Curcumin Figures
37 figures issues de recherches évaluées par des pairs
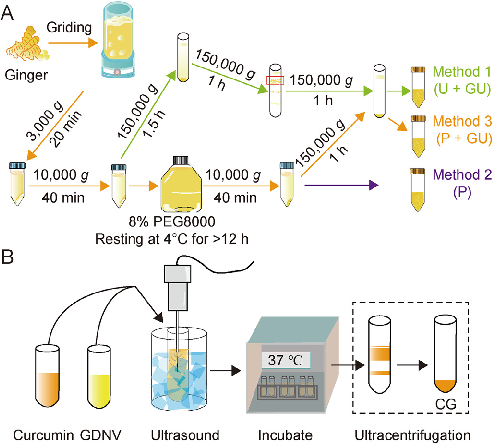
Turmeric-derived nanoparticles (TDNPs 2) are isolated through sucrose gradient ultracentrifugation and administered orally to target inflamed colonic tissue in a murine colitis model. The schematic outlines the isolation workflow from edible turmeric to purified nanovesicles.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
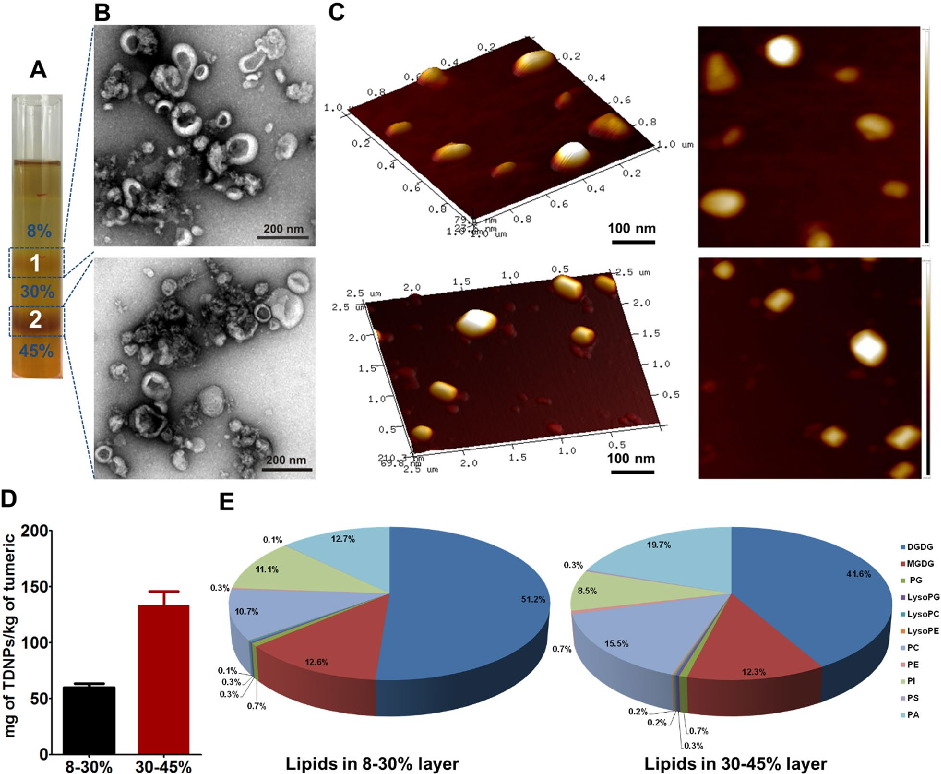
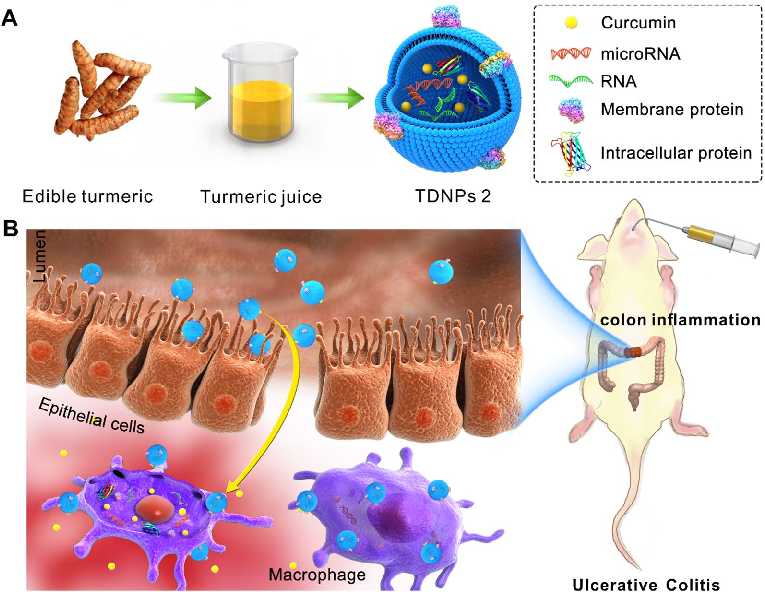
Characterization of turmeric-derived nanoparticles reveals two distinct bands (TDNPs 1 and TDNPs 2) at the 8%/30% and 30%/45% sucrose gradient interfaces, respectively. TDNPs 