Quy trình nghiên cứu
477 hình ảnh từ nghiên cứu có bình duyệt

Fecal calprotectin and other inflammatory biomarkers may serve as objective measures for monitoring probiotic treatment response in IBD patients. Reductions in these markers correlate with clinical improvement in several studies.
Role of prebiotics, probiotics, and synbiotics in management of inflammatory bowel disease: …
Mechanisms of probiotic action in poultry include competitive exclusion of pathogens, production of bacteriocins, enhancement of intestinal barrier integrity, and stimulation of mucosal immune responses.
Probiotics, Prebiotics, and Phytogenic Substances for Optimizing Gut Health in Poultry.
Prebiotic supplementation in poultry diets selectively promotes beneficial cecal microorganisms, particularly Bifidobacterium and Lactobacillus, leading to increased short-chain fatty acid production and improved gut barrier function.
Probiotics, Prebiotics, and Phytogenic Substances for Optimizing Gut Health in Poultry.
Essential oil compounds from oregano, thyme, and cinnamon demonstrate dose-dependent antimicrobial activity against common poultry pathogens. Their mode of action involves disrupting bacterial cell membrane integrity.
Probiotics, Prebiotics, and Phytogenic Substances for Optimizing Gut Health in Poultry.
Synbiotic combinations in poultry nutrition show additive or synergistic effects compared to individual probiotic or prebiotic supplementation. Performance metrics including feed conversion ratio and weight gain may improve with optimized formulations.
Probiotics, Prebiotics, and Phytogenic Substances for Optimizing Gut Health in Poultry.
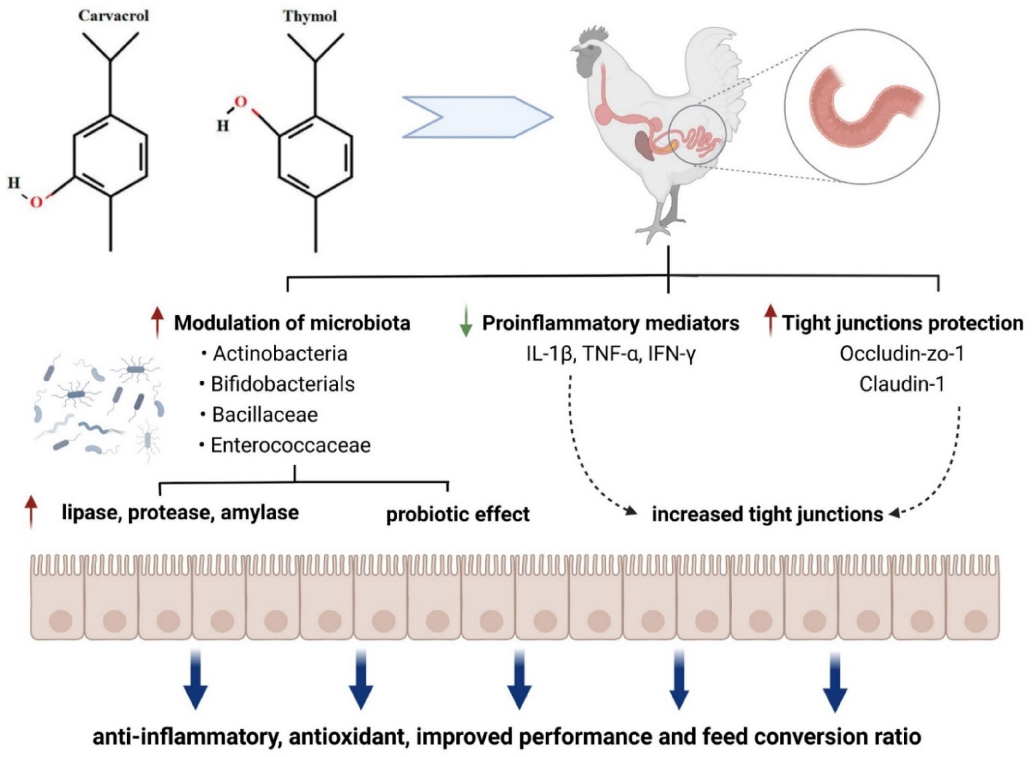
A comprehensive overview of gut health optimization strategies in poultry production compares the efficacy of probiotics, prebiotics, phytogenics, and synbiotics across key performance indicators and immune parameters.
Probiotics, Prebiotics, and Phytogenic Substances for Optimizing Gut Health in Poultry.
