N-Acetyl Cysteine (NAC) Hình ảnh
43 hình ảnh từ nghiên cứu có bình duyệt
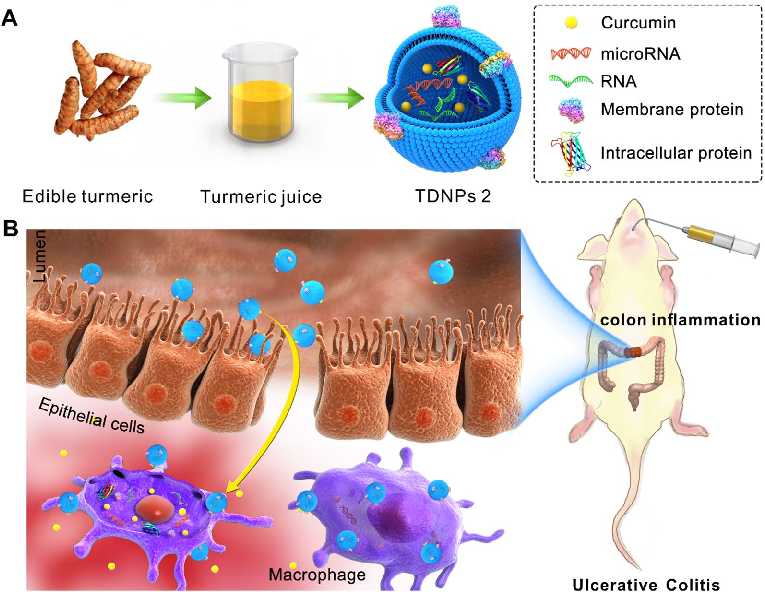
Turmeric-derived nanoparticles (TDNPs 2) are isolated through sucrose gradient ultracentrifugation and administered orally to target inflamed colonic tissue in a murine colitis model. The schematic outlines the isolation workflow from edible turmeric to purified nanovesicles.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
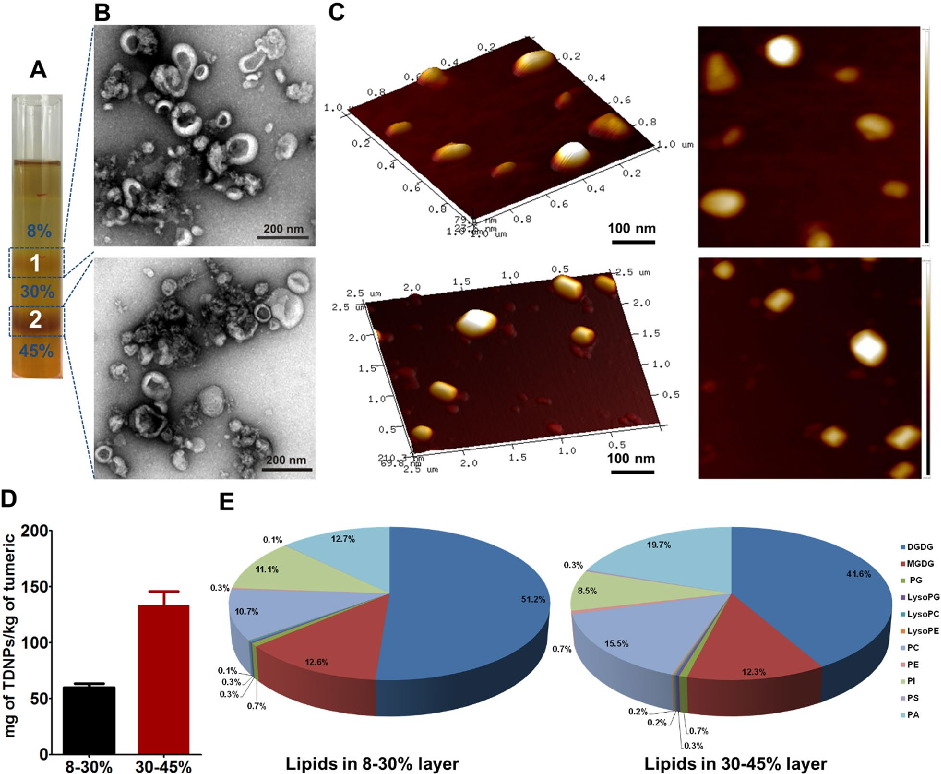
Characterization of turmeric-derived nanoparticles reveals two distinct bands (TDNPs 1 and TDNPs 2) at the 8%/30% and 30%/45% sucrose gradient interfaces, respectively. TDNPs 2 demonstrate appropriate size distribution and surface charge for oral drug delivery applications.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
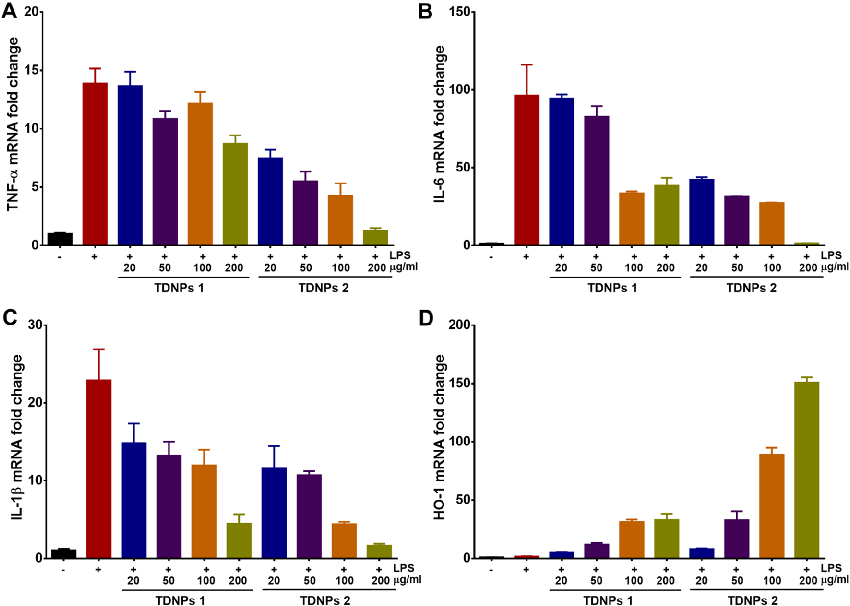
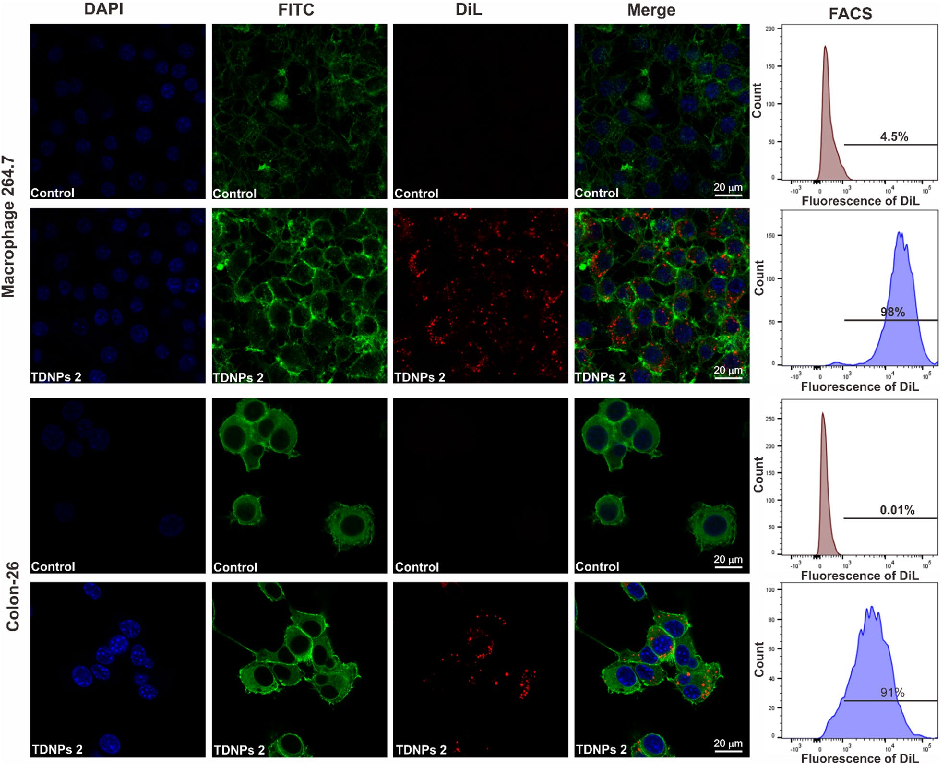
