Efficacy and tolerability of α-galactosidase in treating gas-related symptoms in children: a randomized, double-blind, placebo controlled trial.
Study Design
- Çalışma Türü
- Randomized Controlled Trial
- Popülasyon
- Children with gas-related GI symptoms
- Süre
- 2 weeks
- Müdahale
- Efficacy and tolerability of α-galactosidase in treating gas-related symptoms in children: a randomized, double-blind, placebo controlled trial. None
- Karşılaştırıcı
- Placebo
- Birincil Sonuç
- Gas-related symptoms in children
- Etki Yönü
- Positive
- Yanlılık Riski
- Low
Abstract
BACKGROUND: Gas-related symptoms represent very common complaints in children. The reduction of gas production can be considered as a valuable target in controlling symptoms. α-galactosidase has been shown to reduce gas production and related symptoms in adults. To evaluate the efficacy and tolerability of α-galactosidase in the treatment of gas-related symptoms in pediatric patients. METHODS: Single center, randomized, double-blind, placebo-controlled, parallel group study performed in tertiary care setting. Fifty-two pediatric patients (32 female, age range 4-17) with chronic or recurrent gas-related symptoms were randomized to receive placebo (n = 25) or α-galactosidase (n = 27). Both treatments were given as drops or tablets, according to body weight for 2 weeks. The primary endpoint was the reduction in global distress measured by the Faces Pain Scale-Revised (FPS-R) at the end of treatment compared to baseline. Secondary endpoints were the reduction in severity and frequency of gas-related symptoms as recorded by parents and/or children. RESULTS: α-galactosidase significantly reduced global distress (p = 0.02) compared to placebo. The digestive enzyme decreased the number of days with moderate to severe bloating (p = 0.03) and the proportion of patients with flatulence (p = 0.02). No significant differences were found for abdominal spasms and abdominal distension. No adverse events were reported during treatment. CONCLUSIONS: Although larger and longer trials are needed to confirm this result, α-galactosidase seems to be a safe, well tolerated and effective treatment for gas-related symptoms in the pediatric population. TRIAL REGISTRATION: ClinicalTrials.gov, NCT01595932.
Kısaca
Although larger and longer trials are needed to confirm this result, α-galactosidase seems to be a safe, well tolerated and effective treatment for gas-related symptoms in the pediatric population.
Full Text
RESEARCH ARTICLE Open Access
Figure 2 Change of FPS-score in any patient before and after the treatment.
Figure 3 Physician overall evaluation at end of treatment and at follow-up.
of gas by colonic bacteria may have been expected to have other beneficial effects on distension of colon segments and abdomen.
The strength of our findings is mitigated by the findings in some secondary efficacy endpoints. No significant changes were observed between α-galactosidase and placebo for the “physician’s overall evaluation” at end of treatment and at follow-up. This may be related to the fact that this global evaluation may include other symptoms (in addition to bloating) which are poorly responsive to α-galactosidase such as symptoms that are not gas-related. The effect of α-galactosidase on gas related symptoms does not appear to be long-lasting. The improvement observed at the end of treatment was not sustained after 2 weeks of follow-up but this is consistent with the mechanism of action of the enzyme.
The limitations of our study are the short duration of treatment (2 weeks only) and the relatively small sample compared to trials in adult populations.
There is not much information about placebo response on gas related symptoms in children in literature. In our study, we did not see apparent difference in the placebo response between children of less and above 8 years. There were no reported adverse effects during the study. Based on our study there is no safety issue related to the use of α-galactosidase in children with bloating and gas-related symptoms. This is also supported by clinical practice and post-marketing experience on the use of α-galactosidase in children as well as in adults.
Oral α-galactosidase was effective in the short-term treatment of gas-related symptoms in children who were referred to specialist care. Its use is also supported by non-toxicity, good tolerability and availability of the formulation in drops, suitable for pediatric patients.
Further longer and larger randomized controlled clinical trials are needed to assess the efficacy and usefulness of α-galactosidase in children with gas-related symptoms and to identify subgroups of patients who are more likely to respond (or fail) to this agent.
A detailed medical history may help physicians to recognize children in whom, based on the medical history, food appears to induce or favor gas-related symptoms.