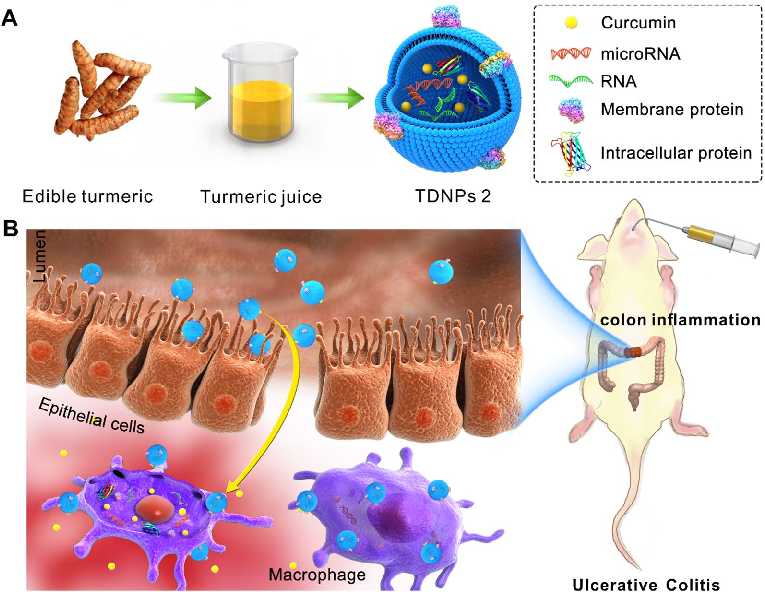
Turmeric-derived nanoparticles (TDNPs 2) are isolated through sucrose gradient ultracentrifugation and administered orally to target inflamed colonic tissue in a murine colitis model. The schematic outlines the isolation workflow from edible turmeric to purified nanovesicles.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
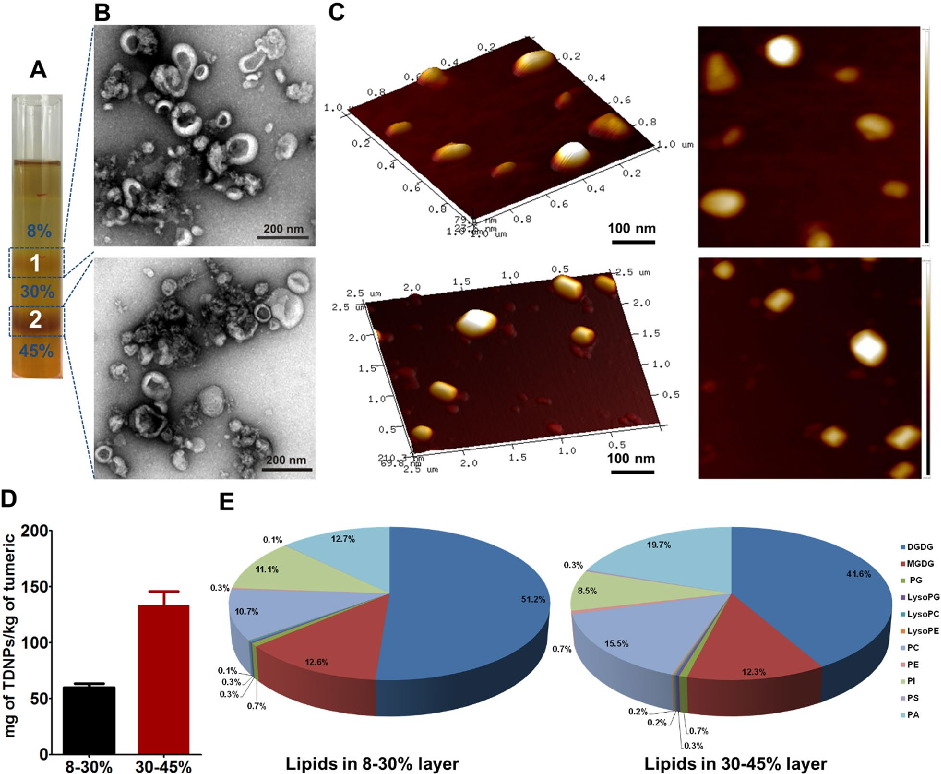
Characterization of turmeric-derived nanoparticles reveals two distinct bands (TDNPs 1 and TDNPs 2) at the 8%/30% and 30%/45% sucrose gradient interfaces, respectively. TDNPs 2 demonstrate appropriate size distribution and surface charge for oral drug delivery applications.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