In vitro assessment of turmeric-derived nanovesicles demonstrates anti-inflammatory activity, including suppression of pro-inflammatory cytokine production in activated macrophages. Dose-dependent reductions in TNF-alpha and IL-6 secretion are observed.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
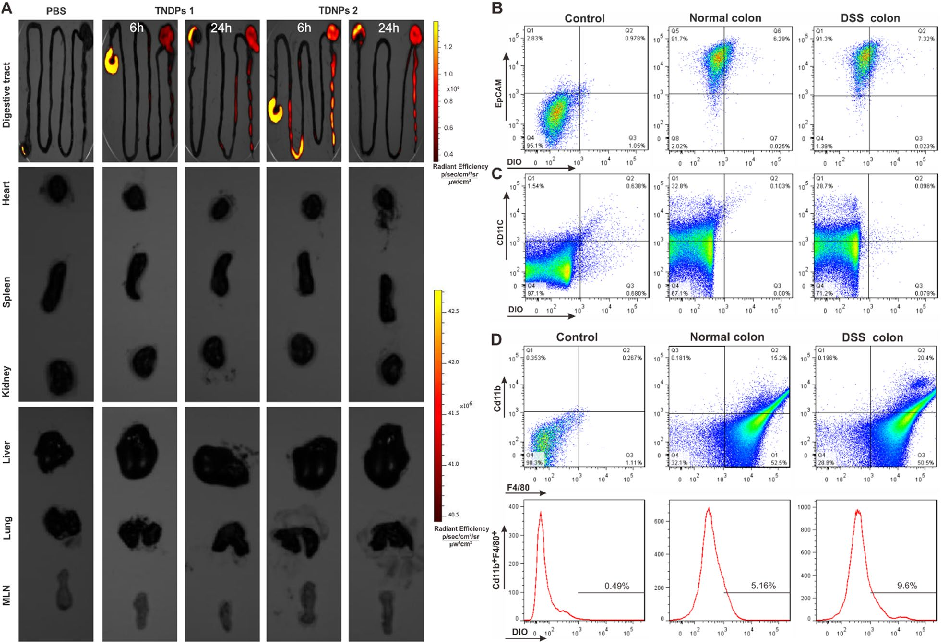
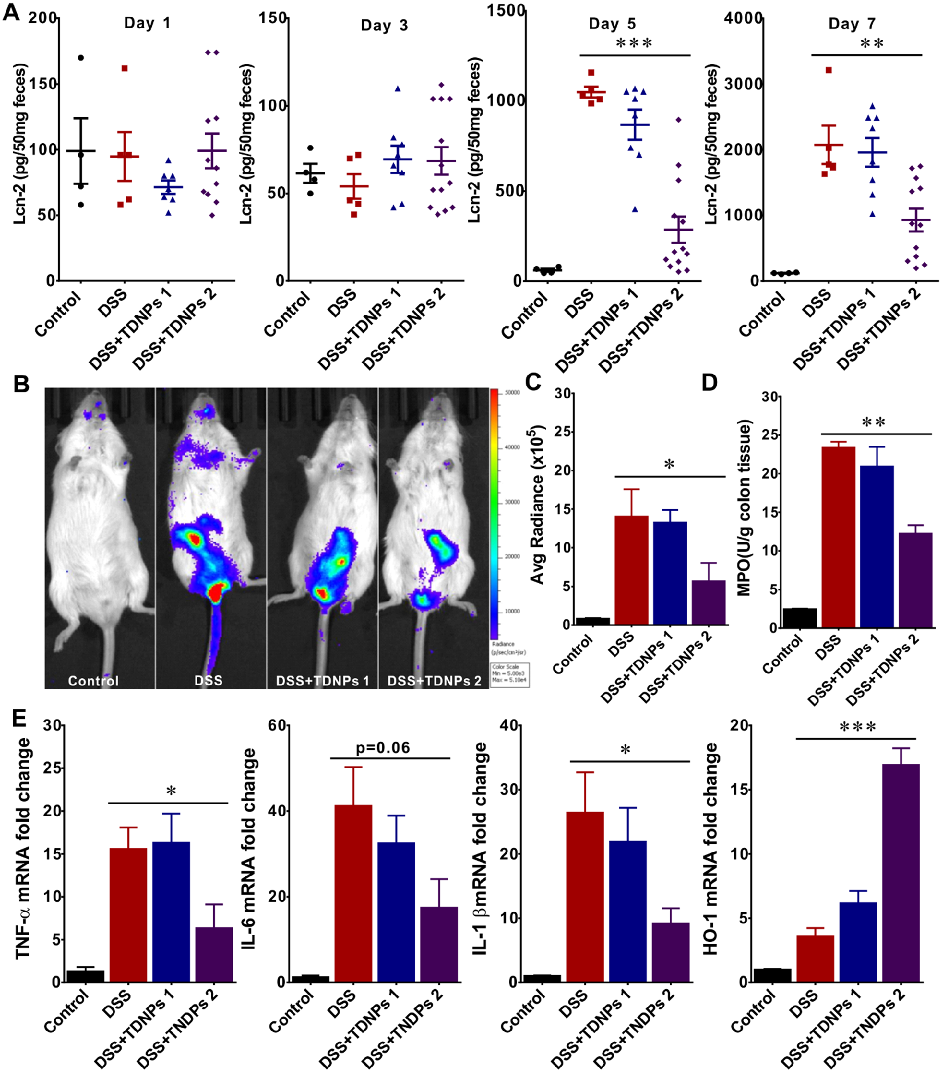
Biodistribution imaging using IVIS Spectrum reveals that TDNPs 2 preferentially accumulate in inflamed colonic tissue following oral administration. Fluorescence signals are minimal in non-target organs including heart, liver, spleen, lung, and kidney.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
Oral TDNPs 2 administration significantly attenuates disease activity index scores and colon shortening in DSS-induced colitis mice. Body weight recovery is also improved compared to untreated colitis controls.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
