italy149 (73/76)symptomatic
Mirzaee[41]2012Iran68 (34/34)symptomatic
Shavakhi[45]2013Iran180 (90/90)symptomatic
children
children
adults
adults
adults
adults
adults
(references)Year Area Case Number
(treatment/
Dajani[46]2013United
Arab
Table 1.Cont.
Authors
(no. of patients)
% Side Effects
(treatment/
control)
% Rradication
of Control
patients)
(no. of
% Eradication
of Treatment
patients)
(no. of
23.5% (12/51)
64.4% (29/45) 10.2% (5/49)
(29/51) PP
83.7% (26/43) ITT 56.9%
(36/49) PP
ITT 73.5%
each packs of probiotics containing 2 gL.
‘62 gB. bifidum-12(4.3108 cfu/100 g),
69 cfu/100 g) andacidophilus-5(4.710
,.5 years old, qd;5 years old, bid,
MethodsEradication TherapyProbiotics Supplementation
14 days
amoxicillin 30–50 mg/kg
bid, and metronidazole
(6 weeks later) PPI 0.6–0.8 mg/kg bid,
15–20 mg/kg bid for
10–15 mg/kg bid,
children, 14 days
penicillin allergic
clarithromycin
control)Patients Diagnositic
children UBT/UBT
Wang YH[47]2013China100 (49/51)symptomatic
(references)Year Area Case Number
(treatment/
doi:10.1371/journal.pone.0111030.t001
Table 1.Cont.
Authors
in an intervention group who received probiotics plus the identical antibiotic therapy; 4) there should be data on successful eradication rates available; 5) for the analysis of side effects data on occurrence of side effects during treatment were required as well. Exclusion criteria were as follows: 1) the design and the definition of the trials were obviously different from those specified above; 2) essential information was not provided; 3) papers were letters, commentaries, editorials, reviews and duplicate publications; 4) articles were written in a language other than English.
Data Extraction
Two authors extracted data independently from all eligible papers. For conflicting evaluations, another author was consulted to solve the dispute and a final decision was made by the majority of the votes. The data extracted included authors, year of publication, base characteristics of the patients, details of the H. pylori eradication therapy, details related to interventions, primary and secondary outcomes, and confirmation methods of H. pylori infection. Extracted information was entered into a database.
Assessment of Study Quality
We used the Cochrane Tool of Bias [15] to assess study quality. Two authors independently evaluated all studies. Results were then compared and discussed to form consensus. If consensus could not be reached, another author was consulted and a decision made by the majority of the votes.
Statistical Analysis
Statistical heterogeneity was analyzed with Chi-squared distributed Chochran’s Q and the I-squared statistics (I2=100%6 (Qdf)/Q). I-square indicates the percentage of variation across studies due to hetereogeneity as opposed to chance. We assumed sufficient homogeneity to apply a fixed-effects model (Mantel-Haenszel) for the meta-analysis if I-square was under 40% and/or Q was not significant at p,0.05. Otherwise, we opted for a random-effects model (DerSimonian and Laird). The jacknife was used to assess the influence of individual studies, i.e. estimation of the overall effect was repeated while omitting one study at each time. Funnel plots and Harbord’s modified test for small study effects were used to assess publication bias [16]. The influence of the probiotic strain applied (multi-strain interventions were excluded from this analysis), blinded vs. non-blinded trials, and in the case of eradication rates, the effectiveness of antibiotic therapy in the control group (eradication rate .80% vs. ,80%) was assessed with sub-group analysis. Concerning eradication rates, metaregression was used to determine the influence of studies in pediatric vs. adult as well as symptomatic and asymptomatic populations. All statistical analysis was performed with STATA 12.0 (Stata Corporation, Texas, USA).
Results Study Characteristics
The bibliographic search yielded 1114 articles, 40 of which were reviewed in full text (Figure 1) [17–56]. Of these studies, 33 RCTs [17–49] met the inclusion criteria for the analysis of eradication rates and 20 were eligible for analyzing side effects. These trials randomized a total of 4459 patients, 4261 of which were followed up. Nine studies enrolled only children [24– 26,33,34,36,44,45,49] and 24 were undertaken exclusively with adults [17–23,27–32,35,37–43,46–48]. From all the included trials, 29 used PPI-triple therapy [17–25,27–29,31–41,43– 46,48,49], three used sequential therapy [26,30,42] and one used bismuth-quadruple therapy [47]. These studies were undertaken
- Figure 2. Risk of bias graph (A): review authors’ judgements about each risk of bias item presented as percentages across all included studies. (B) Risk of bias summary: review authors’ judgements about each risk of bias item for each included study. (+) = low risk of bias, (?) = unclear, (-) = high risk of bias.
- doi:10.1371/journal.pone.0111030.g002
in Argentina [25], Brazil [46], mainland China [41,49], China (Taiwan) [21], Finland [23], France [38], Iran [43,45,47], Italy [17–20,22,26,29,30,32,42,44], Japan [40], Korea [31,35], Poland [27,34], Romania [33], Turkey [28,36,37,39], United Kingdom [24] and the United Arabian Emirates [48].(Table 1)
Trial Quality Assessment
Figure 2A shows authors’ judgements for each Cochrane risk of bias item and Figure 2B presents the Cochrane risk of bias score for each citation included.
Eradication Rates
Eradication risk ratios (RRs) were available for 4459 patients (2189 in the probiotics supplementation group and 2270 in the control group). Heterogeneity was found to be low for the overall incidence of eradication rates in ITT analysis (x2=42.97, p=0.167, I2=18.5%) but higher for PP analysis (x2=58.55, p=0.006, I2=41.9%). Therefore, a fixed effects model based on Mantel-Haenszel’s estimation method was used in the case of ITT and a random effects model in the case of PP analysis. Nonetheless, the pooled RRs from intention-to-treat (ITT) and from pre-protocol (PP) analysis for the probiotics supplementation group over controls were very similar with 1.122 (95% CI 1.086– 1.159) and 1.114 (95% CI 1.070–1.159) respectively (Figure 3A&B).
