Butyrate (Sodium/Calcium Butyrate) Figuras
89 figuras de pesquisa revisada por pares
Differential abundance analysis identifying specific bacterial taxa that are significantly enriched or depleted in the intestinal microbiome of treatment-naive pediatric Crohn's disease patients.
Differences in the intestinal microbiome of healthy children and patients with newly …
Correlation analysis or network diagram showing relationships between specific gut bacterial taxa and clinical parameters in pediatric Crohn's disease at the time of diagnosis.
Differences in the intestinal microbiome of healthy children and patients with newly …

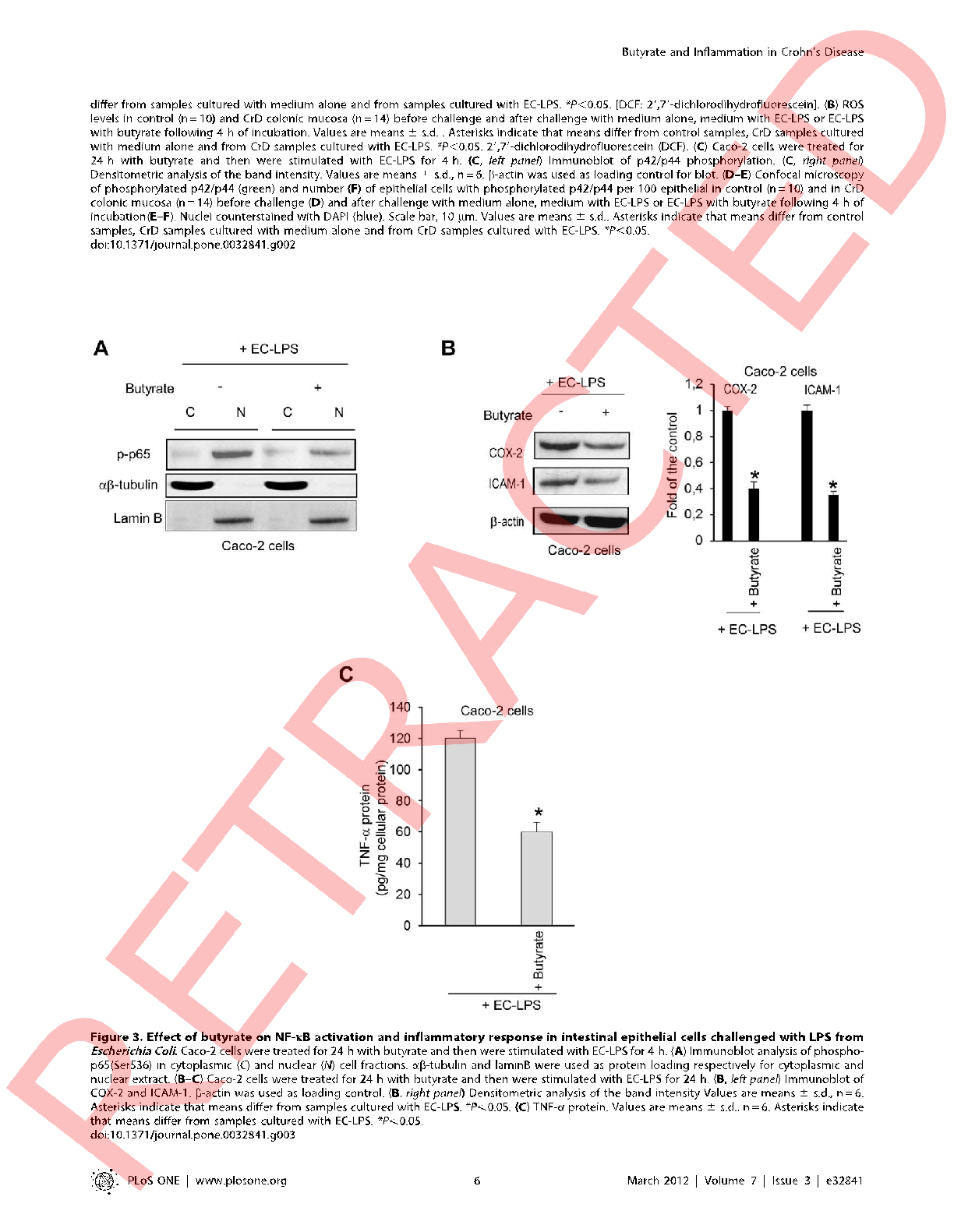
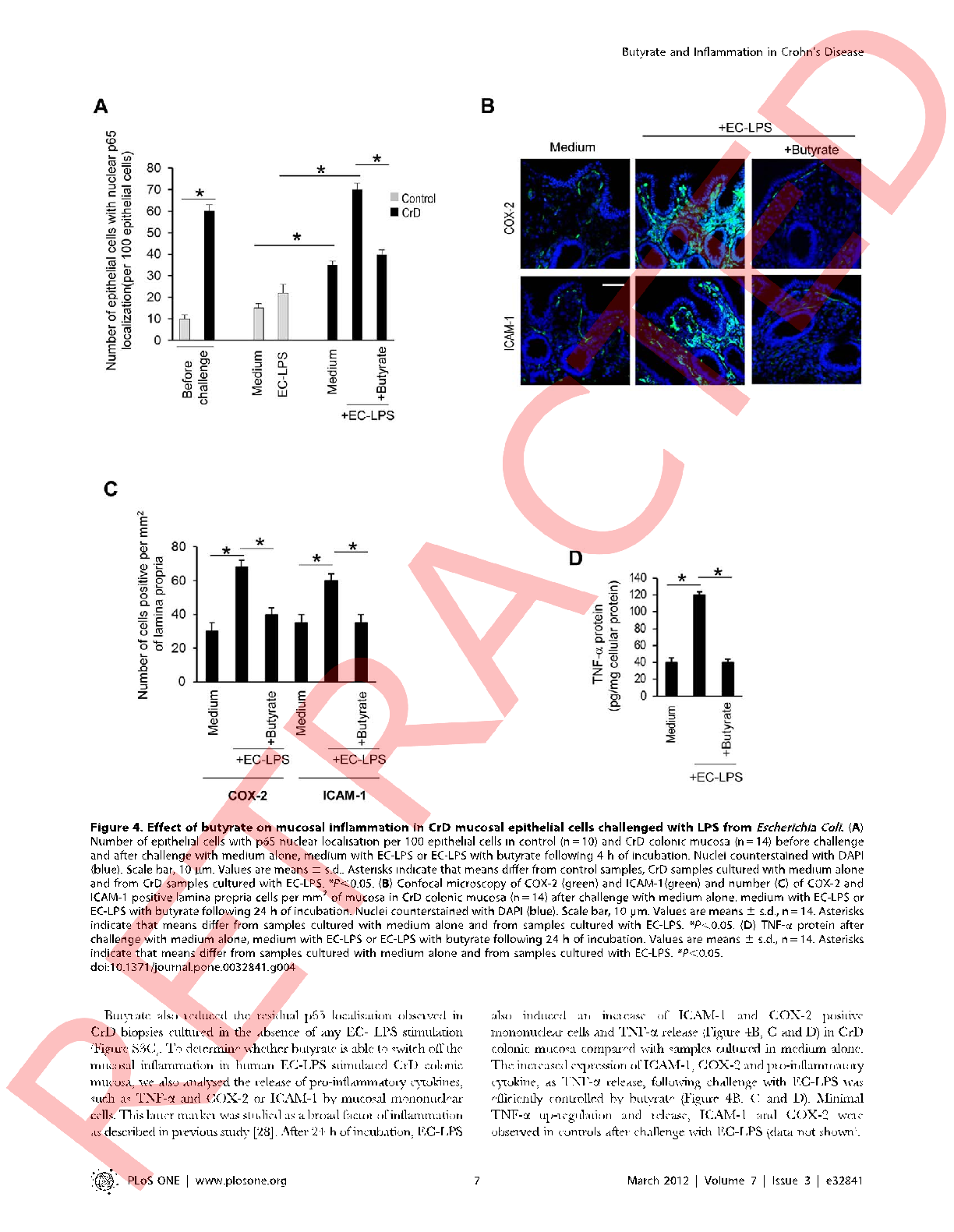
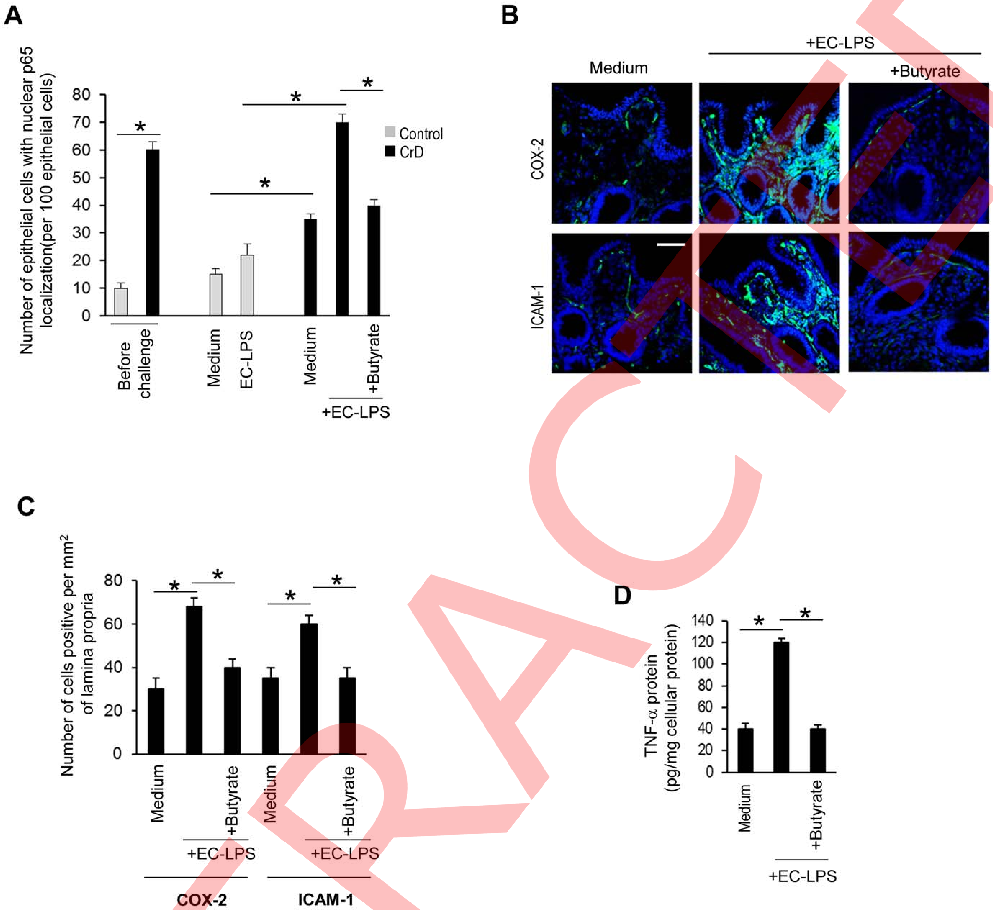