Intestinal permeability assessment indicates that TDNPs 2 treatment preserves gut barrier integrity in colitis mice. Tight junction protein expression, including ZO-1 and occludin, is maintained at near-normal levels.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
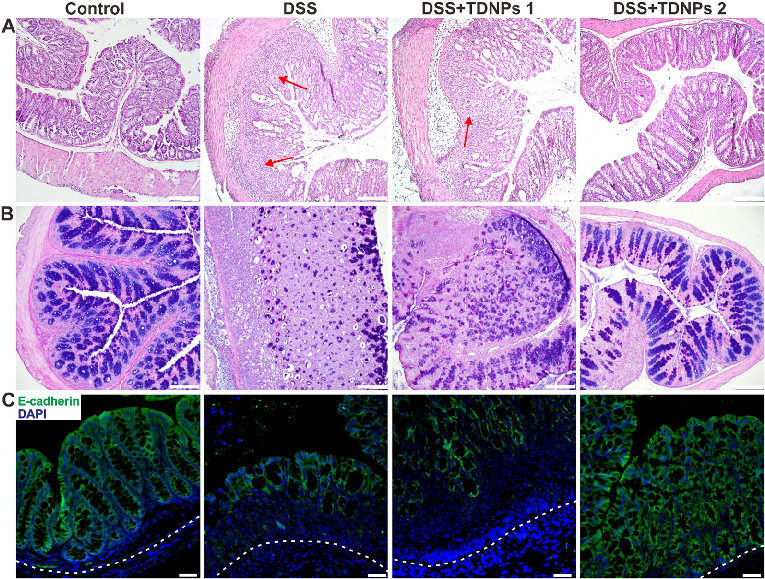
Histological examination with H&E staining reveals markedly reduced inflammatory cell infiltration and preserved goblet cell density in TDNPs 2-treated colitic mice. Colonic tissue architecture remains largely intact compared to severe disruption in untreated animals.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
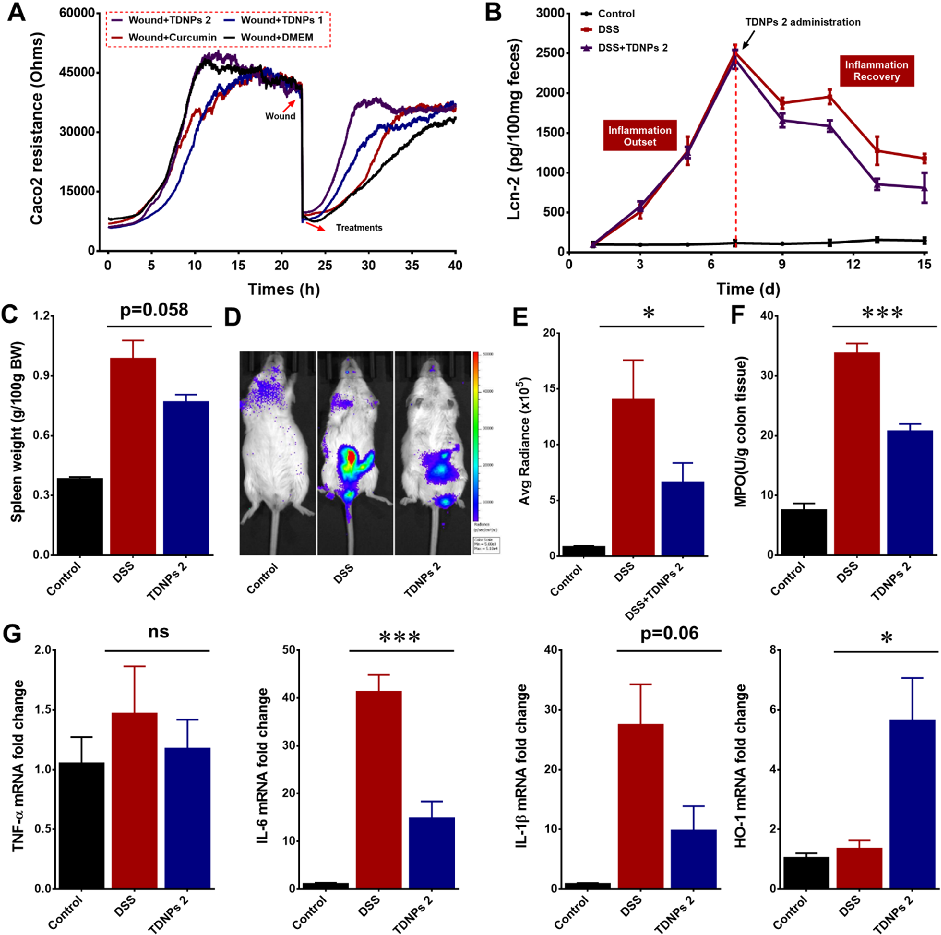