Omitting individual studies from the meta-analysis did not change occurrence of RRs being significantly higher than 1. Funnel-plot and Harbord’s modified test showed no evidence for publication bias (p =0.64). Neither the age of the population (pediatric vs. adult, p=0.127) nor symptom status (p=0.314) played a role according to the meta-regression.
Studies used different strains of probiotics with 13 applying Lactobacillus [17–20,24,26,27,30,32,34,38,40,41], two using Bifidobacterium [36,48], one using Bacillus clausii [22], five using Saccharomyces [20,28,33,35,39] and 15 using multistrain [20,21,23,26,29,31,32,37,42–47,49]. Among those studies applying individual probiotic supplementation, four specific species were effective: Lactobacillus acidophilus (pooled RR=1.235, 95% CI 1.090–1.400), Lactobacillus casei DN-114001 (pooled RR=1.473, 95% CI 1.13–1.949), Lactobacillus gasseri (pooled RR=1.192, 95% CI 1.028–1.382), Bifidobacterium infantis 2036 (pooled RR=1.205, 95% CI 1.031–1.408). While double-blinded (pooled RR=1.118, 1.045–1.196) and non-blinded trials (pooled RR=1.120, 95% CI 1.080–1.162) basically arrived at the same results, sub-group analysis by effectiveness of the antibiotic therapy in the control group revealed that supplementation with probiotics solely increased eradication rates in relatively ineffective therapies.
Another sub-group analysis was done according to the effectiveness of eradication regimens. The less effective antibiotic therapies were, the more useful probiotic supplementation was: when eradication rate ,60%, pooled RR=1.28, 1.12–1.45; when eradication rate within 60%–69%, pooled RR=1.18, 1.10–1.27; when eradication rate within 70%–79%, pooled RR=1.11, 1.06– 1.17; while if eradication rate over 80%, the supplementation was useless (pooled RR=1.01, 0.96–1.77). (Figure 4)
Side Effects
There were 20 trials (2487 patients, 1269 in the probiotics supplementation group and 1218 in the control group) which
provided data on the overall incidence of side effects. Significant heterogeneity was found for the overall occurrence of side effects (I2=72.2%, P,0.001). Therefore, the random effects model was used. The pooled RR in the probiotics supplementation over control was 0.735 (95% CI 0.598–0.902) (Figure 5). While Harbord’s modified test for publication bias was insignificant (p =0.17), the Funnel Plot did reveal some asymmetry (Figure 6): Small studies showing a considerable reduction in side-effects occur more often than those only showing a small reduction. When stratified by probiotics, only the pooled RR for Saccharomyces boulardii indicates a significant reduction in side effects (0.335, 95% CI 0.220–0.510; LB: 0.892, 95% CI 0.632–1.259; MS: 0.760, 95% CI 0.568–1.017; BB: 0.895: 0.737–1.087).
Only eight studies reporting the overall incidence of side effects were blinded and solely results from non-blinded trials provided evidence for the reduction of side-effects (pooled RR=0.589, 95% CI: 0.412–0.842), while double blinded studies did not (pooled RR=0.889, 95% CI: 0.728–1.085) (Figure 7).
Discussion
This is the largest meta-analysis on the effects of probiotics on H. pylori eradication and side effects conducted to date. The quality of the trials was medium to low and no study had a low risk of bias across all Cochrane criteria. Overall, this analysis of 33 RCTs suggests that supplementation of antibiotic therapy with probiotics increases eradication rates compared to a placebo or no intervention. However, regarding individual probiotic strains, this could only be confirmed for several strains of Lactobacilus and one strain of Bifidobacterium. Moreover, probiotics only demonstrated additional effects if the eradication rates in the control groups were relatively low, i.e. cases where the antibiotic therapy was relatively ineffective. Although, we found an overall decrease of total side effects, this held true only for Saccharomyces boulardii and nonblinded RCTs.
In comparison with previously published meta-analysis
- [8,9,10,11,12,13,14], our updated study represents the most comprehensive analysis. Various potential influence factors were taken into consideration such as age, symptom status, eradication therapy regime, eradication rate, bacterial strain and blinding method. Moreover, from the previous studies, six
- [8,10,11,12,13,14] excluded trials using Quadruple therapy as the co-intervention and none performed subgroup analysis with regard to the efficacy of antibiotic therapy. Moreover, sensitivity analysis for side effects had not been conducted so far. Our results confirm previous meta-analyses only to some extent. At a first glance, probiotics seem effective in increasing eradication rates and decreasing side effects. At a closer look, however, evidence only supports these claims for specific probiotic strains, ineffective antibiotic therapies and low-quality trials (i.e. non-blinded studies) as far as side effects are concerned.
Before Marshall found H. pylori in 1984 [57], the stomach was considered to be a sterile organ due to its low pH level. In the aftermath, ‘H. pylori has been intensively studied and recent sequencing analysis of other gastric microbiota shows that H. pylori is not alone’ [58]. Although there are only 10
2-4 cfu/g in the gastric mucosa [59], these commensal bacteria can play an important role in maintaining human health. Probiotic supple-
‘
- Figure 3. The effect of probiotics supplementation vs. without probiotics on eradication rates by intention-to-treat analysis in (A). RR, risk ratio; CI, confidence interval. The triangles represent individual studies and the size of the triangle represents the weight given to each study in the meta-analysis. The diamond represents the combined results. (B) The effect of probiotics supplementation vs. without probiotics on eradication rates by pre-protocol analysis. RR, risk ratio; CI, confidence interval. The triangles represent individual studies and the size of the triangle represents the weight given to each study in the meta-analysis. The diamond represents the combined results.
mentation as an approach to manipulate gastrointestinal flora has been intensively studied.