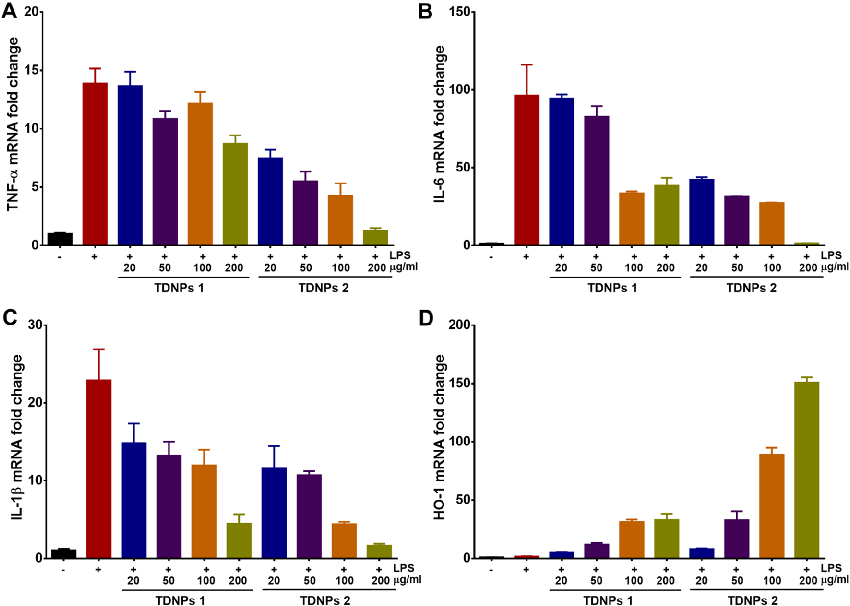
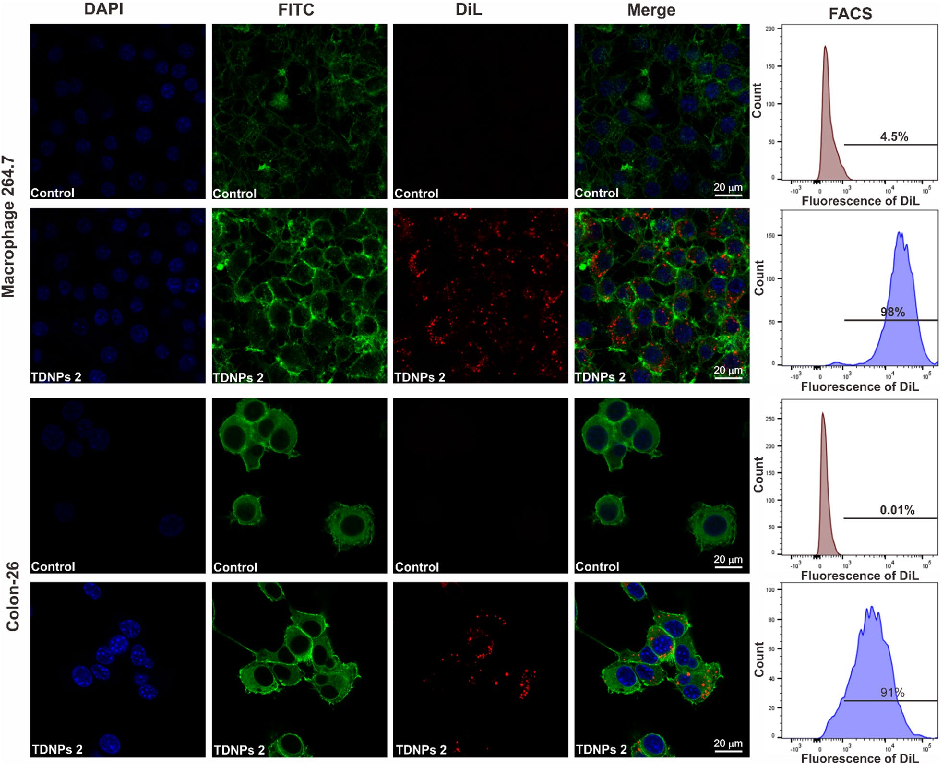
In vitro assessment of turmeric-derived nanovesicles demonstrates anti-inflammatory activity, including suppression of pro-inflammatory cytokine production in activated macrophages. Dose-dependent reductions in TNF-alpha and IL-6 secretion are observed.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
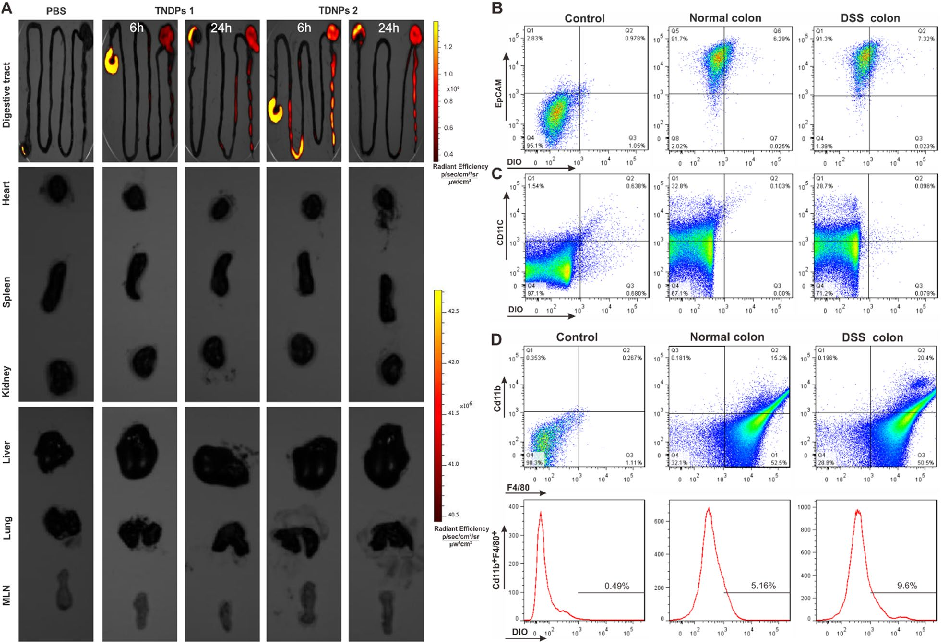
Biodistribution imaging using IVIS Spectrum reveals that TDNPs 2 preferentially accumulate in inflamed colonic tissue following oral administration. Fluorescence signals are minimal in non-target organs including heart, liver, spleen, lung, and kidney.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