Wound healing assays and fecal lipocalin-2 quantification demonstrate that TDNPs 2 accelerate resolution of intestinal inflammation. Lcn-2 levels, a sensitive marker of intestinal inflammation, decrease significantly with treatment.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
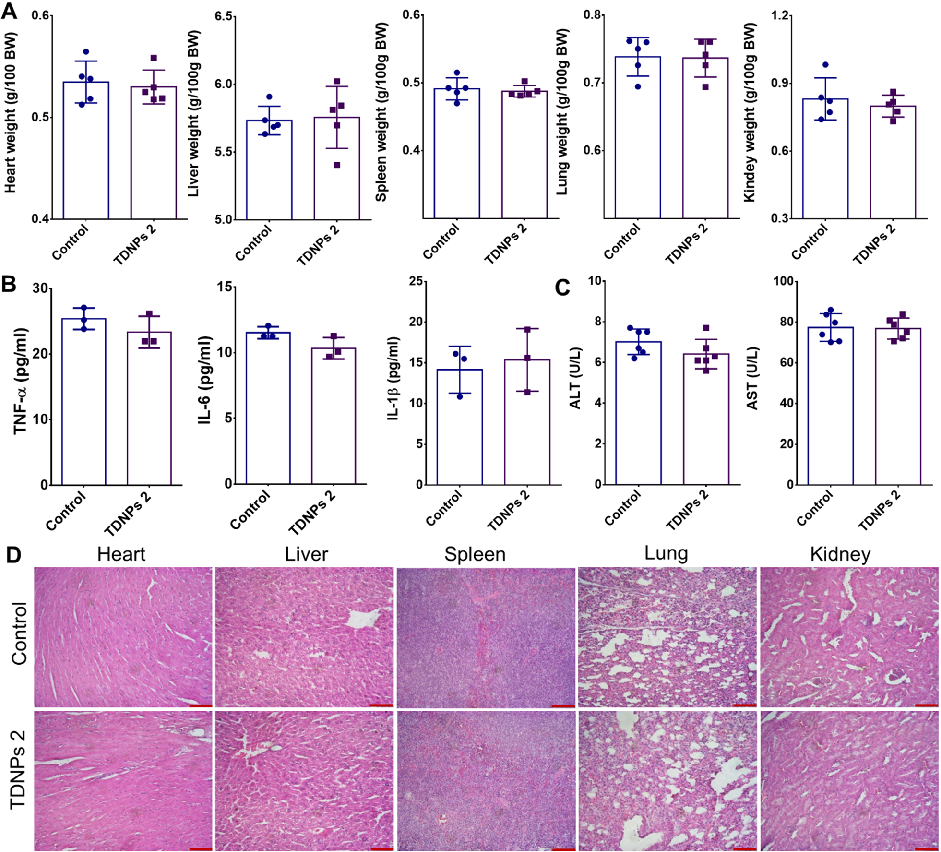
Biocompatibility evaluation shows no significant changes in vital organ weights, pro-inflammatory cytokines, or liver function indicators in TDNPs 2-treated mice. H&E staining of major organs confirms absence of toxicity.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
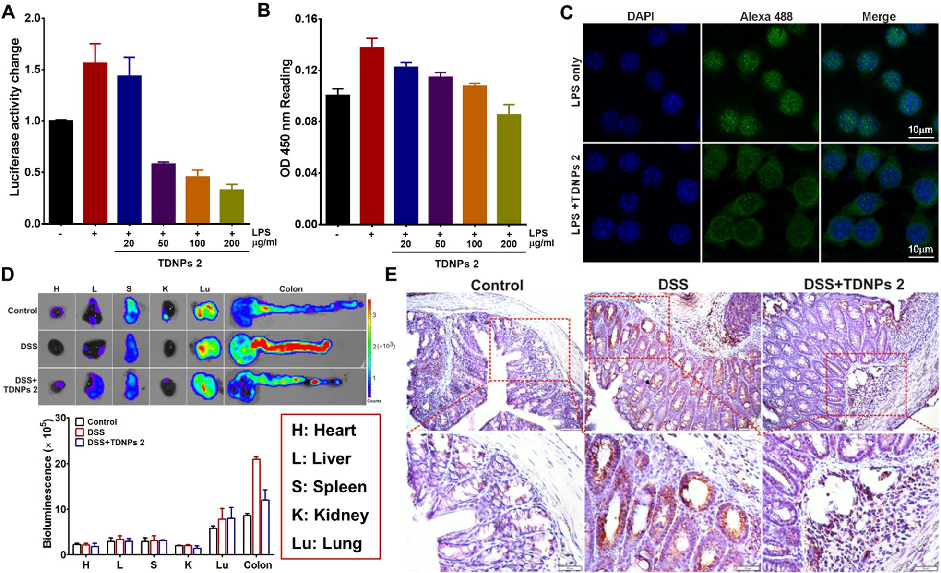
TDNPs 2 exert their protective effect at least partly through inactivation of the NF-kB signaling pathway. Reduced phospho-NF-kB p65 expression and decreased nuclear translocation indicate suppression of this key inflammatory cascade.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
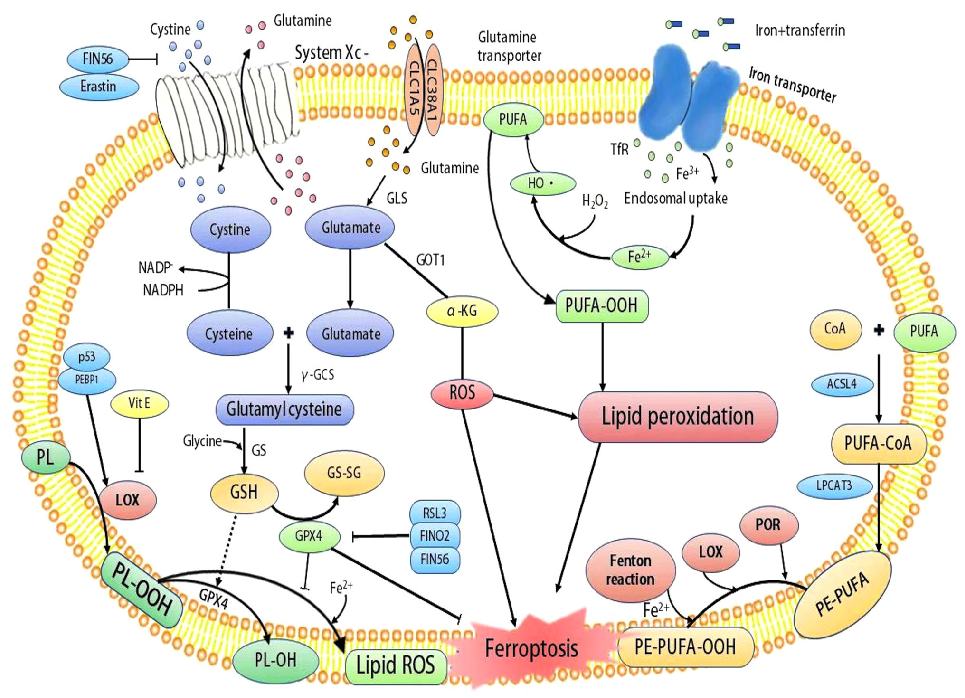