While all previously conducted meta-analysis concluded that supplementation of antibiotic therapy with probiotics is effective in increasing eradication rates [8,9,10,11,12], our study identified only four effective individual strain: Lactobacillus acidophilus (Pubmed ID: 190198), Lactobacillus casei DN-114001 (Pubmed ID: 35628), Lactobacillus gasseri (Pubmed ID: 35528), Bifidobac-
terium infantis 2036 (Pubmed ID: 41468). However, more than one trial was conducted only for Lactobacillus acidophilus so far, limiting the generalizability of the findings regarding the other three strains. While Lactobacillus GG had previously been reported to be an effective strain [8], we do reach the same conclusion here.
Moreover, our results suggest that probiotic supplementation is only useful in less effective (eradication rate ,80%) antibiotic
- Figure 5. The effect of probiotics supplementation vs. without probiotics on total side effects. RR, risk ratio; CI, confidence interval. The triangles represent individual studies and the size of the triangle represents the weight given to each study in the meta-analysis. The diamond represents the combined results.
therapies. Acceptable success rates have often been defined as 80% or more on an ITT basis [2]. In those effective regimens, probiotic supplementation may not be needed. On the other hand, however, increasing resistance to antibiotics, for example clarithromycin, has been noted [2]. Probiotics supplementation may be a potential approach for eliminating resistant strains. However, no study included in this meta-analysis reported the detection of antibiotic resistance. Further research is warranted to clarify this issue.
The occurrence of adverse effects is one of the major drawbacks of antibiotic treatment. Although antibiotics may modify the composition of intestinal bacteria, broad spectrum antibiotics also often lead to gastrointestinal side effects [20,60]. Results regarding the effectiveness of probiotics in reducing side effects from previous meta-analyses have been inconclusive [8,9,10,11,12]. While we found that probiotics reduce the overall occurrence of side effects in the pooled data, this result must be taken with caution. Only one specific strain significantly reduced side effects and this overall result was only confirmed for non-blinded trials. While eradication rates can be determined with objective measures, assessment of side effects must rely on patients’ subjective reporting. When patients are not blinded, their reports are likely to be biased due to awareness of a respective intervention.
- Figure 7. Meta-analysis of side effects by double-blinded and nondouble-blinded trials. RR, risk ratio; CI, confidence interval. The triangles represent individual studies and the size of the triangle represents the weight given to each study in the meta-analysis. The diamond represents the combined results.
- doi:10.1371/journal.pone.0111030.g007
Limitations of this meta-analysis include a limited number of high quality trials that could be analyzed, particularly regarding individual probiotic strains and side effects. Only one controlled trial was conducted for the following specific species Bacillus clausii, Bifidobacterium DN-173 010, Lactobacillus casei DN114001, Lactobacillus gasseri, Bifidobacterium infantis 2036. Accordingly, results regarding those strains need to be interpreted with caution. Thirteen of the analyzed trials did not provide data on overall occurrence of side effects. Moreover, tools used for measuring side effects and reporting individual side effects largely differed across studies. From 20 studies reporting on the overall incidence of side effects, only eight were blinded.
Conclusions
The pooled data suggests that supplementation with specific strains of probiotics compared with eradication therapy may be considered as an option for increasing eradication rates of H.
References
- 1. Go MF (2002) Review article: natural history and epidemiology of Helicobacter pylori infection. Aliment Pharmacol Ther 16: 3–15.
- 2. Malfertheiner P, Meqraud F, O’Morain CA, Atherton J, Axon AT, et al. (2012) Management of Helicobacter pylori infection–the Masstricht IV/Florence Consensus Report. Gut 61: 646–64.
pylori, particularly when antibiotic therapies are relatively ineffective.
Supporting Information
Checklist S1 PRISMA Checklist. (DOC)
Acknowledgments
We acknowledge the help of Dr. Andrew Pennycott in correcting the manuscript for grammar and style.
Author Contributions
Conceived and designed the experiments: YD GZ. Performed the experiments: YD XZ. Analyzed the data: YD JDR. Contributed reagents/materials/analysis tools: XZ. Wrote the paper: YD JDR GZ.
- 3. Meqraud F (2004) Basis of the management of drug-resistant Helicobacter pylori infection. Drugs 64: 1893–904.
- 4. Yoqueswaran K, Chen G, Cohen L, Cooper MA, Yong E, et al. (2011) How well is Helicobacter pylori treated in usual practice? Can J Gastroenterol 25: 543–6.
- 5. Lionetii E, Indrio F, Pavone L, Borrello G, Cavallo L, et al. (2010) Role of probiotics in pediatric patients with Helicobacter pylori infection: a comprehensive review of the literature. Helicobacter 15: 79–87.
- 6. FAO/WHO (2001) Report on Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria. Cordoba, FAO/WHO, 2001.
- 7. Saxelin M, Tynkkynen S, Mattila-Sandholm T, de Vos WM (2005) Probiotic and other functional microbes: from markets to mechanisms. Curr Opin Biotechnol 16: 204–11.
- 8. Tong JL, Ran ZH, Shen J, Zhang CX, Xiao SD (2007) Meta-analysis: the effect of supplementation with probiotics on eradication rates and adverse events during Helicobacter pylori eradication therapy. Aliment Pharmacol Ther 25: 155–68.
- 9. Zou J, Dong J, Yu X (2009) Meta-analysis: Lactobacillus containing quadruple therapy versus standard triple first-line therapy for Helicobacter pylori eradication. Helicobacter 14: 449–459.
