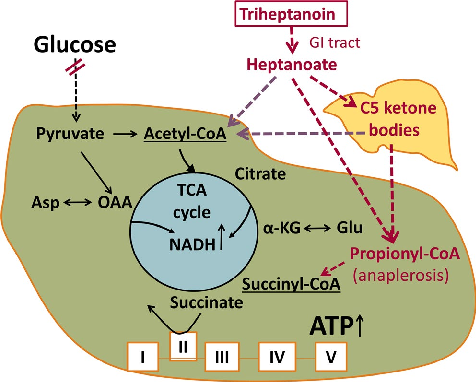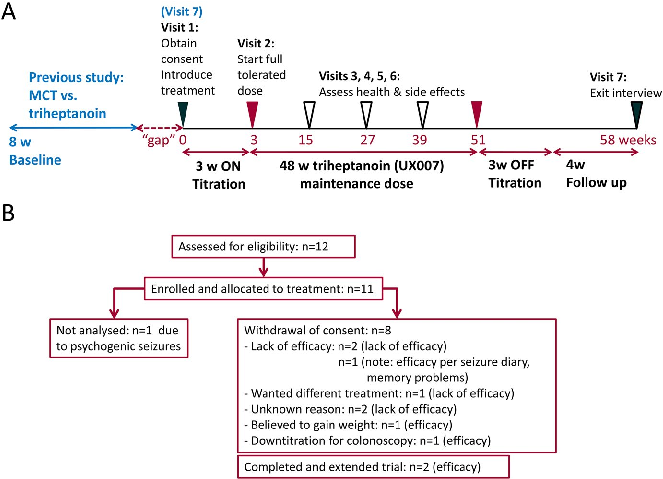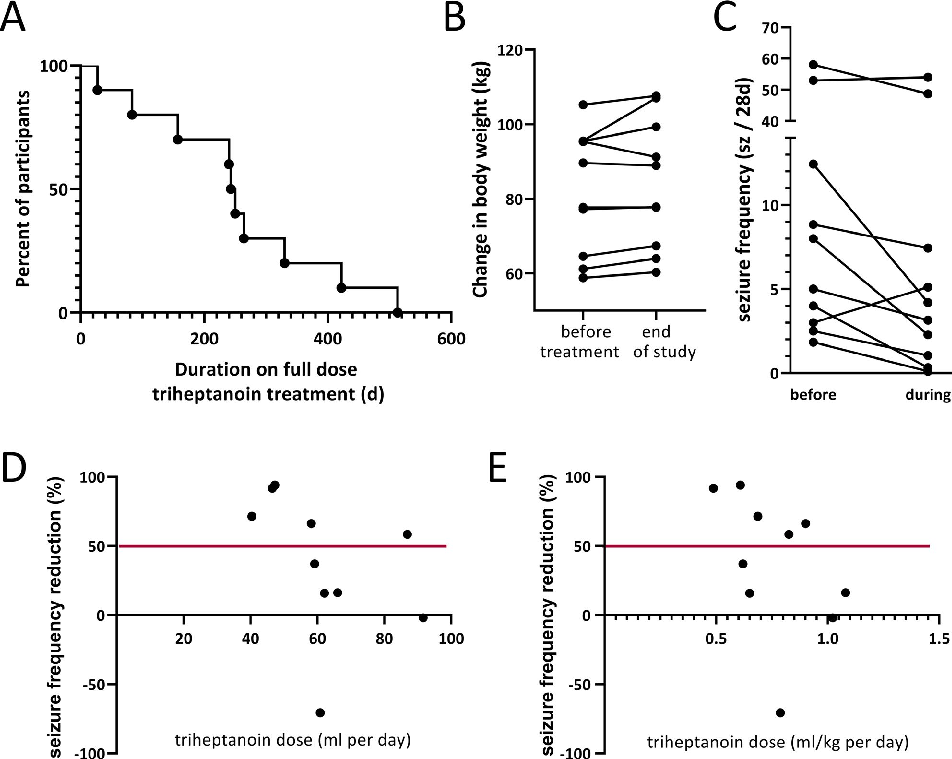
Figure 4
952 × 759px
· 145,8 KB
Source Paper
Open-label long-term treatment of add-on triheptanoin in adults with drug-resistant epilepsy.Cite This Figure
 > Source: Karin Borges et al. "Open-label long-term treatment of add-on triheptanoin in adults with drug-resist." *Epilepsia open*, 2020. PMID: [32524048](https://pubmed.ncbi.nlm.nih.gov/32524048/)
<figure> <img src="https://pdfs.citedhealth.com/figures/32524048/168.png" alt="FIGURE 3 A, Duration on full treatment dose and B the changes in body weight are shown for all participants. B, The changes in body weight (kg) from the beginning of the study until the end of the triheptanoin treatment vs until the end of the full study " /> <figcaption>Figure 4. FIGURE 3 A, Duration on full treatment dose and B the changes in body weight are shown for all participants. B, The changes in body weight (kg) from the beginning of the study until the end of the triheptanoin treatment vs until the end of the full study <br> Source: Karin Borges et al. "Open-label long-term treatment of add-on triheptanoin in adults with drug-resist." <em>Epilepsia open</em>, 2020. PMID: <a href="https://pubmed.ncbi.nlm.nih.gov/32524048/">32524048</a></figcaption> </figure>