Mechanistic diagram illustrating the role of ferroptosis — characterized by iron accumulation, lipid peroxidation, and ROS production — in the pathogenesis of inflammatory bowel disease and its potential as a therapeutic target.
Role of ferroptosis in the pathogenesis and as a therapeutic target of …
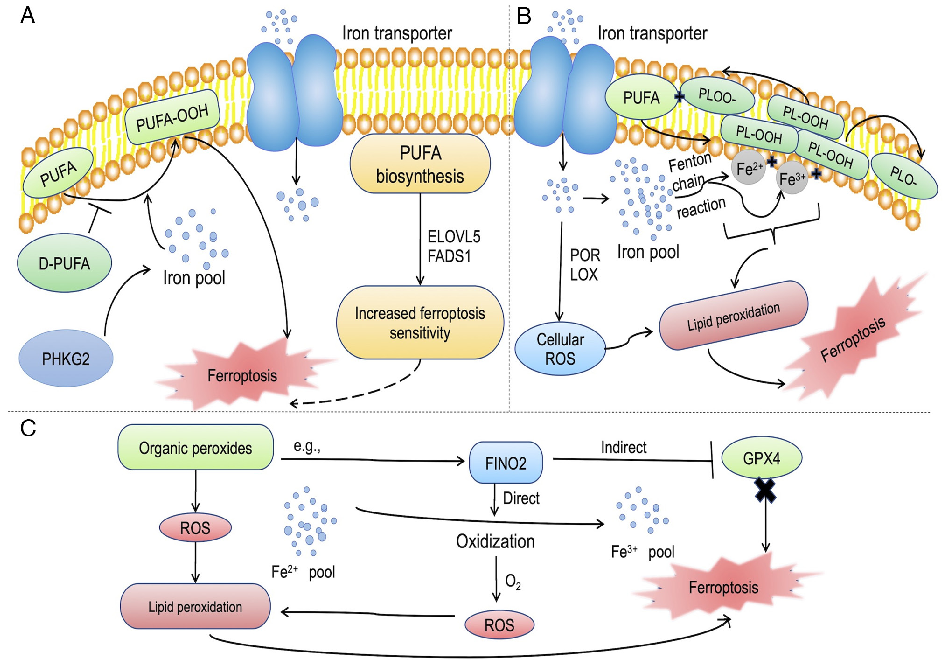
Mechanistic diagram illustrating the role of ferroptosis — characterized by iron accumulation, lipid peroxidation, and ROS production — in the pathogenesis of inflammatory bowel disease and its potential as a therapeutic target.
Role of ferroptosis in the pathogenesis and as a therapeutic target of …
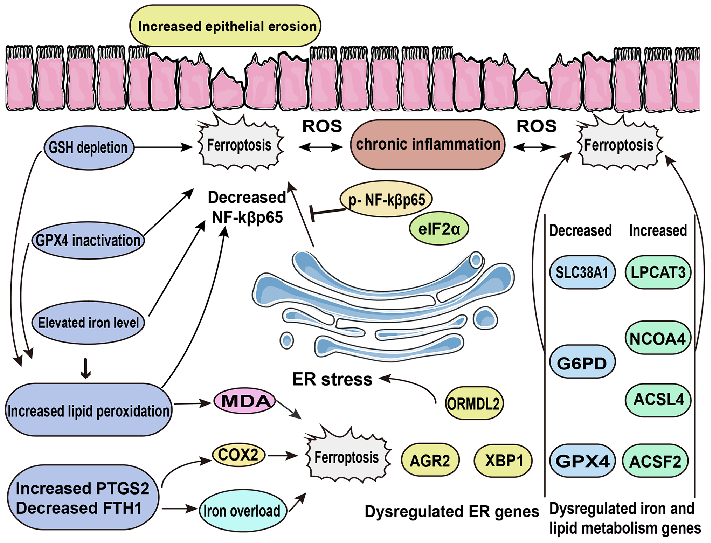
Mechanistic diagram illustrating the role of ferroptosis — characterized by iron accumulation, lipid peroxidation, and ROS production — in the pathogenesis of inflammatory bowel disease and its potential as a therapeutic target.