A symptomatic treatment approach for these children could be beneficial: if there is no significant improvement in the main target symptom or overall symptoms after 2 weeks, a different alternative approach should be considered.
This study was placebo-controlled, as currently there is no reference treatment for gas-related symptoms in FGID. The evidence of efficacy of antiflatulents such as simethicone and activated charcoal is weak, together with that of probiotics, such as different strains of Lactobacillus or Bifidobacterium spp. Similarly, there does not appear to be any robust evidence for using antispasmodics,
prokinetics or non-absorbable antibiotics if the target symptom is bloating. Our study was carried out in children who predominantly had bloating and gas-related symptoms, with no significant abdominal pain. Consequently, the beneficial results obtained in this study cannot be considered valid for children suffering from different variants of functional gastrointestinal disorder (IBSpredominant constipation or IBS-predominant diarrhea).
We acknowledge the limitations of the present study. The number of included patients is limited and their age range is wide. In addition, although the “gas-related syndrome” may be considered as a functional digestive syndrome characterized by non specific gastrointestinal symptoms, including bloating, flatulence, abdominal distension and discomfort that the patient attributes to an excess of abdominal gas, however this condition is poorly defined and overlaps with IBS. All our patients meet Rome III criteria for IBS and the American College of Gastroenterology IBS Task Force recommends that further investigations are unnecessary in young patients without alarm features with the exception of celiac disease serology. However, different diagnoses, including lactose intolerance, fructose intolerance, small bowel bacterial overgrowth and aerophagia, were not formally investigated in our patients. Finally, although we instructed the parents of our patients to continue with their usual diet over the study period, the dietary habits of our children were not formally recorded in diary during the study. For this reason, we can not relate the symptoms to the type of diet (high and low fiber intake).
This study has shown for the first time that pediatric patients with predominant gas-related symptoms had a better response than placebo in the short term use of oral α-galactosidase. The improvement of symptoms becomes evident in a few days, in particular the reduction of the severity and frequency of bloating and flatulence. This effect tends to disappear in half of patients 2 weeks after treatment withdrawal.
Conclusions
In conclusion, oral α-galactosidase was effective and very well tolerated in the treatment of bloating and gas-related symptoms in children and adolescents aged 4 – 17 years.
Competing interests The authors declare that they have no competing interests.
Authors’ contributions DNG and OS designed the study and wrote the manuscript. MS and FF followed-up patients. CC, BG and AM designed the study. CS is the head of the Pediatric Gastroenterology Unit, approved the study design and strongly revised a draft of the paper. Guarantor of the article: CS. All authors read and approved the final manuscript.
Author details
Pediatric Gastroenterology and Liver Unit, Sapienza – University of Rome, Rome, Italy. 2Department of Medical Science, University of Bologna, Bologna, Italy.
Received: 15 November 2012 Accepted: 18 September 2013 Published: 24 September 2013
References
- 1. Longstreth GF, et al: Functional bowel disorders. Gastroenterology 2006, 130:1480 1491.
- 2. Sandler RS, Stewart WF, Liberman JN, et al: Abdominal pain, bloating and diarrhea in the United States: prevalence and impact. Dig Dis Sci 2000, 45:1166–1171.
- 3. Youssef NN, et al: Quality of life for children with functional abdominal pain: a comparison study of patients’ and parents’ perceptions. Pediatrics 2006, 117:54 59.
- 4. Spiller R: New insights into bloating and abdominal distension: is it all outlet obstruction? Am J Gastroenterol 2010, 105:888 889.
- 5. Lea R, Whorwell PJ: Expert commentary – bloating, distension, and the irritable bowel syndrome. MedGenMed 2005, 7:18.
- 6. Issa B, Wafaei NA, Whorwell PJ: Abdominal bloating and distension: what is the role of the microbiota. Dig Dis Sci 2012, 57:4 8.
- 7. Fink RN, Lembo AJ: Intestinal gas. Curr Treat Options Gastroenterol. 2001 Aug, 4:333 337.
- 8. Jones M: Bloating and intestinal gas. Curr Treat Options Gastroenterol 2005, 8:311 318.
- 9. Friis H, Bode S, Rumessen JJ, et al: Effect of simethicone on lactuloseinduced H2 production and gastrointestinal symptoms. Digestion 1991, 49:227–230.