NF-κB signaling pathway activation in intestinal cells is measured with and without butyrate pre-treatment following LPS challenge, demonstrating the short-chain fatty acid's inhibitory capacity.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …

Gene expression profiles for inflammatory mediators in LPS-stimulated intestinal cells are compared across varying butyrate concentrations, revealing a clear anti-inflammatory dose response.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …

Cell viability assays confirm that butyrate concentrations used in the inflammation experiments are non-cytotoxic, validating the specificity of observed anti-inflammatory effects.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
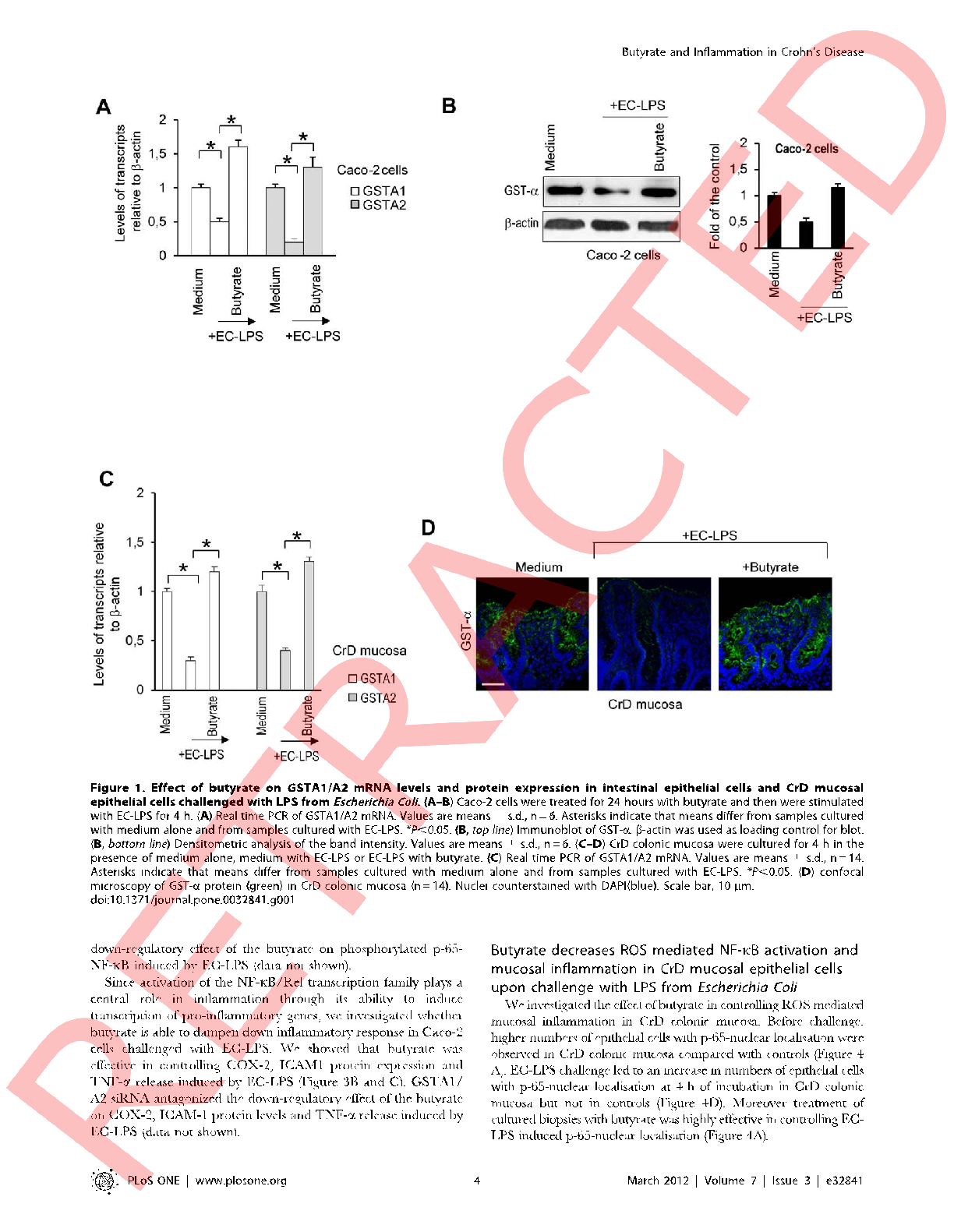
Transepithelial electrical resistance measurements across intestinal cell monolayers demonstrate butyrate's capacity to maintain epithelial integrity during inflammatory challenge.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
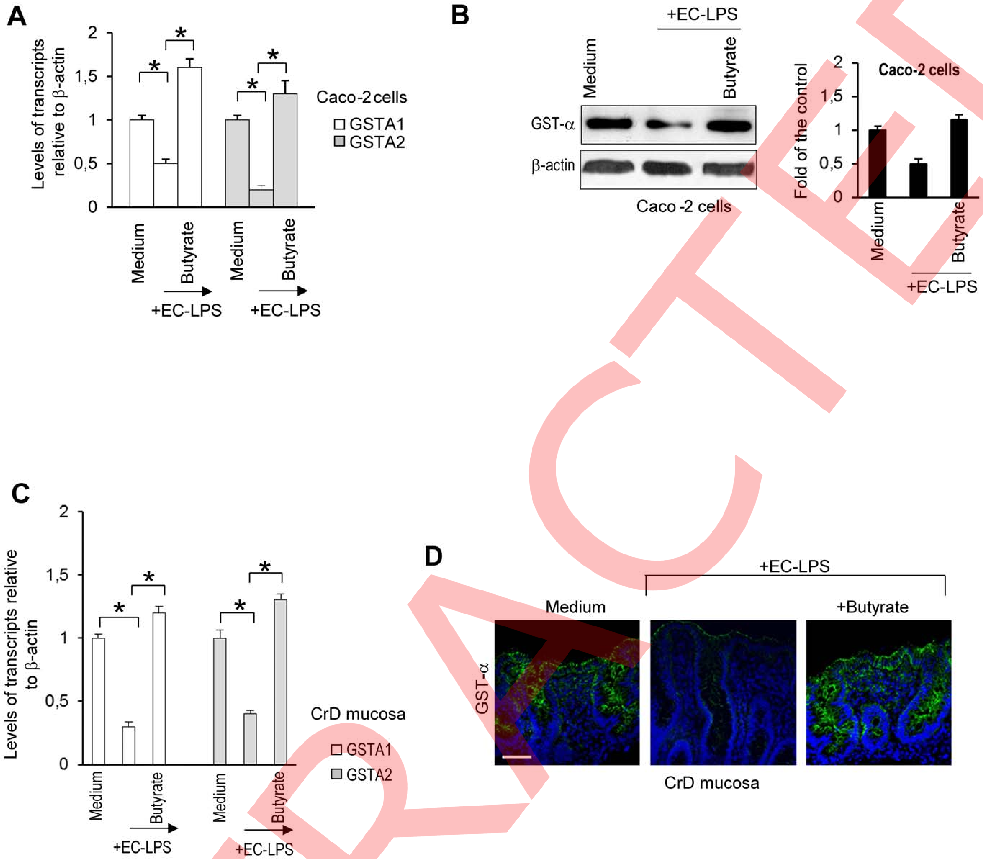
Western blot analysis of phosphorylated signaling proteins in LPS-treated intestinal cells shows reduced inflammatory cascade activation in the presence of butyrate.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
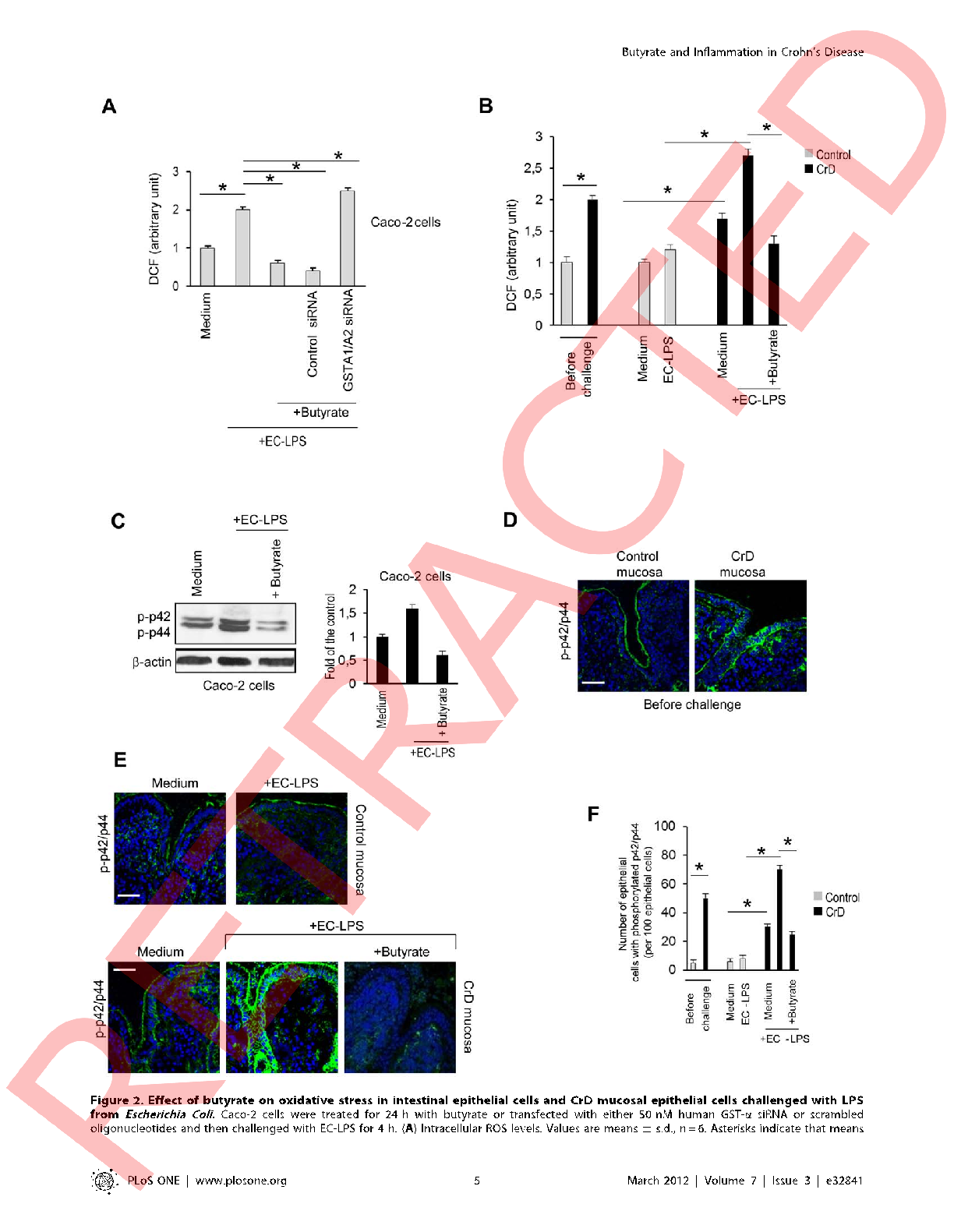
Cytokine secretion profiles including IL-6, IL-8, and TNF-α are quantified in cell culture supernatants, with butyrate-treated cells showing significantly attenuated inflammatory responses.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