Oral TDNPs 2 administration significantly attenuates disease activity index scores and colon shortening in DSS-induced colitis mice. Body weight recovery is also improved compared to untreated colitis controls.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
Intestinal permeability assessment indicates that TDNPs 2 treatment preserves gut barrier integrity in colitis mice. Tight junction protein expression, including ZO-1 and occludin, is maintained at near-normal levels.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
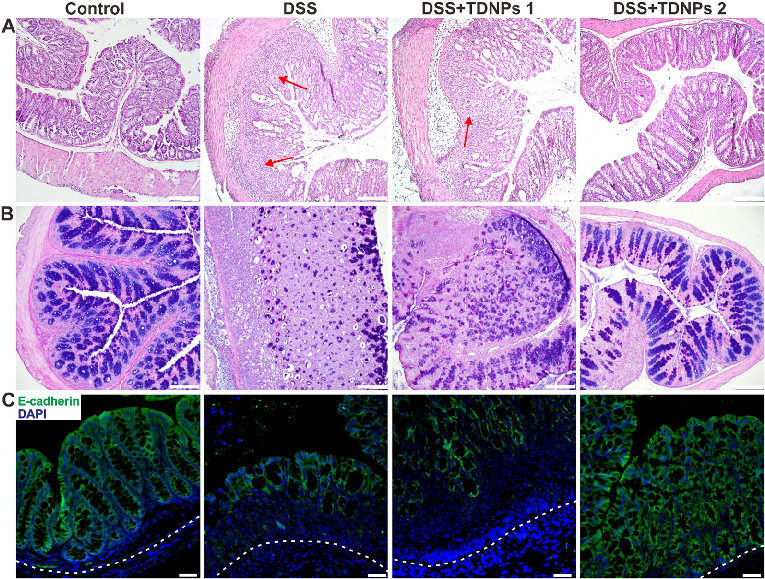
Histological examination with H&E staining reveals markedly reduced inflammatory cell infiltration and preserved goblet cell density in TDNPs 2-treated colitic mice. Colonic tissue architecture remains largely intact compared to severe disruption in untreated animals.