- 10. Sachdeva A, Naqpal J (2009) Effect of fermented milk-based probiotic preparations on Helicobacter pylori eradication: a systematic review and metaanalysis of randomized-controlled trials. Eur J Gastroenterol Hepatol 21: 45–53.
- 11. Szajewska H, Horvath A, Piwowarczyk A (2010) Meta-analysis: the effects of Saccharomyces boulardii supplementation on Helicobacter pylori eradication rates and side effects during treatment. Aliment Pharmacol Ther 32: 1069–79.
- 12. Wang ZH, Gao QY, Fang JY (2013) Meta-analysis of the efficacy and safety of Lactobacillus-containing and Bifidobacterium-containning probiotic compound preparation in Helicobacter pylori eradication therapy. J Clin Gastroenterol 47: 25–32.
- 13. Zheng X, Lyu L, Mei Z (2013) Lactobacillus-containingprobiotic supplementation increases Helicobacter plyori eradication rate: evidence from a metaanalysis. Rev Esp Enferm Dig 105: 445–53.
- 14. Li S, Huang XL, Sui JZ, Chen SY, Xie YT, et al. (2014) Meta-analysis of randomized controlled trails on the efficacy of probiotics in Helicobacter pylori eradication therapy in children. Eur J Pediatr 173: 153–61.
- 15. Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, et al. (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomized trials. BMJ 343: d5928.
- 16. Roger MH, Ross JH, Jonathan AC (2009) Updated tests for small-study effects in meta-analyses. The Stata Journal 9: 197–210.
- 17. Canducci F, Armuzzi A, Cremonini F, Cammarota G, Bartolozzi F, et al. (2000) A lyophilized and inactivated culture of Lactobacillus acidophilus increases Helicobacter pylori eradication rates. Aliment Pharmacol Ther 14: 1625–9.
- 18. Armuzzi A, Cremonini F, Bartolozzi F, Canducci F, Candelli M, et al. (2001) The effect of oral administration of Lactobacillus GG on antibiotic-associated gastrointestinal side-effects during Helicobacter pylori eradication therapy. Aliment Pharmacol Ther 15: 163–9.
- 19. Armuzzi A, Cremonini F, Ojetti V, Bartolozzi F, Canducci F, et al. (2001) Effect of Lactobacillus GG supplementation on antibiotic-associated gastrointestinal side effects during Helicobacter pylori eradication therapy: a pilot study. Digestion 63: 1–7.
- 20. Cremonini F, Di Caro S, Covino M, Armuzzi A, Gabrielli M, et al. (2002) Effect of different probiotic preparations on anti-Helicobacter pylori therapy-related side effects: a parallel proup, triple blind, placebo-controlled study. Am J Gastroenterol 97: 2744–9.
- 21. Sheu BS, Wu JJ, Lo CY, Wu HW, Chen JH, et al. (2002) Impact of supplement with Lactobacillus- and Bifidobacterium-containing yogurt on triple therapy for Helicobacter pylori eradication. Aliment Pharmacol Ther 16: 1669–75.
- 22. Nista EC, Candelli M, Cremonini F, Cazzato IA, Zocco MA, et al. (2004) Bacillus clausii therapy to reduce side-effects of anti-Helicobacter pylori treatment: randomized, double-blind, placebo controlled trial. Aliment Pharmacol Ther 20: 1181–8.
- 23. Myllyluoma E, Veijola L, Ahlroos T, Tynkkynen S, Kankuri E, et al. (2005) Probiotic supplementation improves tolerance to Helicobacter pylori eradication therapy-a placebo-controlled, double-blind randomized pilot study. Aliment Pharmacol Ther 21: 1263–72.
- 24. Sykora J, Valeckova K, Amlerova J, Siala K, Dedek P, et al. (2005) Effects of a specially designed fermented milk product containing probiotic Lactobacillus casei DN-114 001 and the eradication of H. pylori in children: a prospective randomized double-blind study. J Clin Gastroenterol 39: 692–8.
- 25. Goldman CG, Barrado DA, Balcarce N, Rua EC, Oshiro M, et al. (2006) Effect of a probiotic food as an adjuvant to triple therapy for eradication of Helicobacter pylori infection in children. Nutrition 22: 984–8.
- 26. Lionetti E, Miniello VL, Castellaneta SP, Maqista AM, de Canio A, et al. (2006) Lactobacillus reuteri therapy to reduce side-effects during anti-Helicobacter pylori treatment in children: a randomized placebo controlled trial. Aliment Pharmacol Ther 24: 1461–8.
- 27. Ziemniak W (2006) Efficacy of Helicobacter pylori eradication taking into account its resistance to antibiotics. J Physiol Pharmacol 57: 123–41.
- 28. Cindoruk M, Erkan G, Karakan T, Dursun A, Unal S (2007) Efficacy and safety of Saccharomyces boulardii in the 14-day triple anti-Helicobacter pylori therapy: a prospective randomized placebo-contrilled double-blind study. Helicobacter 12: 309–16.
- 29. de Bortoli N, Leonardi G, Ciancia E, Merlo A, Bellini M, et al. (2007) Helicobacter pylori eradication: a randomized prospective study of triple therapy versus triple therapy plus lactoferrin and probiotics. Am J Gastroenterol 102: 951–6.
- 30. Francavilla R, Lionetii E, Casrellaneta SP, Maqusta AM, Mauroqivanni G, et al.
- 31. Kim MN, Kim N, Lee SH, Park YS, Hwang JH, et al. (2008) The effects of probiotics on PPI-triple therapy for Helicobacter pylori eradication. Helicobacter 13: 261–8.
- 32. Scaccianoce G, Zullo A, Hassan C, Gentili F, Cristofari F, et al. (2008) Triple therapies plus different probiotics for Helicobacter pylori eradication. Eur Rev Med Pharmacol Sci. 12: 251–6.