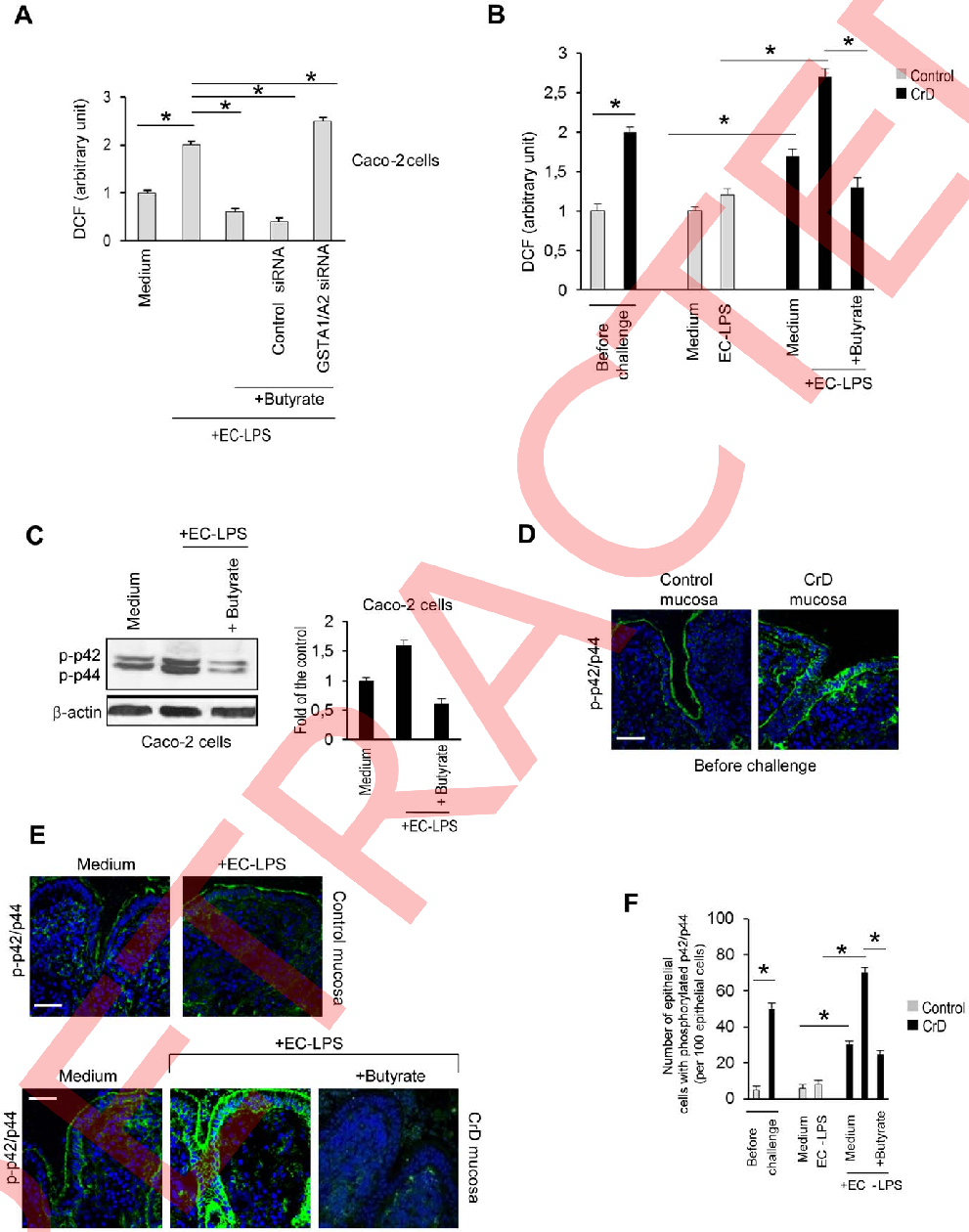
Histone deacetylase activity in intestinal epithelial cells is measured following butyrate exposure, linking epigenetic modulation to the observed anti-inflammatory phenotype.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
Intracellular reactive oxygen species levels are visualized in LPS-challenged intestinal cells with and without butyrate treatment, showing reduced oxidative stress in supplemented cultures.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
Prostaglandin E2 production by intestinal cells under inflammatory conditions is attenuated by butyrate, as measured by ELISA across multiple treatment concentrations.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