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
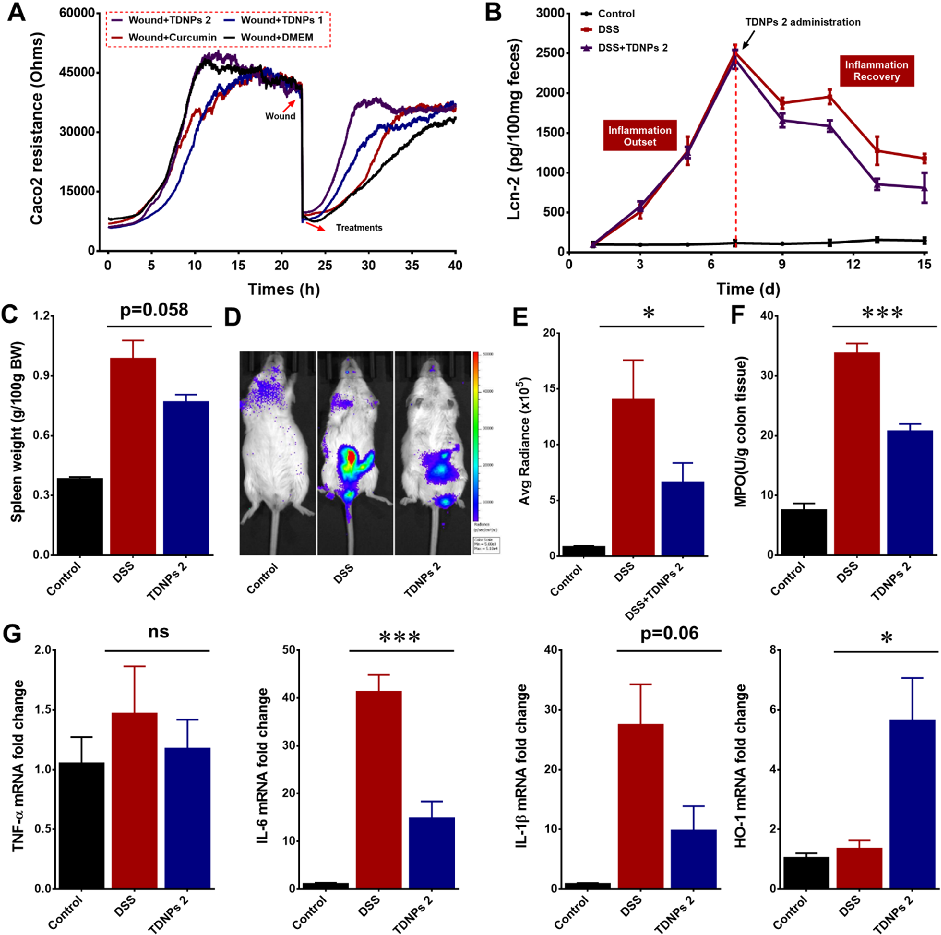
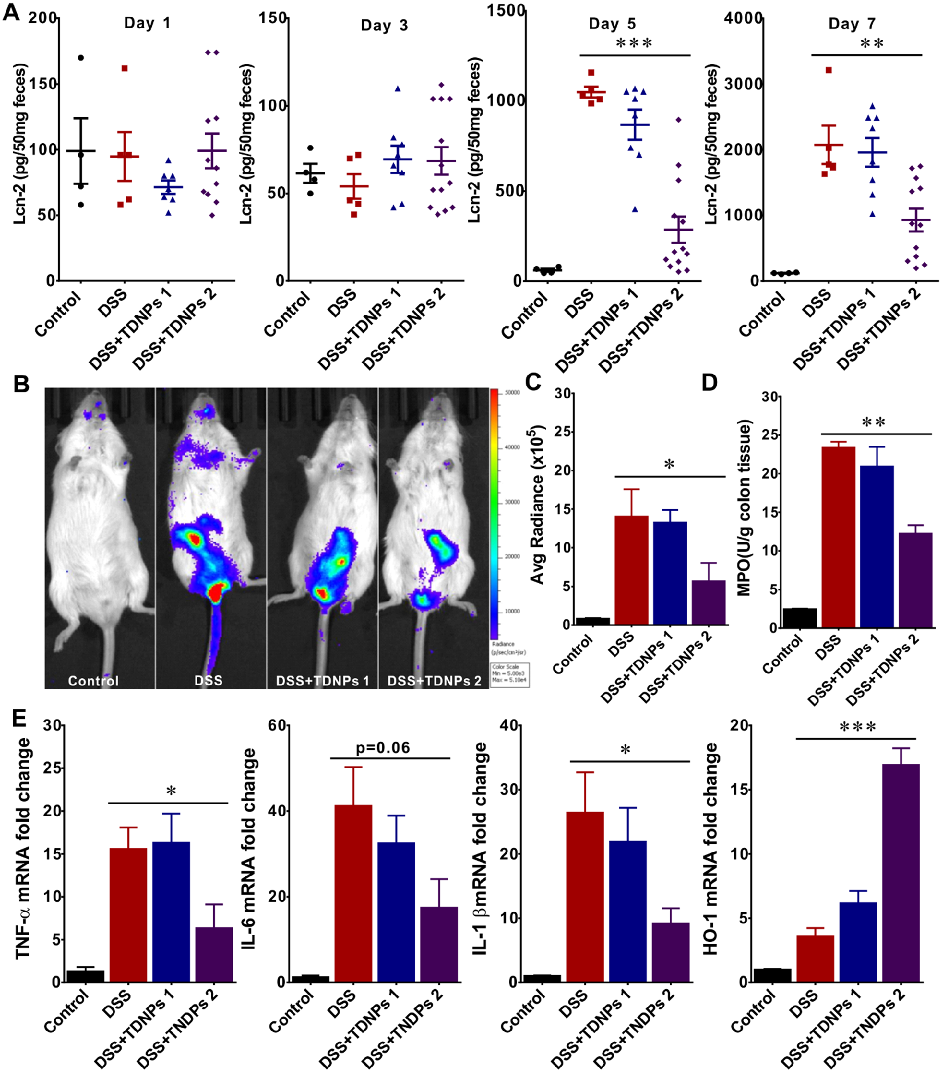
Wound healing assays and fecal lipocalin-2 quantification demonstrate that TDNPs 2 accelerate resolution of intestinal inflammation. Lcn-2 levels, a sensitive marker of intestinal inflammation, decrease significantly with treatment.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
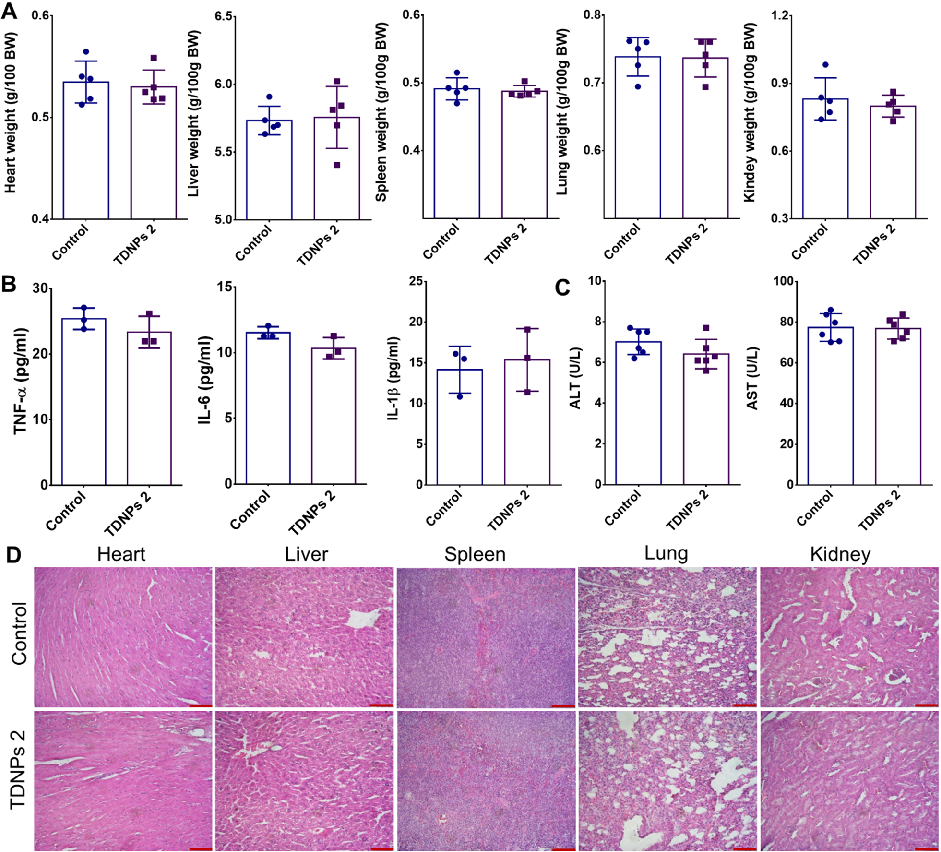
Biocompatibility evaluation shows no significant changes in vital organ weights, pro-inflammatory cytokines, or liver function indicators in TDNPs 2-treated mice. H&E staining of major organs confirms absence of toxicity.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