2 demonstrate appropriate size distribution and surface charge for oral drug delivery applications.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
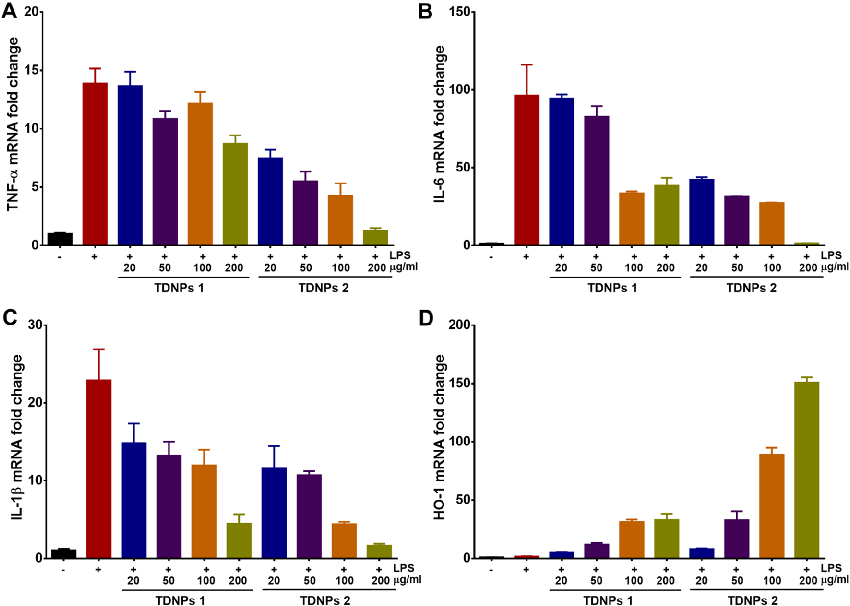
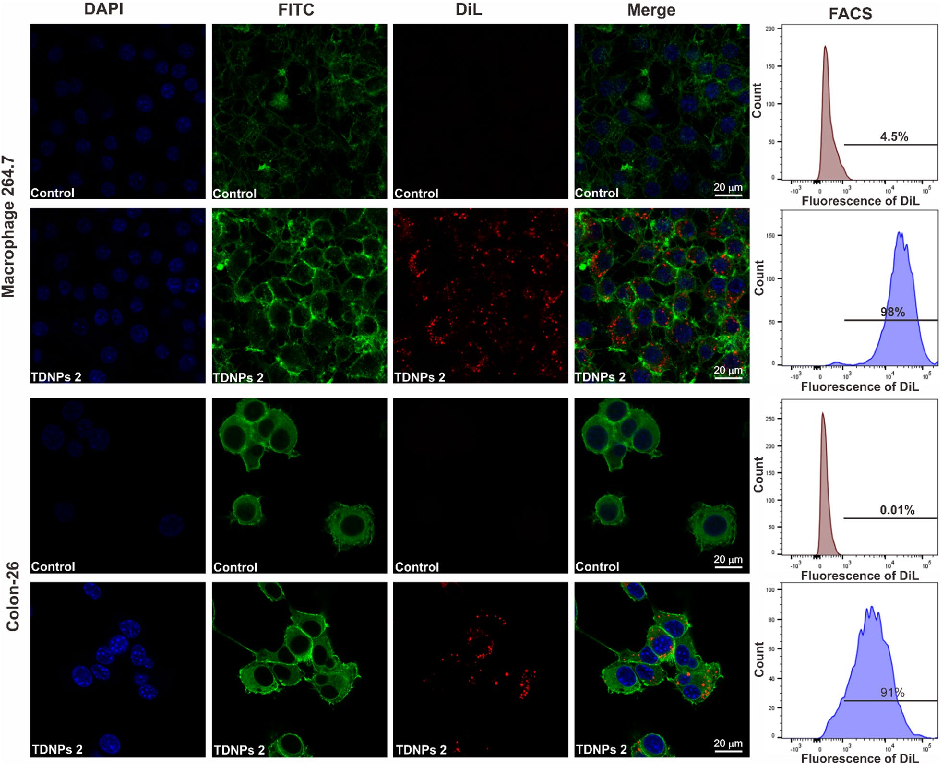
In vitro assessment of turmeric-derived nanovesicles demonstrates anti-inflammatory activity, including suppression of pro-inflammatory cytokine production in activated macrophages. Dose-dependent reductions in TNF-alpha and IL-6 secretion are observed.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
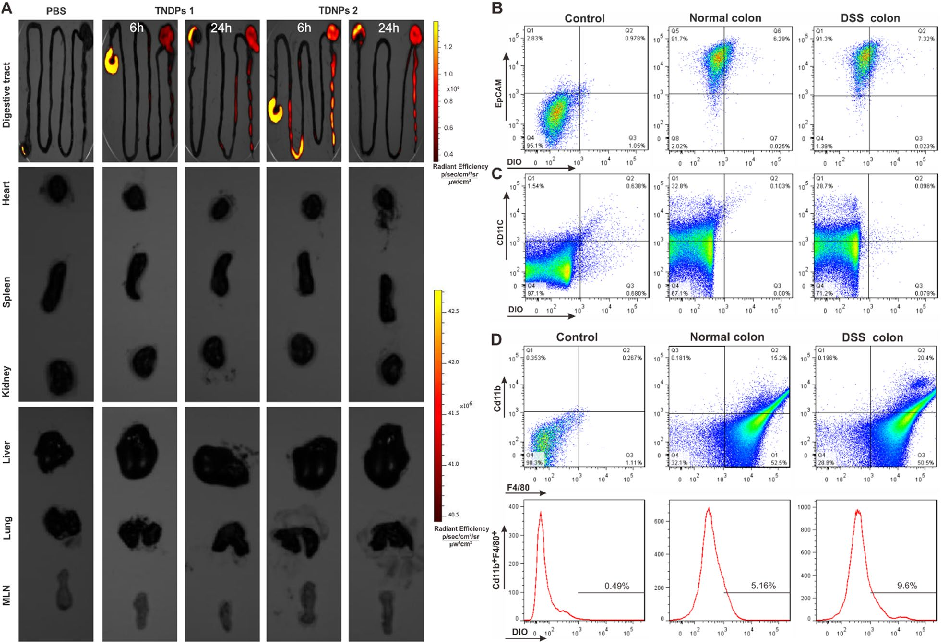