Role of ferroptosis in the pathogenesis and as a therapeutic target of …
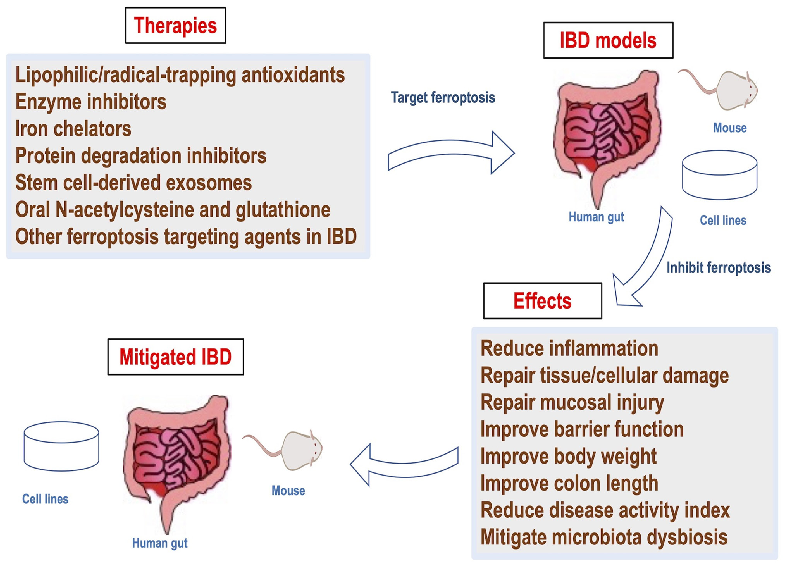
Mechanistic diagram illustrating the role of ferroptosis — characterized by iron accumulation, lipid peroxidation, and ROS production — in the pathogenesis of inflammatory bowel disease and its potential as a therapeutic target.
Role of ferroptosis in the pathogenesis and as a therapeutic target of …
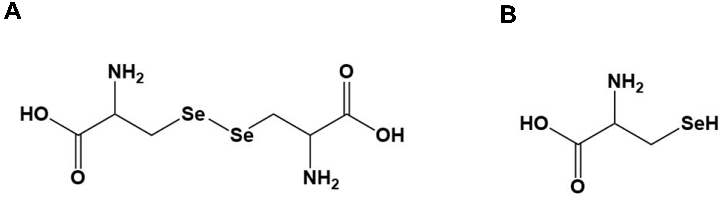
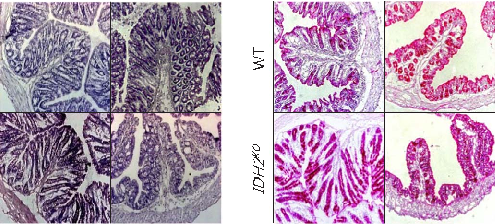
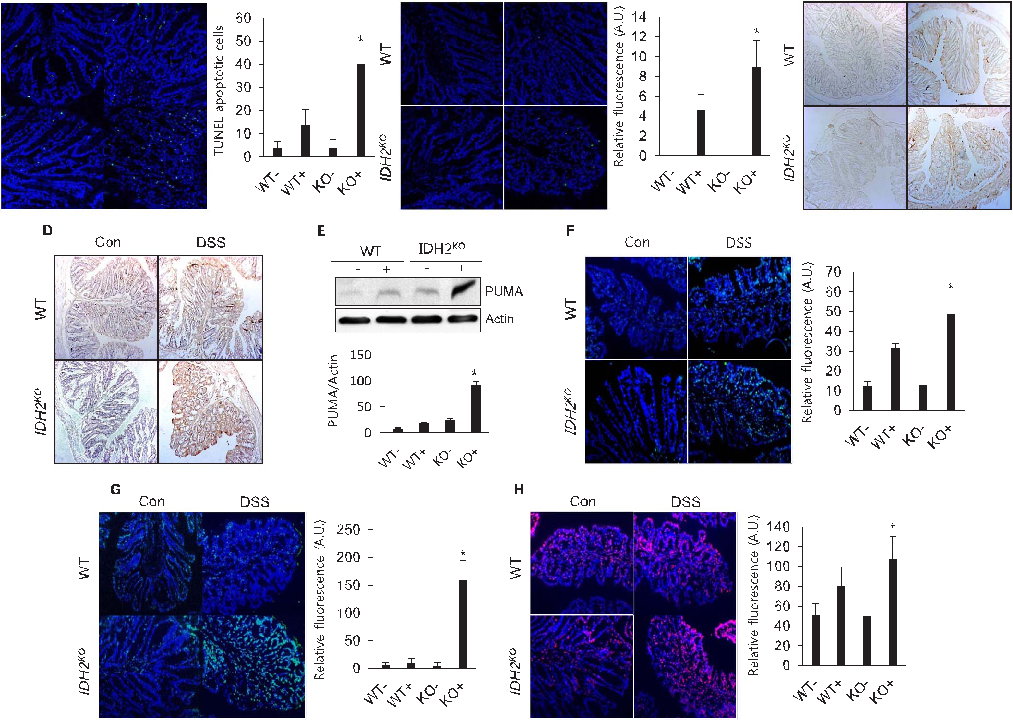
Selenium-Containing Amino Acids Protect Dextran Sulfate Sodium-Induced Colitis via Ameliorating Oxidative Stress …