- 33. Hurduc V, Plesca D, Dragomir D, Sajin M, Vandenplas Y (2009) A randomized, open trial evaluating the effect of Saccharomyces boulardii on the eradication rate of Helicobacter pylori infection children. Acta Paediatr 98: 127–31.
- 34. Szajewska H, Albrecht P, Topczewska-Cabanek A (2009) Randomized, doubleblind, placebo-controlled trial: effect of lactobacillus GG supplementation on Helicobacter pylori eradication rates and side effects during treatment in children. J Pediatr Gastroenterol Nutr 48: 431–6.
- 35. Song MJ, Park DI, Park JH, Kim HJ, Cho YK, et al. (2010) The effect of probiotics and mucoprotective agents on PPI-based triple therapy for eradication of Helicobacter pylori. Helicobacter 15: 206–13.
- 36. Yasar B, Abut E, Kayadibi H, Toros B, Sezikli M, et al. (2010) Efficacy of probiotics in Helicobacter pylori eradication therapy. Turk J Gastroenterol 21: 212–7.
- 37. Bekar O, Yilmaz Y, Gulten M (2011) Kefir improves the efficacy and tolerability of triple therapy in eradicating Helicobacter pylori 14: 344–7.
- 38. Medeiros JA, Goncalves TM, Boyanova L, Pereira MI, de Carvalho JN, et al.
- 39. Ozdil K, Calhan T, Sahin A, Senates E, Kahraman R, et al. (2011) Levofloxacin based sequential and triple therapy compared with standard plus probiotic combination for Helicobacter pylori eradication. Hepatogastroenterology 58: 1148–52.
- 40. Deguchi R, Nakaminami H, Rimbara E, Noquchi N, Sasatsu M, et al. (2012) Effect of pretreatment with Lactobacillus gasseri OLL2716 on first-line Helicobacter pylori eradication therapy. J Gastroenterol Hepatol 27: 888–92.
- 41. Du YQ, Su T, Fan JG, Lu YX, Zheng P, et al. (2012) Adjuvant probiotics improve the eradication effect of triple therapy for Helicobacter pylori infection. World J Gastroenterol 18: 6302–7.
- 42. Manfredi M, Bizzarri B, Sacchero RI, Maccari S, Calabrese L, et al. (2012) Helicobacter pylori infection in clinical practice: probiotics and a combination of probiotics + lactoferrin improve compliance, but not eradication, in sequential therapy. Helicobacter 17: 254–63.
- 43. Mizaee V, Rezahosseini O (2012) Randomized control trial: Comparison of Triple Therapy plus Probiotic Yogurt vs. Standard Triple Therapy on Helicobacter pylori Eradication. Iran Red Crescent Med J 14: 657–66.
- 44. Tolone S, Pellino V, Vitaliti G, Lanzafame A, Tolone C (2012) Evaluation of Helicobacter pylori eradication in pediatric patients by triple therapy plus lactoferrin and probiotics compared to triple therapy alone. Ital J Pediatr 38: 63.
- 45. Ahmad K, Fatemeh F, Mehri N, Maryam S (2013) Probiotics for the treatment of pediatric Helicobacter pylori infection: a randomized double blind clinical trial. Iran H Pediatr 23: 79–84.
- 46. Navarro-Rodriquez T, Silva FM, Barbuti RC, Mattar R, Moraes-Filho JP, et al.
- 47. Shavakhi A, Tabesh E, Yaqhoutkar A, Hashemi H, Tabesh F, et al. (2013) The effects of multistrain probiotics compound on bismuth-containing quadruple therapy for Helicobacter pylori infection: A randomized placebo-controlled triple-blind study. Helicobacter 18: 280–4.
- 48. Dajani AI, Abu Hammour AM, Yang DH, Chung PC, Nounou MA, et al.
- 49. Wang YH, Huang Y (2013) Effect of Lactobacillus acidophilus and Bifidobacterium bifidum supplementation to standard triple therapy on Helicobacter pylori eradication and dynamic changes in intestinal flora. World J Microbiol Biotechnol 30: 847–53.
- 50. Sheu BS, Cheng HC, Kao AW, Wang ST, Yang YJ, et al. (2006) Pretreatment with Lactobacillus- and Bifidobacterium-containing yogurt can improve the efficacy of quadruple therapy in eradicating residual Helicobacter pylori infection after failed triple therapy. Am J Clin Nutr 83: 864–9.
- 51. Boonyaritichaikij S, Kuwabara K, Nagano J, Naqano J, Kobayashi K, et al.
- 52. Yoon H, Kim N, Kim JY, Park SY, Park JH, et al. (2011) Effects of multistrain probiotic containing yogurt on second-line triple therapy for Hp infection. J Gastroenterol Hepatol 26: 44–8.
- 53. Selinger CP, Bell A, Cairns A, Lockett M, Sebastian S, et al. (2013) Probiotics VSL#3 prevents antibiotic-associated diarrhea in a double-blind, randomized, placebo-controlled clinical trial. J Hosp Infect 84: 159–65.
- 54. Mehling H, Busjahn A (2013) Non-viable Lactobacillus reuteri DSMZ 17648
- 55. Dore MP, Cuccu M, Pes GM, Manca A, Graham DY (2014) Lactobacillus reuteri in the treatment of Helicobacter pylori infection. Intern Emerg Med 9: 649–54.
- 56. Francavilla R, Polimeno L, Demichina A, Mauroqiovanni G, Principi B, et al.
(2014) Lactobacillus reuteri strain combination in Helicobacter pylori infection: a randomized, double-blind, placebo-controlled study. J Clin Gastroenterol 48: 407–13.