Biodistribution imaging using IVIS Spectrum reveals that TDNPs 2 preferentially accumulate in inflamed colonic tissue following oral administration. Fluorescence signals are minimal in non-target organs including heart, liver, spleen, lung, and kidney.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
Oral TDNPs 2 administration significantly attenuates disease activity index scores and colon shortening in DSS-induced colitis mice. Body weight recovery is also improved compared to untreated colitis controls.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
Intestinal permeability assessment indicates that TDNPs 2 treatment preserves gut barrier integrity in colitis mice. Tight junction protein expression, including ZO-1 and occludin, is maintained at near-normal levels.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
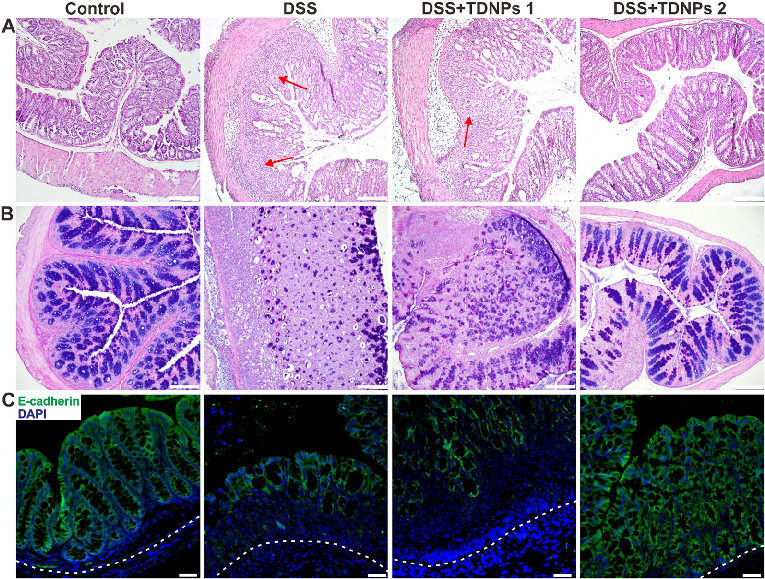
Histological examination with H&E staining reveals markedly reduced inflammatory cell infiltration and preserved goblet cell density in TDNPs 2-treated colitic mice. Colonic tissue architecture remains largely intact compared to severe disruption in untreated animals.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
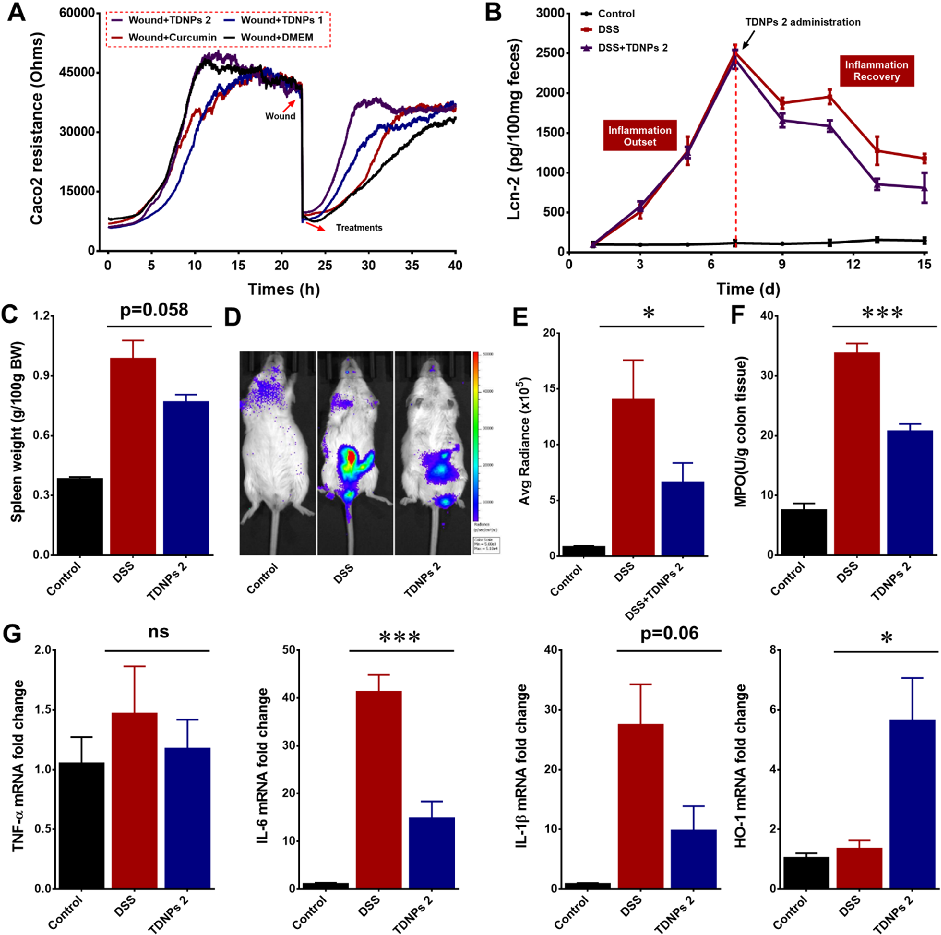
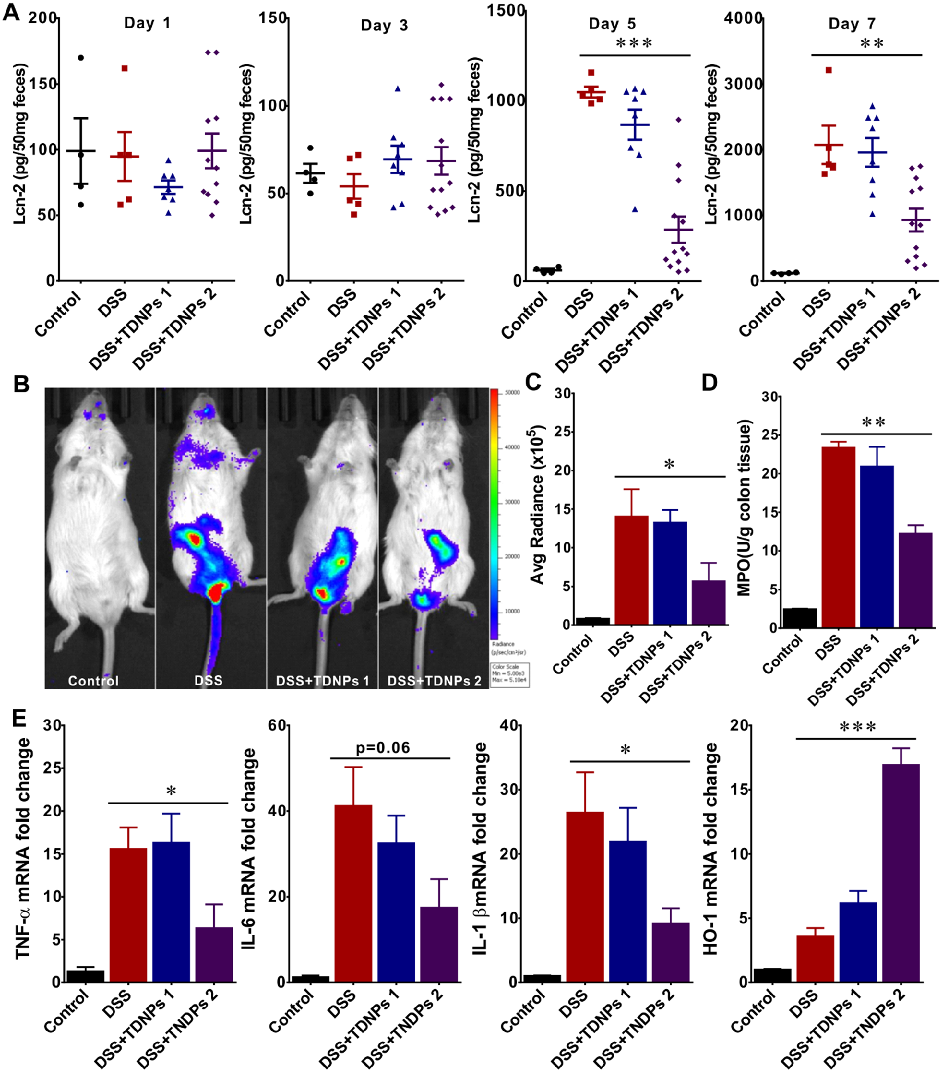
Wound healing assays and fecal lipocalin-2 quantification demonstrate that TDNPs 2 accelerate resolution of intestinal inflammation. Lcn-2 levels, a sensitive marker of intestinal inflammation, decrease significantly with treatment.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