Apoptosis rates in intestinal epithelial cells exposed to LPS are compared between butyrate-treated and control conditions, indicating a cytoprotective effect of the short-chain fatty acid.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …

mRNA expression of tight junction components claudin-1 and occludin is upregulated in butyrate-treated intestinal cells, supporting enhanced paracellular barrier function.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …

Permeability assays using fluorescent dextran tracers demonstrate reduced paracellular flux in butyrate-pretreated intestinal monolayers challenged with LPS.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …

Butyrate's anti-inflammatory effects on intestinal epithelial cells stimulated with lipopolysaccharide are quantified, showing dose-dependent suppression of pro-inflammatory cytokine release.
Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and Crohn's mucosa through modulation …
Specific bacterial taxa responsive to probiotic treatment are identified, showing differential abundance patterns that suggest strain-specific effects on gut ecosystem structure.
The double-edged sword of probiotic supplementation on gut microbiota structure in Helicobacter …
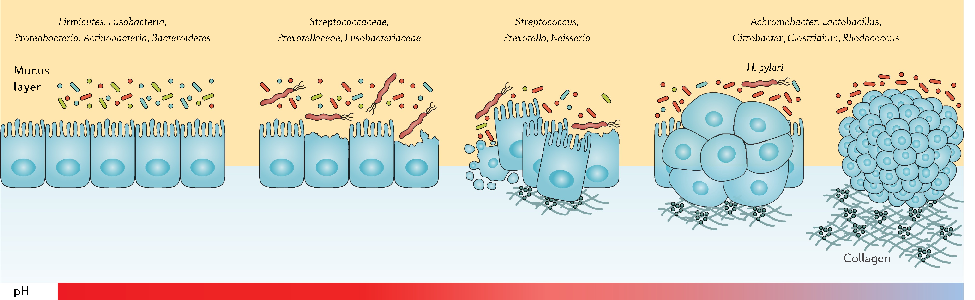
The dual nature of probiotic effects on established gut communities is depicted, with some participants showing enhanced diversity while others experience transient dysbiosis.