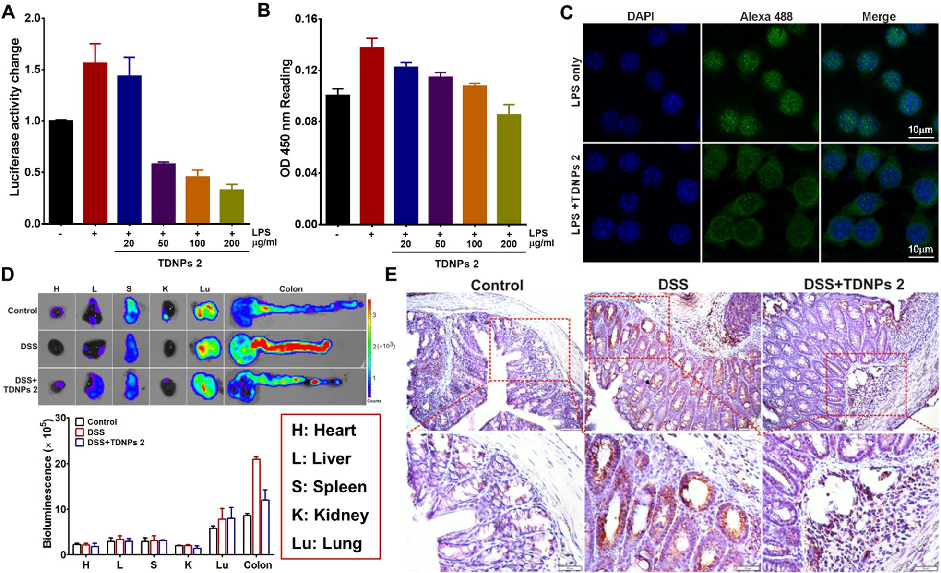
TDNPs 2 exert their protective effect at least partly through inactivation of the NF-kB signaling pathway. Reduced phospho-NF-kB p65 expression and decreased nuclear translocation indicate suppression of this key inflammatory cascade.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
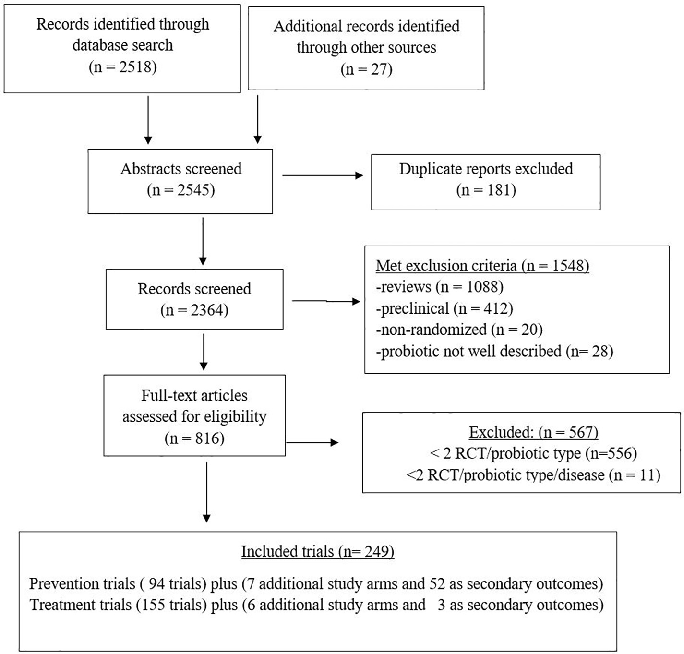
A PRISMA flow diagram details the systematic literature search and study selection process for evaluating probiotic efficacy across multiple clinical conditions. Studies are screened from inception through defined databases with specific inclusion and exclusion criteria.