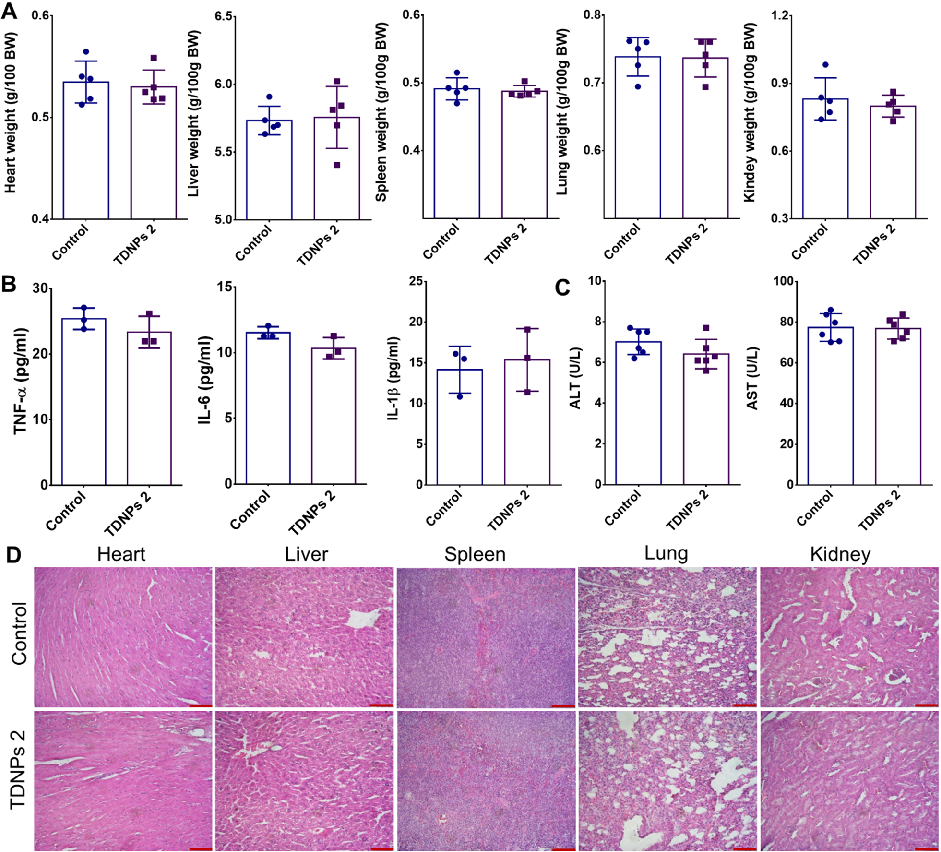
Biocompatibility evaluation shows no significant changes in vital organ weights, pro-inflammatory cytokines, or liver function indicators in TDNPs 2-treated mice. H&E staining of major organs confirms absence of toxicity.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
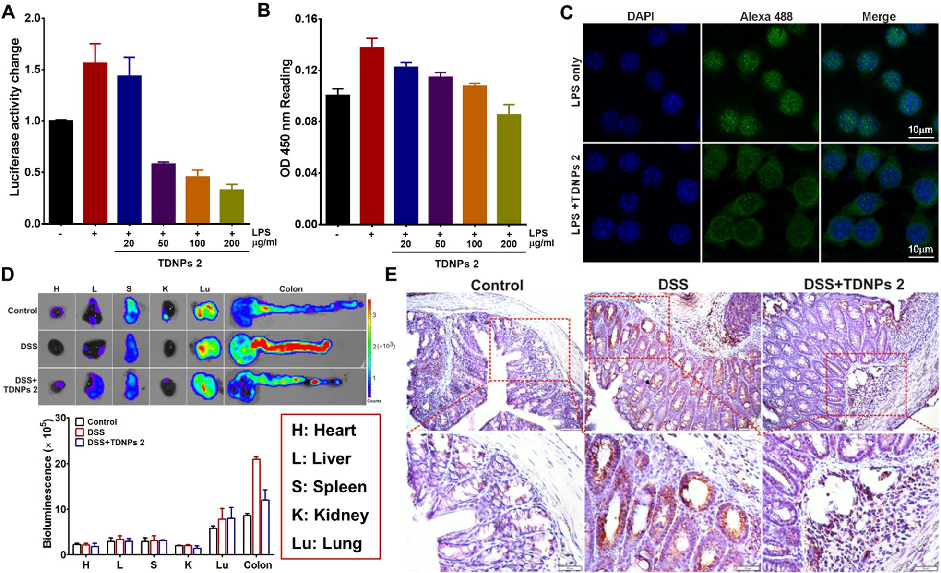
TDNPs 2 exert their protective effect at least partly through inactivation of the NF-kB signaling pathway. Reduced phospho-NF-kB p65 expression and decreased nuclear translocation indicate suppression of this key inflammatory cascade.
Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for …
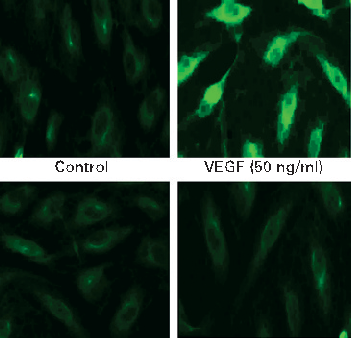
COX-2 protein expression is upregulated by VEGF stimulation in intestinal endothelial cells, and curcumin effectively blocks this induction. Prostaglandin E2 production follows a similar pattern of inhibition.
Curcumin inhibits VEGF-mediated angiogenesis in human intestinal microvascular endothelial cells through COX-2 …

MAPK signaling pathway activation by VEGF is attenuated by curcumin in a time- and dose-dependent manner. Phosphorylation of ERK, p38, and JNK is markedly reduced in curcumin-treated endothelial cells.
Curcumin inhibits VEGF-mediated angiogenesis in human intestinal microvascular endothelial cells through COX-2 …
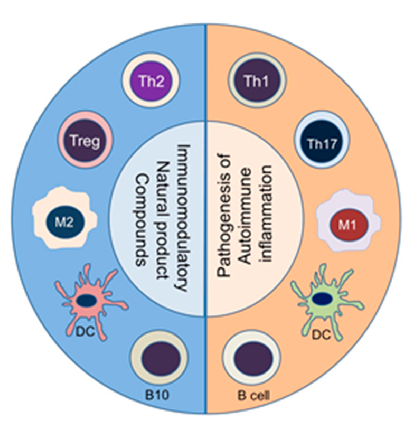
Natural product compounds shift the balance between pathogenic pro-inflammatory immune cells (right panel) and protective anti-inflammatory/immunomodulatory cell subsets (left panel). This diagram illustrates how diverse natural products can rebalance immune cell populations in autoimmune disease.
The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation.
In vivo models of autoimmune diseases have demonstrated therapeutic efficacy of various natural compounds. This figure presents preclinical evidence from animal models of rheumatoid arthritis, multiple sclerosis, or inflammatory bowel disease treated with natural products.
The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation.
Clinical translation of natural anti-inflammatory products requires understanding of bioavailability and dose-response relationships. This figure discusses the therapeutic potential and limitations of natural product-based interventions for autoimmune conditions.
The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation.
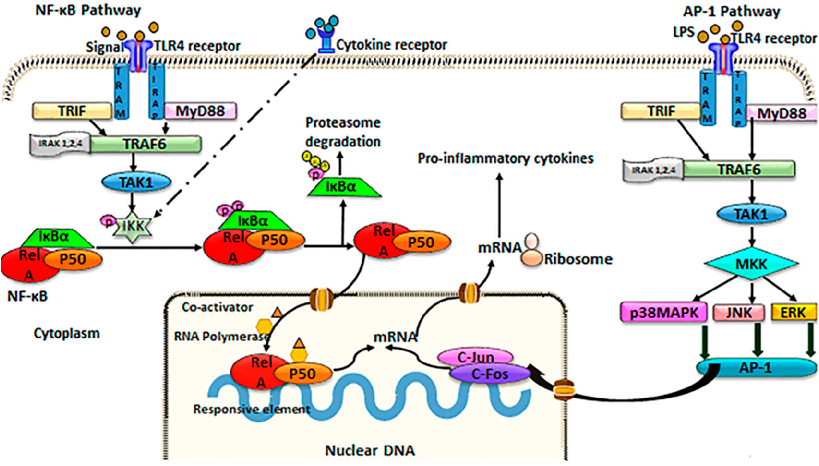
TLR4/NF-kB/AP-1 signaling is a key inflammatory pathway in intestinal disease. This schematic illustrates the cascade from TLR4 receptor activation through NF-kB and AP-1 transcription factors to pro-inflammatory gene expression, and indicates where curcumin may intervene.
Curcumin and Intestinal Inflammatory Diseases: Molecular Mechanisms of Protection.
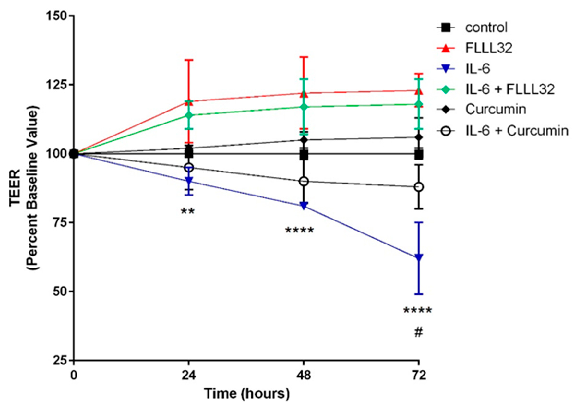
FLLL32 (a curcumin analogue) and curcumin both protect against IL-6-induced reduction of transepithelial electrical resistance (TEER) in T84 cell monolayers. This graph shows that curcumin preserves intestinal barrier integrity by counteracting cytokine-mediated tight junction disruption over 72 hours.