- 57. Marshall BJ, Warren JR (1984) Unidentified curved bacilli in the stomach of patients with gastritis and peptic ulceration. Lancet 1: 1311–5.
- 58. Walker MM, Talley NJ (2014) Review article: bacteria and pathogenesis of disease in the upper gastrointestinal tract—beyond the era of Helicobacter pylori. Aliment Pharmacol Ther 39: 767–79.
- 59. Delqado S, Cabrera-Rubio R, Mira A, Suarez A, Mayo B (2013) Microbiological survey of the human gastric ecosystem using culturing and pyrosequencing methods. Microb Ecol 65: 763–72.
- 60. Patel A, Shah N, Prajapati JB (2014) Clinical appliance of probiotics in the treatment of Helicobacter pylori infection-A brief review. J Microbiol Immunol Infect 47: 429–37.
Figures
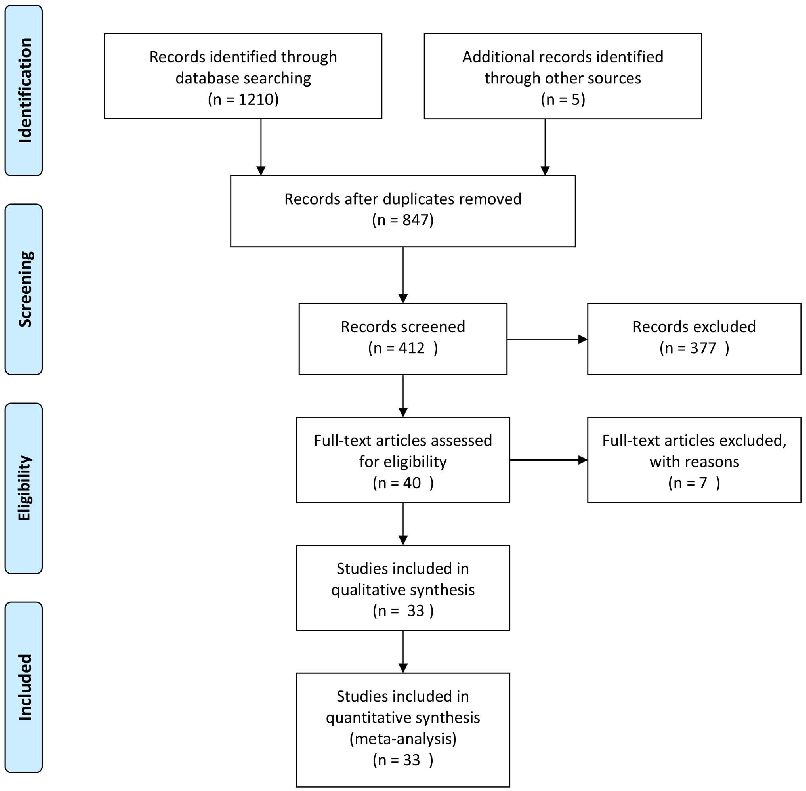
Figure 2
Forest plot of pooled H. pylori eradication rates demonstrates a modest but statistically significant improvement when probiotics are added to standard antibiotic therapy. Effect sizes vary across individual studies depending on probiotic strains used.
forest_plot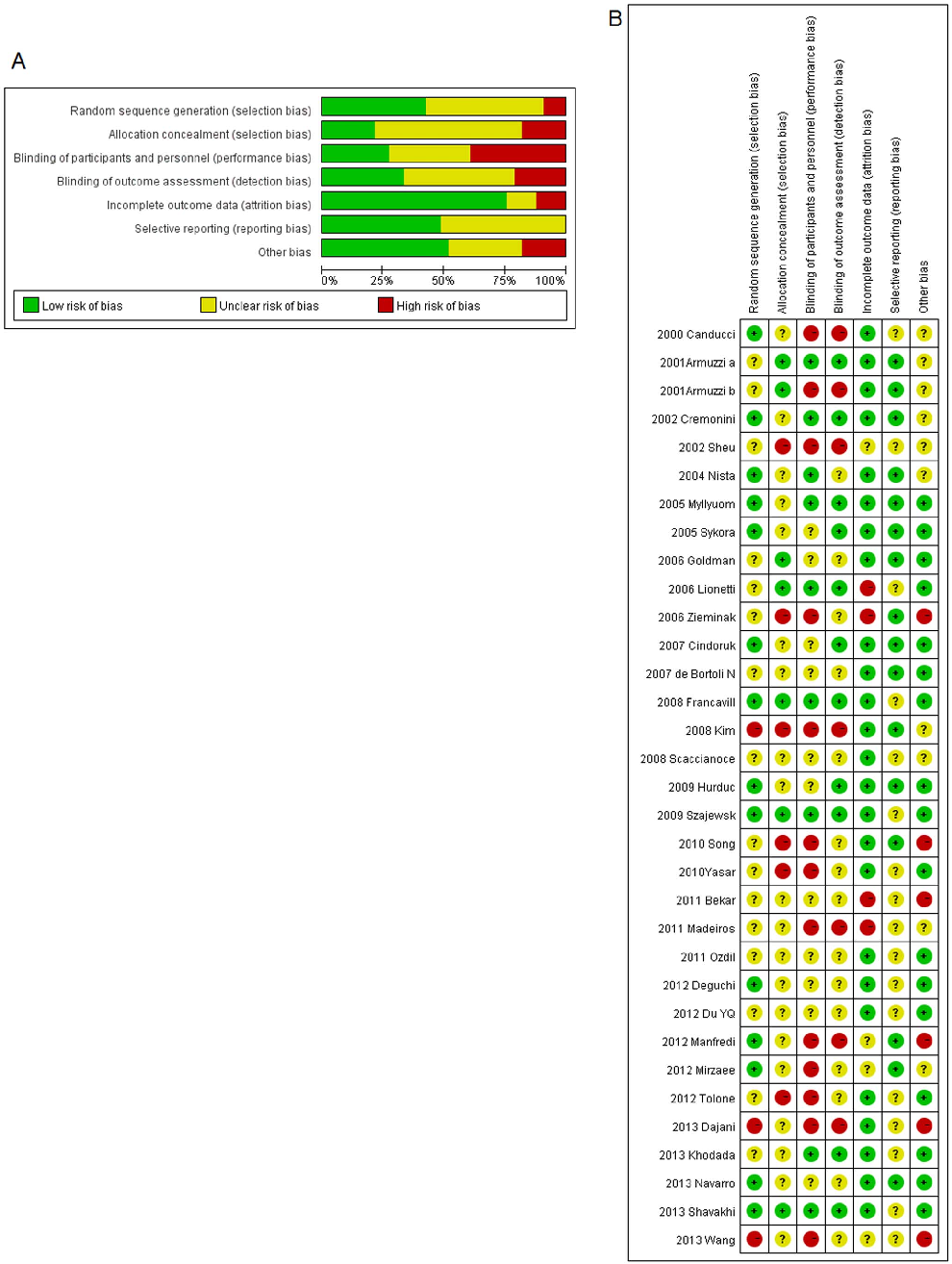
Figure 3
Subgroup analysis by probiotic species reveals that Lactobacillus-based preparations and multi-strain combinations show the most consistent benefit for improving eradication rates. Single-strain Saccharomyces boulardii also demonstrates efficacy.