The double-edged sword of probiotic supplementation on gut microbiota structure in Helicobacter …
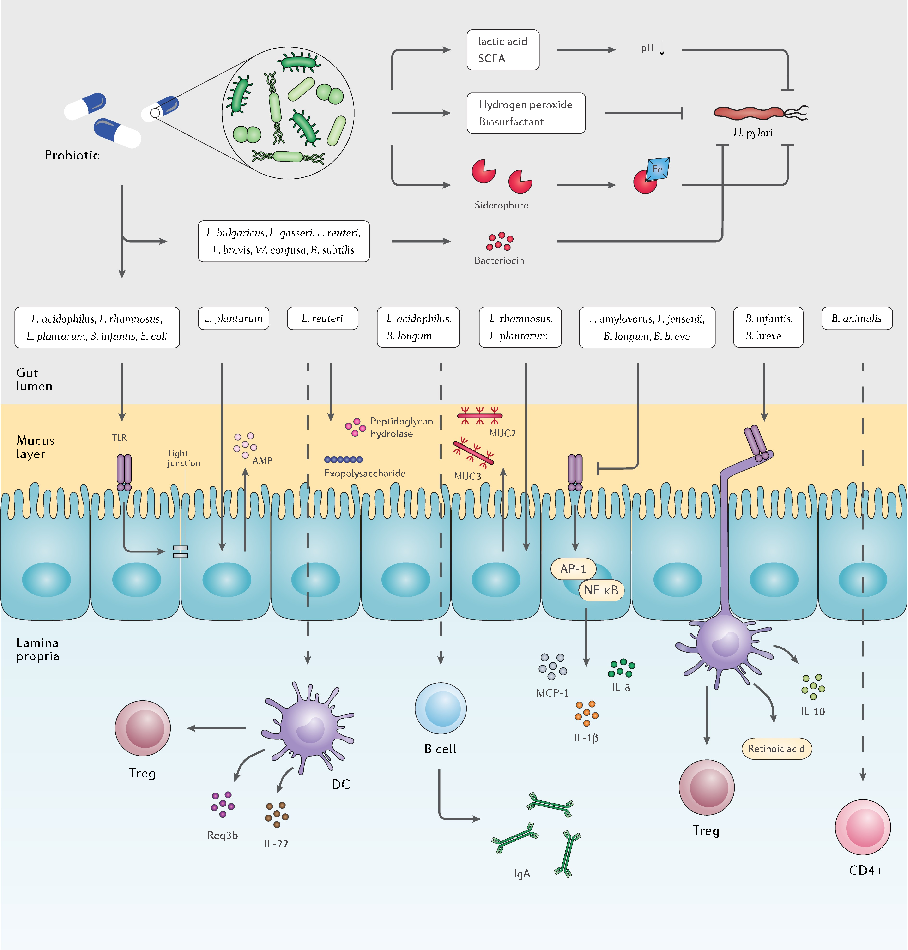
Gut microbiota structural changes following probiotic supplementation are characterized, revealing both beneficial shifts and potential disruptions depending on the host's baseline microbial composition.
The double-edged sword of probiotic supplementation on gut microbiota structure in Helicobacter …
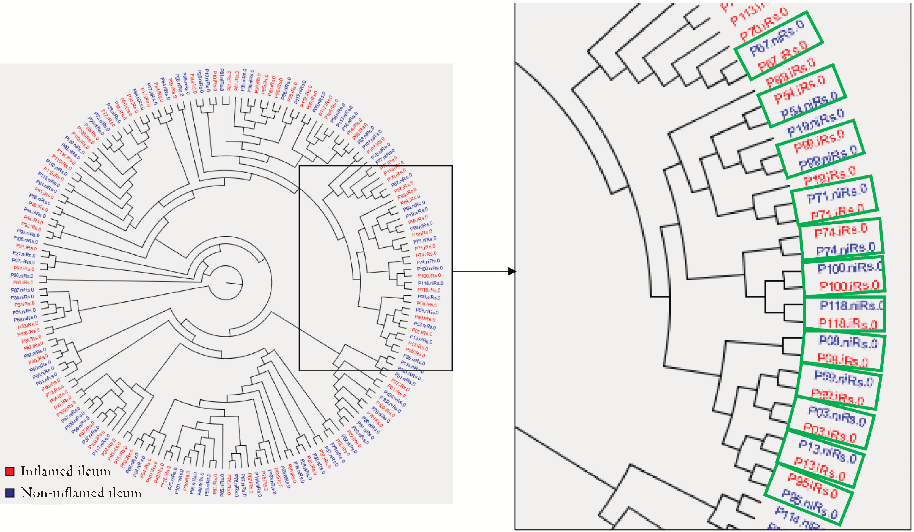
Predictive microbiota signatures for postoperative Crohn's disease recurrence are identified, with specific taxa associated with early endoscopic inflammation at the neoterminal ileum.
Early Postoperative Endoscopic Recurrence in Crohn's Disease Is Characterised by Distinct Microbiota …
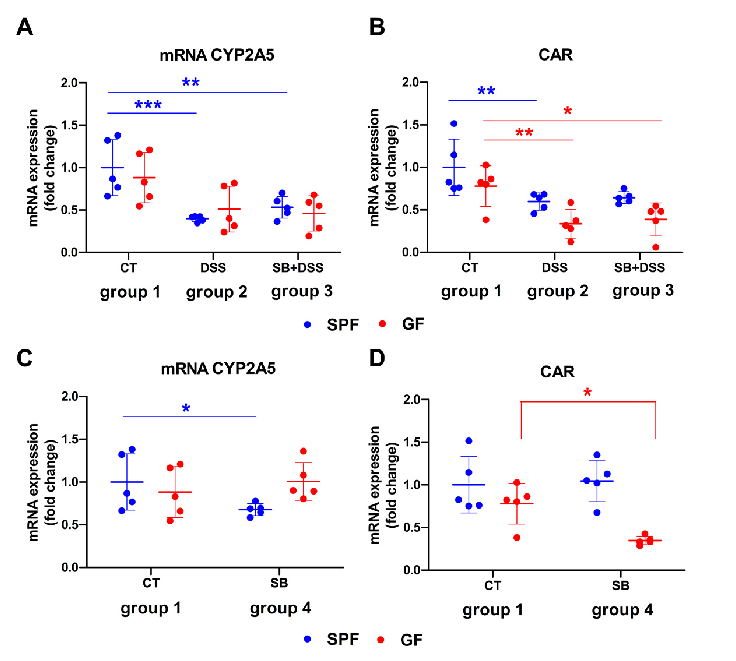
Comparison of mRNA expression of CYP2A5 and CAR in murine liver of GF and SPF mice. Gene expression of (A,C) CYP2A5 and (B,D) constitutive androstane receptor (CAR).