- 10. Suarez FL, Furne J, Springfield J, Levitt MD: Failure of activated charcoal to reduce the release of gases produced by the colonic flora. Am J Gastroenterol 1999, 94:208–212.
- 11. O’Mahony L, McCarthy J, Kelly P, et al: Lactobacillus and bifidobacterium in irritable bowel syndrome: symptom responses and relationship to cytokine profiles. Gastroenterology 2005, 128:541 551.
- 12. Guandalini S, Magazzù G, Chiaro A, et al: VSL#3 improves symptoms in children with irritable bowel syndrome: a multicenter, randomized, placebo-controlled, double-blind, crossover study. J Pediatr Gastroenterol Nutr 2010, 51:24 30.
- 13. Ganiats TG, Norcross WA, Halverson AL, et al: Does beano prevent gas? A double-blind crossover study of oral alfa-galactosidase to treat dietary oligosaccharide intolerance. J Fam Pract 1994, 39:441 445.
- 14. Solomons N, et al: Orally-ingested, microbial alpha-galactosidases produce effective in vivo, intraintestinal digestion of the Bean oligosaccharide, raffinose. Gastroenterology 1991, 100:A251.
- 15. Di Stefano M, Miceli E, Gotti S, et al: The effect of oral α-galactosidase on intestinal gas production and gas-related symptoms. Dig Dis Sci 2007, 52:78 83.
- 16. Rippel SW, et al: Pediatric patients with dyspepsia have chronic symptoms, anxiety, and lower quality of life as adolescents and adults. Gastroenterology 2012, 142:754–761.
- 17. Saps M, et al: Multicenter, randomized, placebo-controlled trial of amitriptyline in children with functional gastrointestinal disorders. Gastroenterology 2009, 137:1261–1269.
- 18. Hicks CL, et al: The faces pain scale-revised: toward a common metric in pediatric pain measurement. Pain 2001, 93:173 183.
- 19. Irvine EJ, et al: Design of treatment trials for functional gastrointestinal disorders. Gastroenterology 2006, 130:1538–1551.
- 20. Spiller R, Aziz Q, Creed F, et al: Guidelines on the irritable bowel syndrome: mechanisms and practical management. Gut 2007, 56:1770 1798.
- 21. Pimentel M, Lembo A, Chey WD, et al: Rifaximin therapy for patients with irritable bowel syndrome without constipation. N Engl J Med 2011, 364:22–32.
- 22. Schmulson M, Chang L: Review article: the treatment of functional abdominal bloating and distension. Aliment Pharmacol Ther 2011, 33:1071 1086.
doi:10.1186/1471-230X-13-142 Cite this article as: Di Nardo et al.: Efficacy and tolerability of αgalactosidase in treating gas-related symptoms in children: a randomized, double-blind, placebo controlled trial. BMC Gastroenterology 2013 13:142.
Submit your next manuscript to BioMed Central and take full advantage of:
- • Convenient online submission
- • Thorough peer review
- • No space constraints or color figure charges
- • Immediate publication on acceptance
- • Inclusion in PubMed, CAS, Scopus and Google Scholar
- • Research which is freely available for redistribution
Submit your manuscript at www.biomedcentral.com/submit
Figures
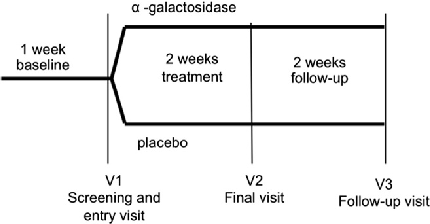
Figure 1
Participant enrollment and randomization flow for the double-blind trial evaluating α-galactosidase in children with gas-related symptoms is depicted.