Choosing an appropriate probiotic product for your patient: An evidence-based practical guide.
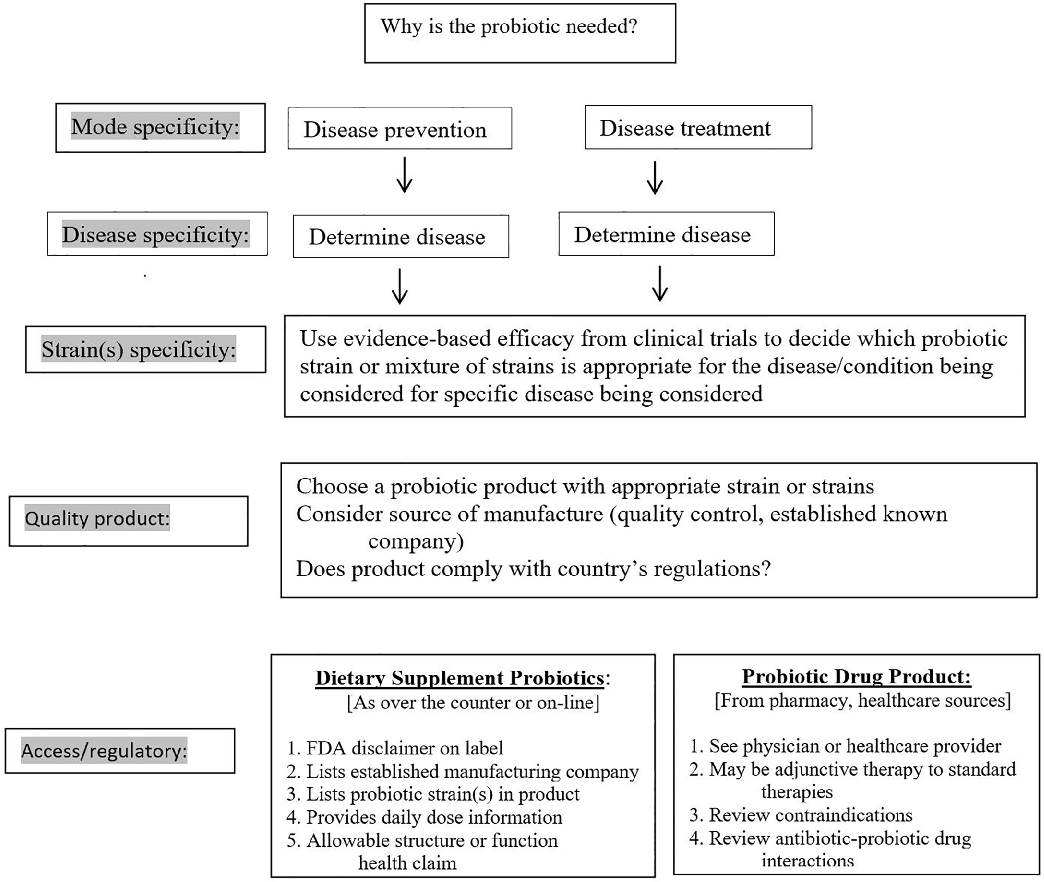
A clinical decision algorithm guides clinicians through the process of selecting an appropriate probiotic product based on the patient's specific condition, available evidence for particular strains, and practical considerations such as product availability and cost.
Choosing an appropriate probiotic product for your patient: An evidence-based practical guide.
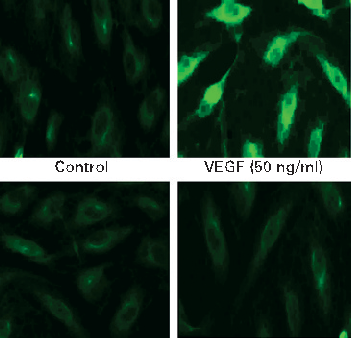
COX-2 protein expression is upregulated by VEGF stimulation in intestinal endothelial cells, and curcumin effectively blocks this induction. Prostaglandin E2 production follows a similar pattern of inhibition.