forest_plot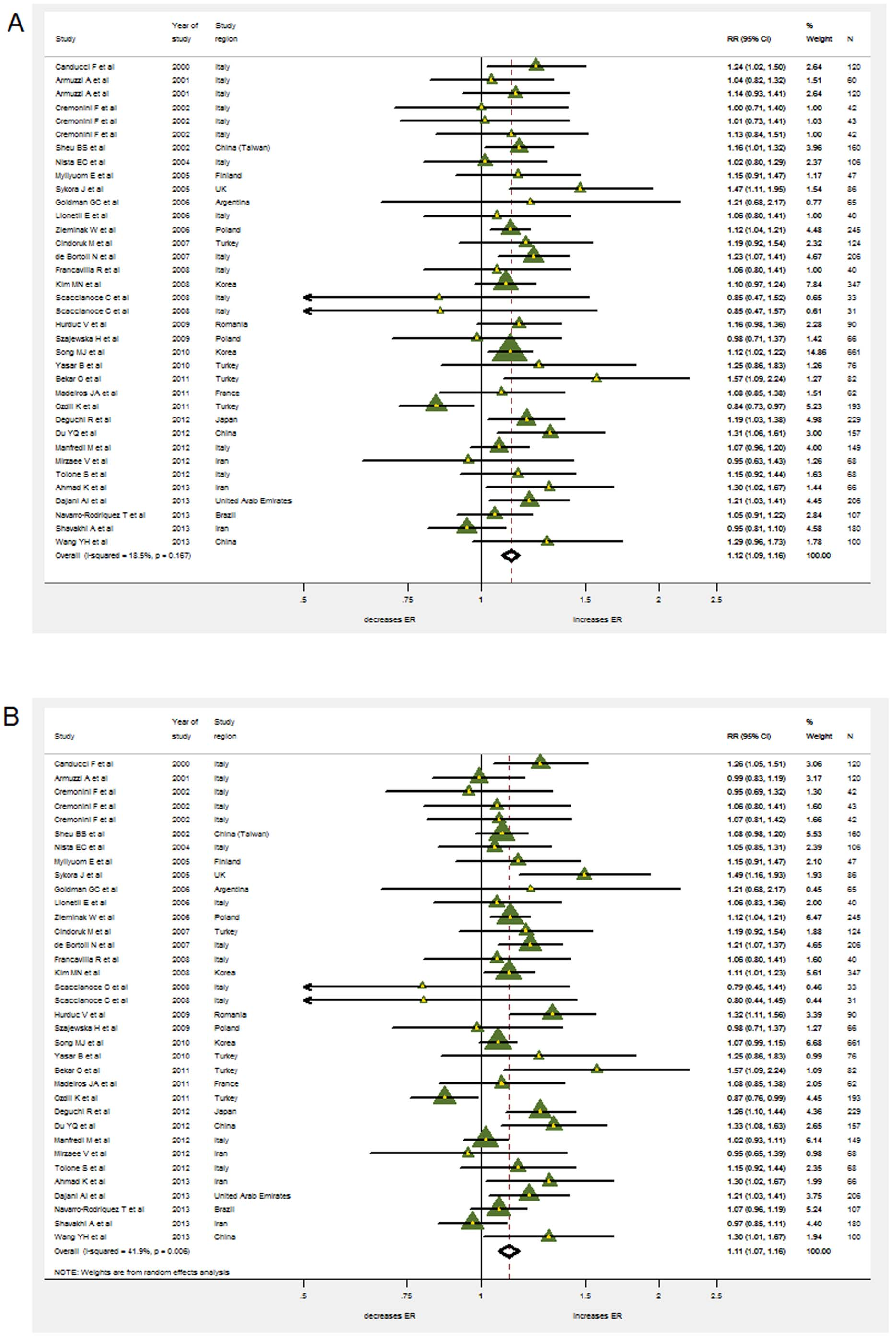
Figure 4
Overall side effect incidence is significantly reduced in patients receiving adjunctive probiotic supplementation during H. pylori eradication therapy. Diarrhea, nausea, and abdominal pain are the adverse events most consistently ameliorated.