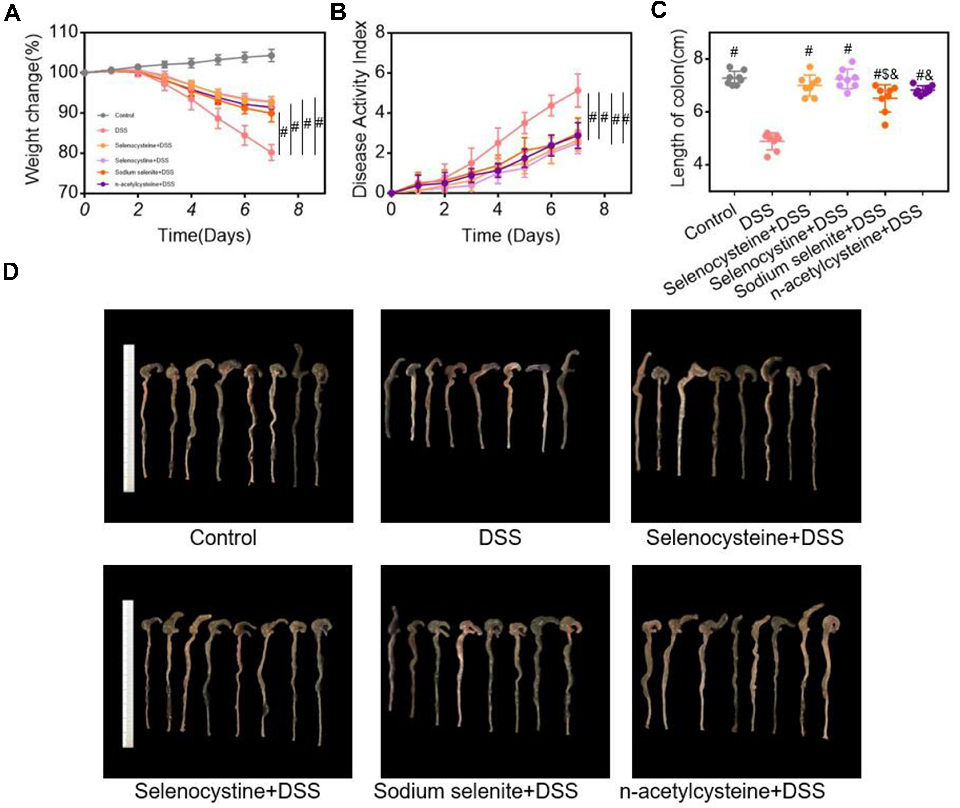
Selenium-Containing Amino Acids Protect Dextran Sulfate Sodium-Induced Colitis via Ameliorating Oxidative Stress …
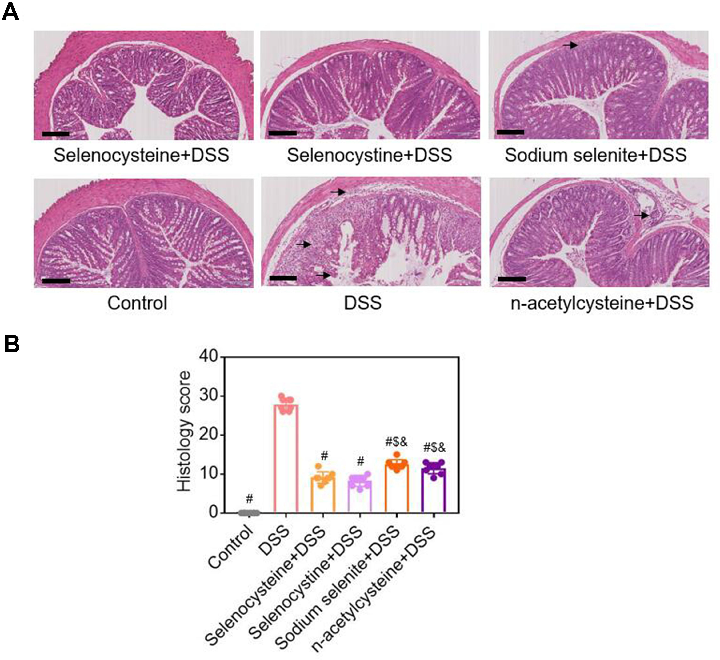
Figure 3 Histological examinations of effects of selenium-containing amino acids on DSS-induced IBD in mice. (A) Representative H&E-stained colon sections of each group (scale bar, 200 μm); (B) Histological scoring …
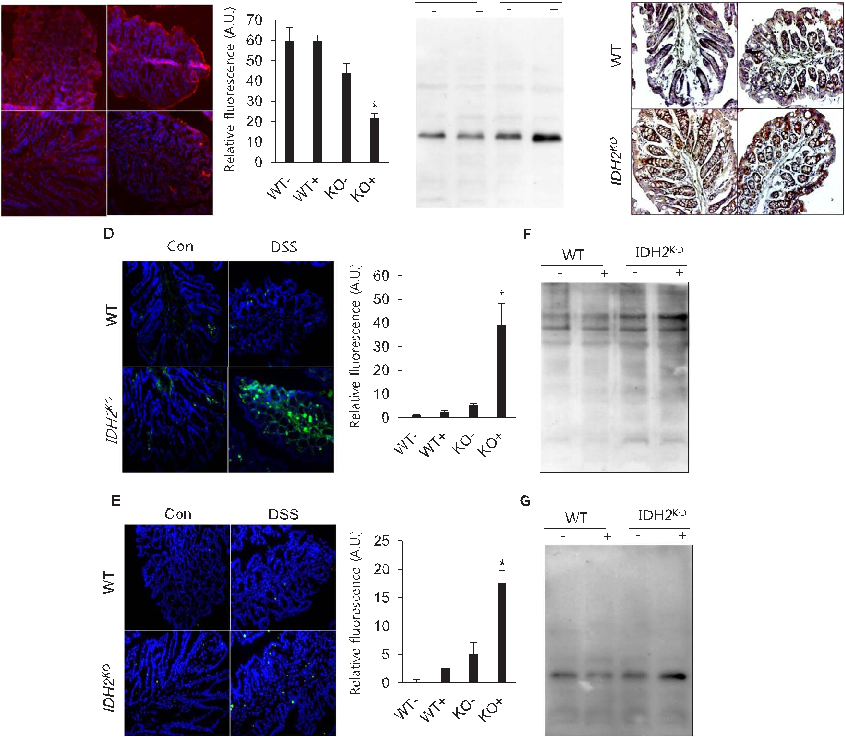
Selenium-Containing Amino Acids Protect Dextran Sulfate Sodium-Induced Colitis via Ameliorating Oxidative Stress …
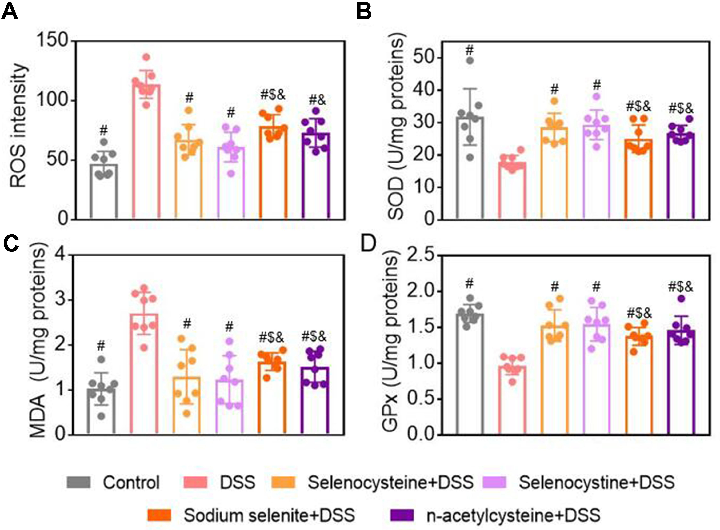