Curcumin and Intestinal Inflammatory Diseases: Molecular Mechanisms of Protection.
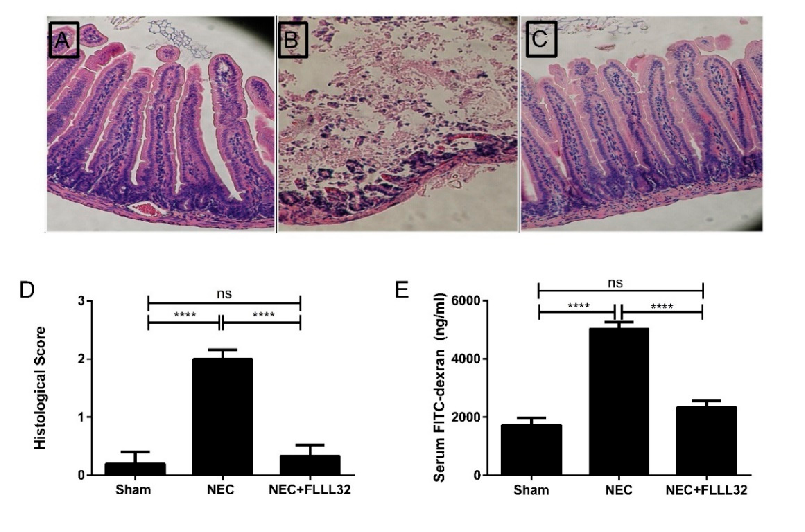
Curcumin's effects on intestinal tight junction proteins have been demonstrated in multiple experimental models. This figure presents protein expression or immunofluorescence data showing curcumin-mediated preservation of barrier function.
Curcumin and Intestinal Inflammatory Diseases: Molecular Mechanisms of Protection.
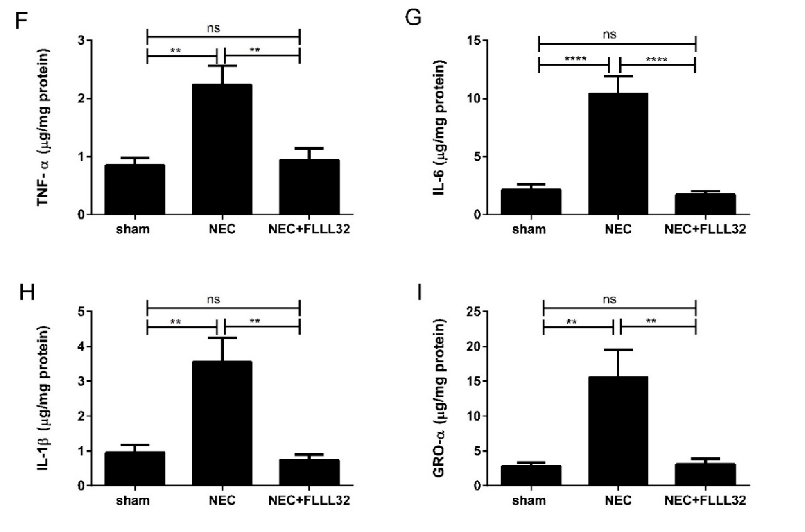
Animal models of necrotizing enterocolitis and colitis respond favorably to curcumin treatment. This figure presents in vivo data on curcumin's protective effects in experimental intestinal inflammation.
Curcumin and Intestinal Inflammatory Diseases: Molecular Mechanisms of Protection.