forest_plot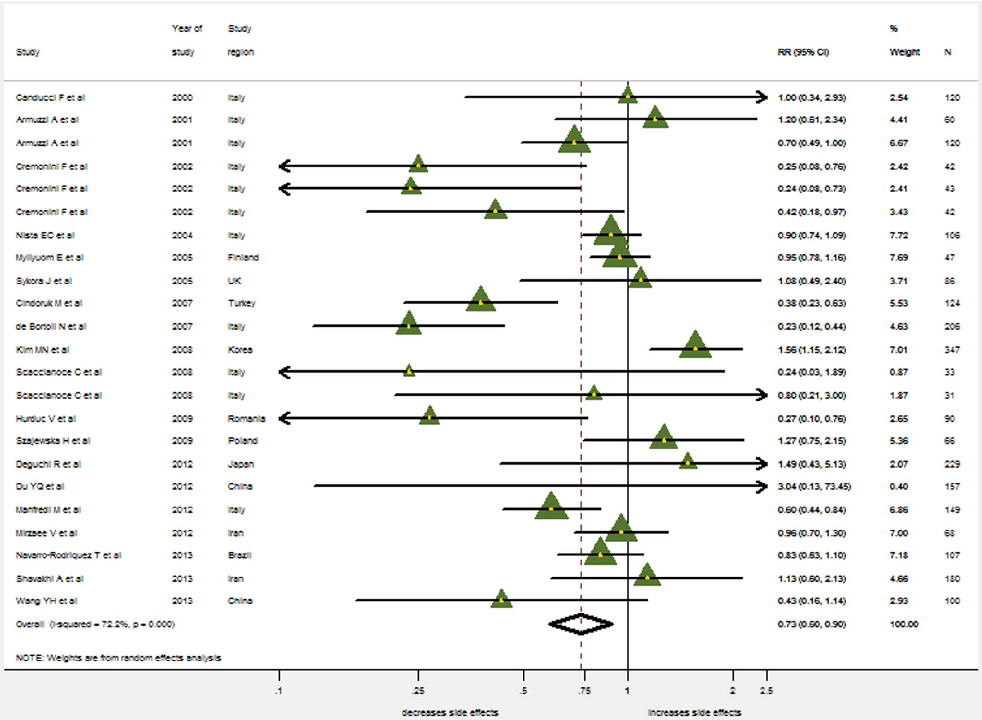
Figure 5
Funnel plot assessment for publication bias in the meta-analysis shows relatively symmetric distribution of studies, suggesting minimal publication bias in the included eradication rate trials.
chart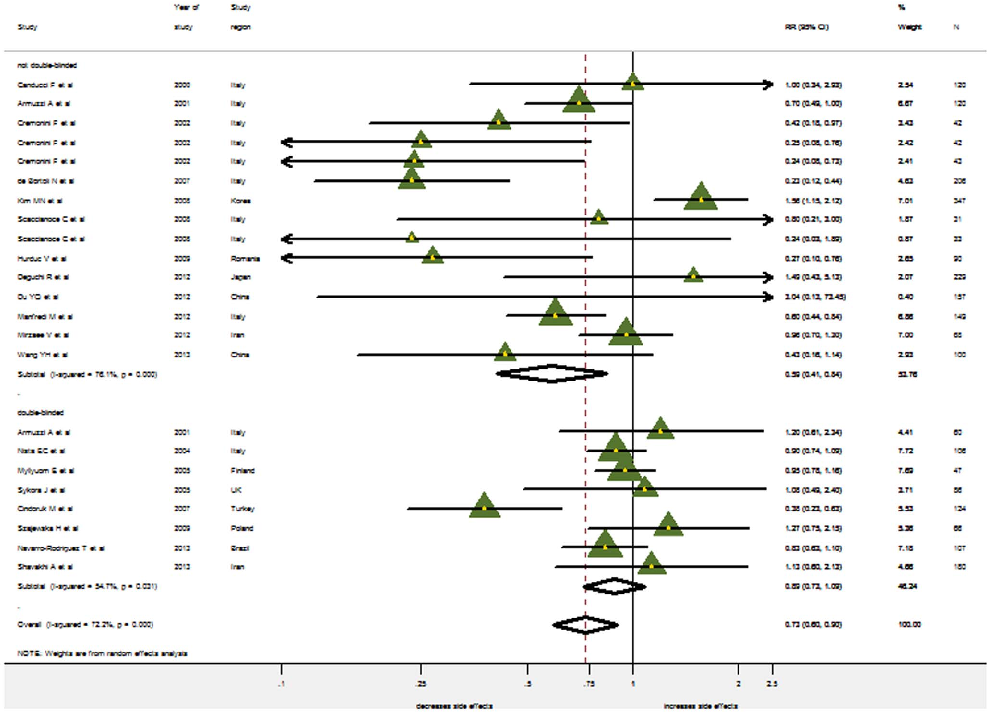
Figure 6
Sensitivity analyses confirm the robustness of the pooled treatment effect, with no single study disproportionately influencing the overall result. Risk of bias assessment across included trials indicates moderate quality evidence.
chartUsed In Evidence Reviews
Similar Papers
The Journal of antimicrobial chemotherapy · 2001
Suppressive effect of Lactobacillus gasseri OLL 2716 (LG21) on Helicobacter pylori infection in humans.
FEMS microbiology reviews · 2013
Genomic and phenotypic evidence for probiotic influences of Lactobacillus gasseri on human health.
Letters in applied microbiology · 2010
Oral administration of lactobacilli from human intestinal tract protects mice against influenza virus infection.
Frontiers in microbiology · 2014
Probiotic and technological properties of Lactobacillus spp. strains from the human stomach in the search for potential candidates against gastric microbial dysbiosis.
Journal of gastroenterology and hepatology · 2003
Lactobacillus gasseri OLL2716 as a probiotic in clarithromycin-resistant Helicobacter pylori infection.
Journal of gastroenterology and hepatology · 2012