Selenium-Containing Amino Acids Protect Dextran Sulfate Sodium-Induced Colitis via Ameliorating Oxidative Stress …
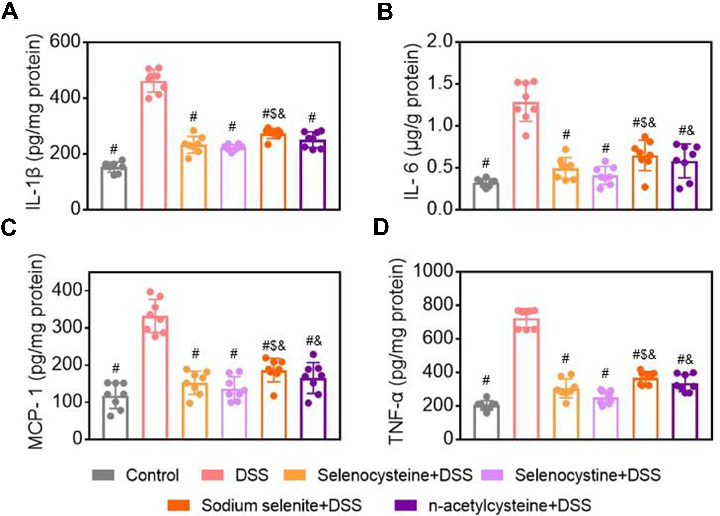
Selenium-Containing Amino Acids Protect Dextran Sulfate Sodium-Induced Colitis via Ameliorating Oxidative Stress …
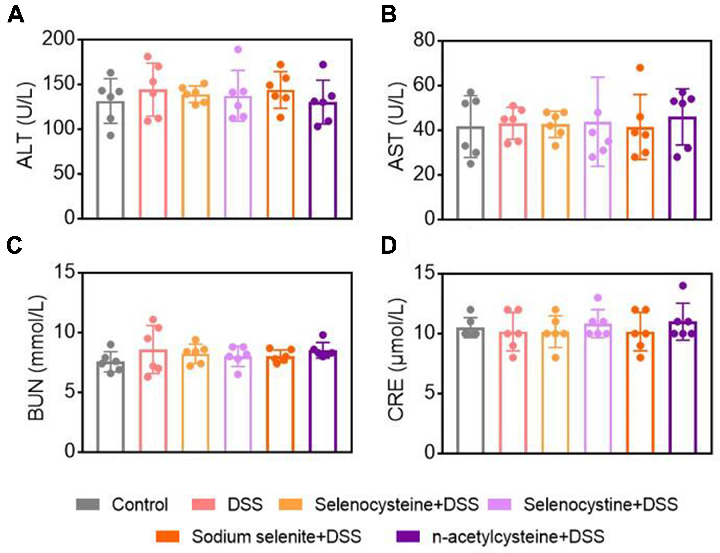
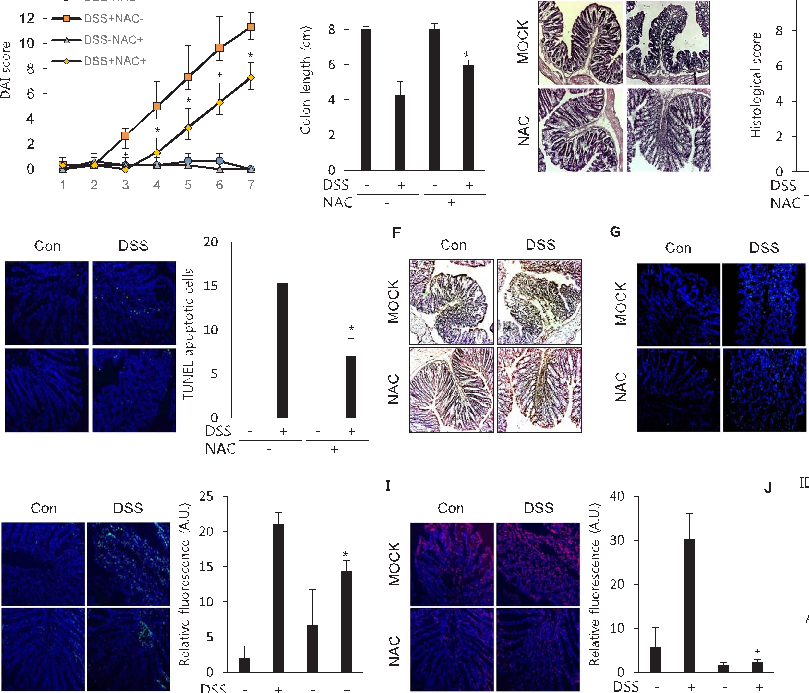
Figure 6 Effects of selenium-containing amino acids on biochemical test parameters in DSS-induced IBD in mice. Serum levels of (A) ALT, (B) AST, (C) BUN, and (D) CRE in different …
Selenium-Containing Amino Acids Protect Dextran Sulfate Sodium-Induced Colitis via Ameliorating Oxidative Stress …
Trang 1 / 2