flowchart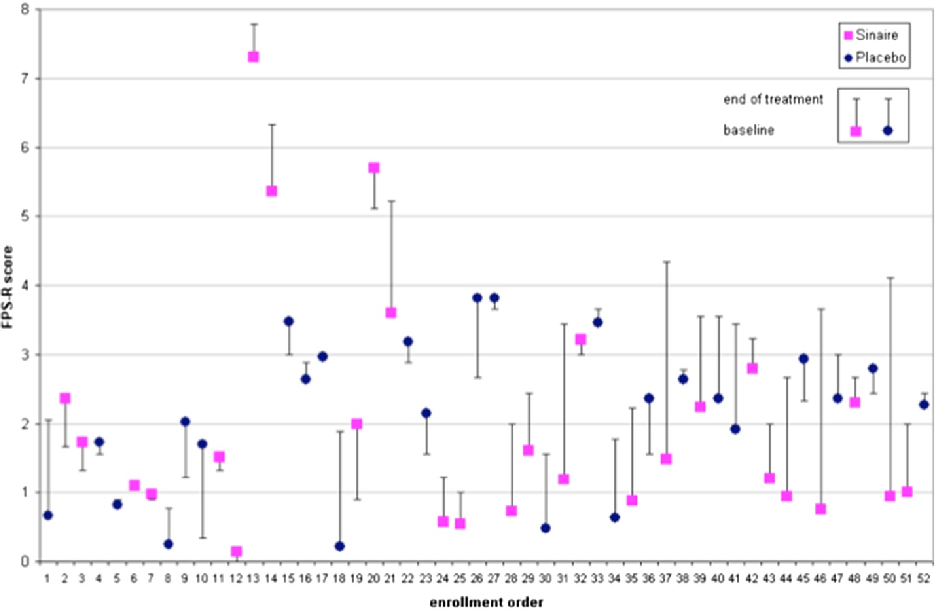
Figure 2
Frequency of gas-related episodes is compared between children receiving α-galactosidase and placebo over the treatment period, indicating changes in symptom burden.
chart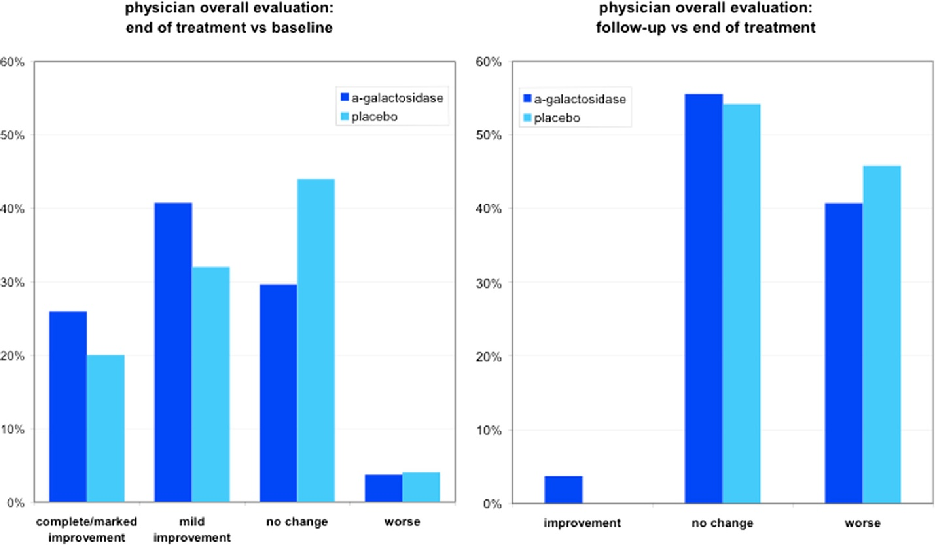
Figure 3
Overall symptom severity scores for gas-related complaints in children are tracked across the study timeline, comparing the α-galactosidase and placebo arms.
chartUsed In Evidence Reviews
Similar Papers
Molecular genetics and metabolism · 2005
Gastrointestinal manifestations of Fabry disease: clinical response to enzyme replacement therapy.
The American journal of gastroenterology · 2018
Increasing Symptoms in Irritable Bowel Symptoms With Ingestion of Galacto-Oligosaccharides Are Mitigated by α-Galactosidase Treatment.
Digestive diseases and sciences · 2007
The effect of oral alpha-galactosidase on intestinal gas production and gas-related symptoms.
Clinical biochemistry · 2015
A family with various symptomatology suggestive of Anderson-Fabry disease and a genetic polymorphism of alpha galactosidase A gene.
Scandinavian journal of gastroenterology · 2016
Does oral α-galactosidase relieve irritable bowel symptoms?
International journal of environmental research and public health · 2021