Curcumin inhibits VEGF-mediated angiogenesis in human intestinal microvascular endothelial cells through COX-2 …

MAPK signaling pathway activation by VEGF is attenuated by curcumin in a time- and dose-dependent manner. Phosphorylation of ERK, p38, and JNK is markedly reduced in curcumin-treated endothelial cells.
Curcumin inhibits VEGF-mediated angiogenesis in human intestinal microvascular endothelial cells through COX-2 …
Animal models of colitis demonstrate that vitamin D deficiency exacerbates intestinal inflammation, while supplementation or VDR activation attenuates disease severity. These findings support a mechanistic link between vitamin D status and IBD pathophysiology.
The Role of Vitamin D in Inflammatory Bowel Disease: Mechanism to Management.
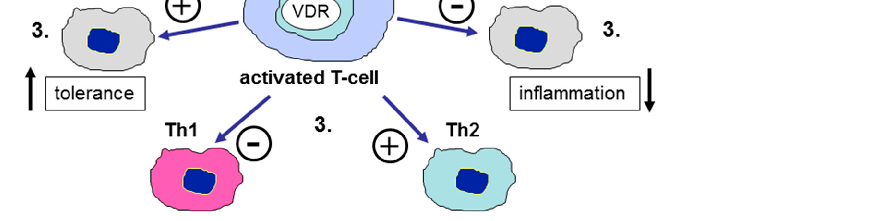
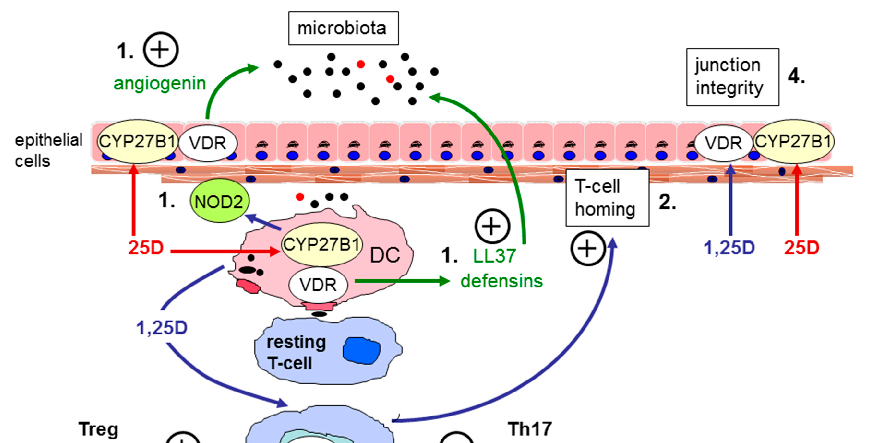
Vitamin D receptor expression and vitamin D-activating enzyme CYP27B1 are present in human colonic epithelial cells, enabling local production of active calcitriol. This paracrine signaling enhances tight junction integrity and antimicrobial defense at the mucosal surface.
The Role of Vitamin D in Inflammatory Bowel Disease: Mechanism to Management.
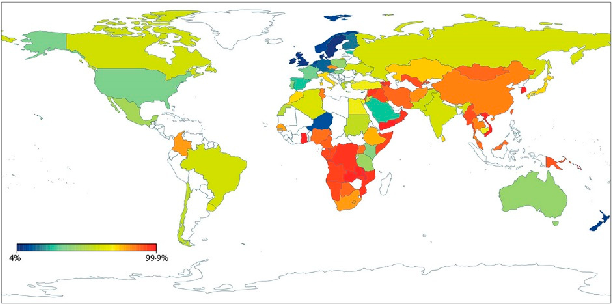
Geographic distribution of lactase non-persistence frequencies reveals wide variation across world populations, with highest persistence rates in Northern European and certain African and Middle Eastern pastoralist populations. East Asian and Native American populations show near-universal non-persistence.
Lactose Intolerance, Dairy Avoidance, and Treatment Options.
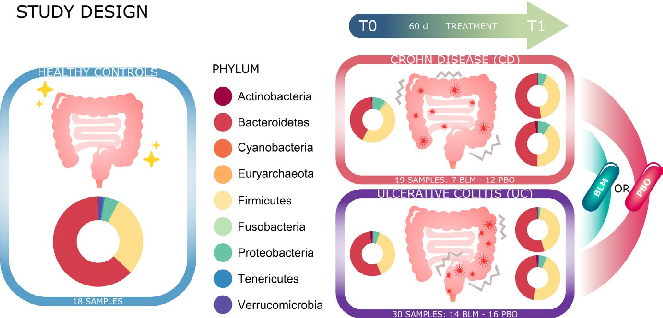
Gut microbiota alpha diversity metrics, including Shannon index and observed species richness, are compared before and after butyrate supplementation. Changes in diversity differ between Crohn's disease and ulcerative colitis subgroups.
Microbiota changes induced by microencapsulated sodium butyrate in patients with inflammatory bowel …
Trang 2 / 20