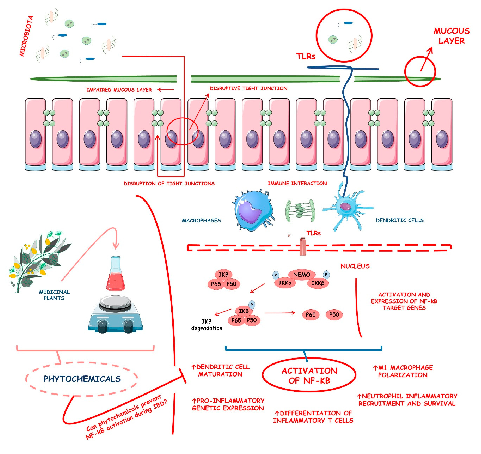
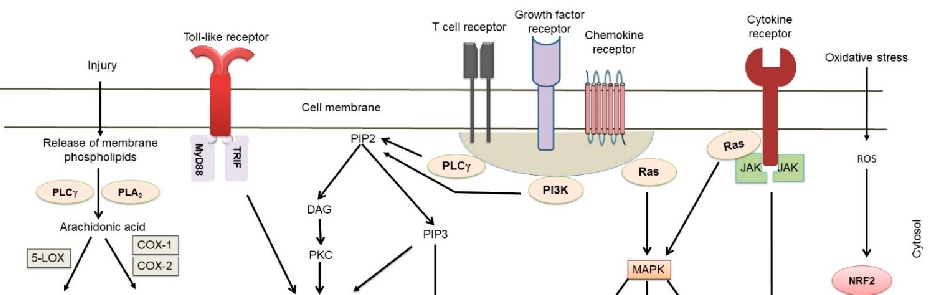
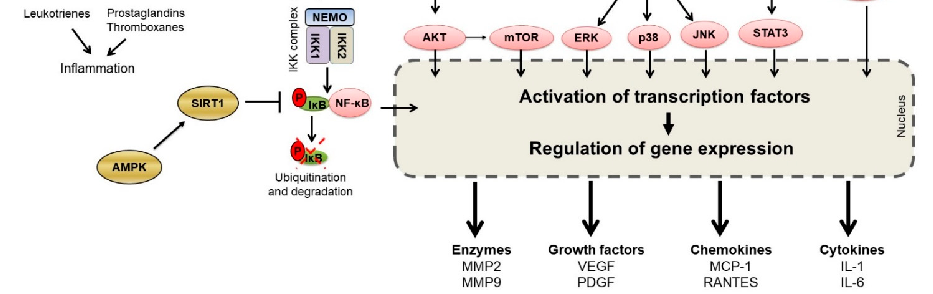
Inflammatory bowel diseases are associated with activation of the NF-kB signaling pathway. This diagram illustrates how various phytochemicals may counteract NF-kB pathway activation through inhibition of Toll-like receptors, IKK complex, and downstream transcriptional targets in IBD.
Phytochemicals and Regulation of NF-kB in Inflammatory Bowel Diseases: An Overview of …
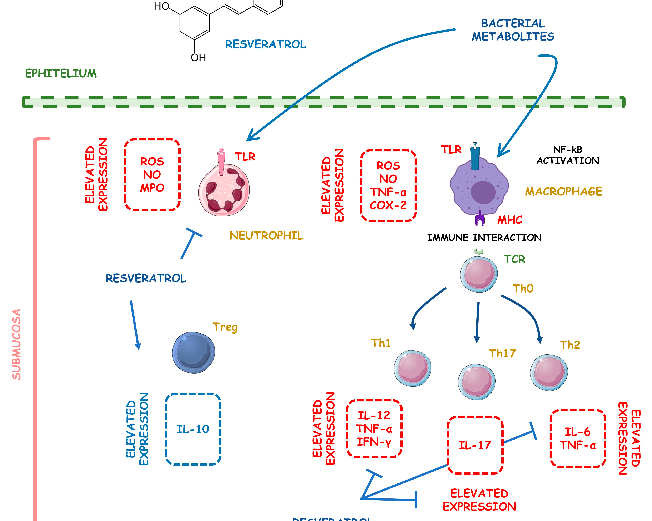
Supplementary figure from a comprehensive review of phytochemical effects on NF-kB regulation in inflammatory bowel disease. The review examines multiple plant-derived compounds with potential to attenuate chronic intestinal inflammation through modulation of key signaling pathways.
Phytochemicals and Regulation of NF-kB in Inflammatory Bowel Diseases: An Overview of …
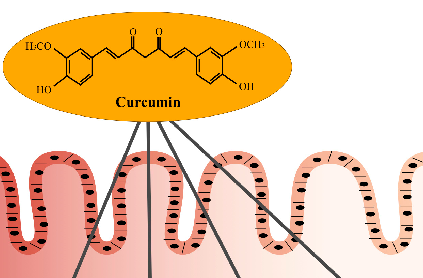
Curcumin's activity in the intestinal mucosa is illustrated, highlighting its anti-inflammatory mechanisms in the gastrointestinal tract. The diagram depicts how curcumin may modulate immune signaling pathways relevant to inflammatory bowel disease, including Crohn's disease and ulcerative colitis.
Curcumin and inflammatory bowel disease: potential and limits of innovative treatments.
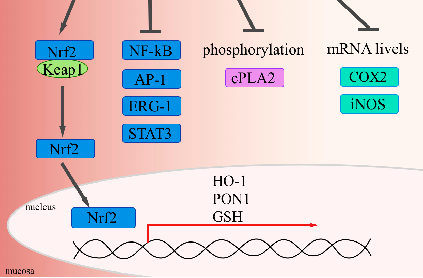
Molecular pathways and cellular targets of curcumin in inflammatory bowel disease are presented. The anti-oxidant and anti-inflammatory properties of curcumin are linked to its potential therapeutic applications in managing chronic gastrointestinal inflammation.
Curcumin and inflammatory bowel disease: potential and limits of innovative treatments.
Page 1 sur 2