Effect of DSS-Induced Ulcerative Colitis and Butyrate on the Cytochrome P450 2A5: …
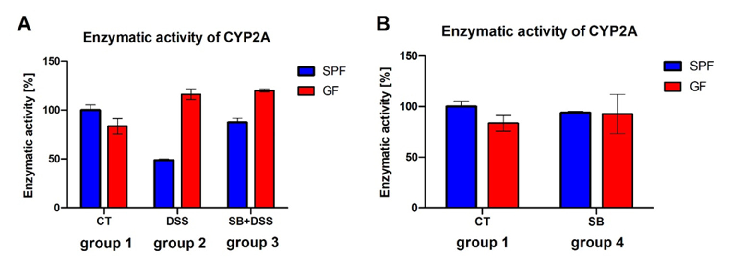
Statistical analysis from research investigating effect of DSS, comparing treatment groups and control conditions.
Effect of DSS-Induced Ulcerative Colitis and Butyrate on the Cytochrome P450 2A5: …
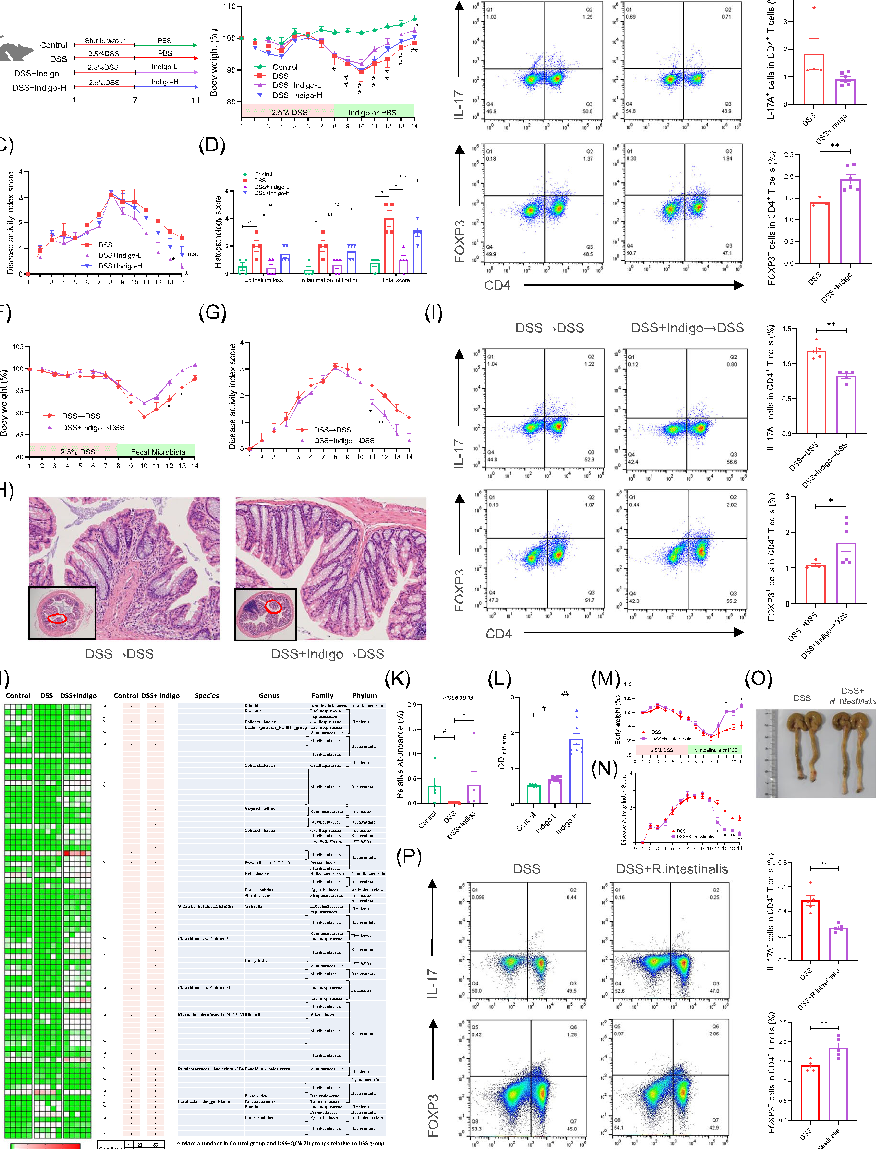
Experimental results examining gut microbiota, with data points illustrating key findings related to this study explored the effect of plant-derived indigo supplementation on intestinal inflammation using in vivo, in v.
Gut microbiota-derived butyrate mediates the anticolitic effect of indigo supplementation through regulating …
Página 2 de 4