Understanding the Immunomodulatory Effects of Bovine Colostrum: Insights into IL-6/IL-10 Axis-Mediated Inflammatory Control.
Study Design
- Tipo de Estudo
- In Vitro
- População
- In vitro (Caco-2/THP-1 co-culture model)
- Intervenção
- Understanding the Immunomodulatory Effects of Bovine Colostrum: Insights into IL-6/IL-10 Axis-Mediated Inflammatory Control. None
- Comparador
- None
- Desfecho Primário
- Cytokine production (IL-6/IL-10 axis)
- Direção do Efeito
- Positive
- Risco de Viés
- Unclear
Abstract
Bovine colostrum (COL), the first milk secreted by lactating cows postpartum, is a rich source of bioactive compounds that exert a significant role in the survival, growth, and immune development of neonatal calves. This study investigated the immunomodulatory effects of COL on cytokine production in vitro using a Caco-2/THP-1 macrophage co-culture model stimulated with Phorbol 12-myristate 13-acetate (PMA). COL pretreatment significantly reduced IL-6 (241.3 pg/mL) production induced by PMA (p < 0.05), while increasing IL-10 production (45.3 pg/mL), in comparison to PMA control (441.1 and 12.5 pg/mL, respectively). Further investigations revealed that the IL-6 suppressive effect of colostrum was heat-sensitive and associated with components of higher molecular mass (100 kDa). Moreover, colostrum primarily influenced THP-1 macrophages rather than Caco-2 epithelial cells. The effects of colostrum on IL-6 production were associated with reduced NF-κB activation in THP-1 macrophages. In calf-FMT transplanted C57BL/6 murine model, colostrum decreased intestinal permeability, reduced immune cell infiltration and intestinal score, and suppressed IL-6 (142.0 pg/mL) production during S. typhimurium infection, in comparison to control animals (215.2 pg/mL). These results suggest the immunomodulatory activity of bovine colostrum and its potential applications in inflammatory disorders. Further studies are needed to elucidate the underlying mechanisms and validate the findings in bovine models.
Resumo Rápido
Investigations revealed that the IL-6 suppressive effect of colostrum was heat-sensitive and associated with components of higher molecular mass (100 kDa) and influenced THP-1 macrophages rather than Caco-2 epithelial cells, which highlights the immunomodulatory activity of bovine colostrums and its potential therapeutic applications in inflammatory disorders.
Full Text
veterinary sciences
Article
Understanding the Immunomodulatory Effects of Bovine Colostrum: Insights into IL-6/IL-10 Axis-Mediated Inflammatory Control
Ramune Grigalevicˇi˙ ut¯ e˙ 1,2,*, Paulius Matusevicˇius 2, Rita Plancˇiunien¯ e˙ 3, Rolandas Stankevicˇius 2, Eivina Radzevicˇiut¯ e-Valcˇiuk˙ e˙ 4 , Austeja Balevicˇi˙ ut¯ e˙ 5 , Augustinas Želvys 4 , Aukse Zinkevicˇien˙ e˙ 4, Vilma Zigmantaite˙ 1 , Audrius Kucˇinskas 1 and Povilas Kavaliauskas 1,6,7,8,*
Citation: Grigalevicˇiut¯ e, R.;˙ Matusevicˇius, P.; Plancˇiunien¯ e, R.;˙ Stankevicˇius, R.; Radzevicˇiut¯ e-˙ Valcˇiuke, E.; Balevicˇi˙ ut¯ e, A.; Želvys,˙ A.; Zinkevicˇiene, A.; Zigmantait˙ e, V.;˙ Kucˇinskas, A.; et al. Understanding the Immunomodulatory Effects of Bovine Colostrum: Insights into IL-6/IL-10 Axis-Mediated Inflammatory Control. Vet. Sci. 2023, 10, 519. https://doi.org/10.3390/ vetsci10080519
Academic Editors: Awad Shehata, Guillermo Tellez-Isaias and Wolfgang Eisenreich
Copyright: © 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https:// creativecommons.org/licenses/by/ 4.0/).
- 1 Biological Research Center, Lithuanian University of Health Sciences, Tilzes Str. 18/7, LT-47181 Kaunas, Lithuania; [email protected] (V.Z.); [email protected] (A.K.)
- 2 Department of Animal Nutrition, Lithuanian University of Health Sciences, Tilzes Str. 18, LT-47181 Kaunas, Lithuania; [email protected] (P.M.); [email protected] (R.S.)
- 3 Institute of Microbiology and Virology, Lithuanian University of Health Sciences, Eiveniu Str. 4, LT-50161 Kaunas, Lithuania; [email protected]
- 4 Centre for Innovative Medicine, Department of Immunology, Santariskiu Str. 5, LT-08410 Vilnius, Lithuania; [email protected] (E.R.-V.); [email protected] (A.Ž.); [email protected] (A.Z.)
- 5 Institute of Environmental Medicine, Toxicology Unit, Karolinska Institutet, Stockholm, Solnavägen 1, 17177 Solna, Sweden; [email protected]
- 6 Joan and Sanford I. Weill Department of Medicine, Weill Cornell University, 1300 York Avenue, New York, NY 10065, USA
- 7 Department of Microbiology and Immunology, University of Maryland Baltimore School of Medicine, Baltimore, MD 21201, USA
- 8 Institute of Infectious Diseases and Pathogenic Microbiology, Birstono Str. 38A, LT-59116 Prienai, Lithuania
* Correspondence: [email protected] (R.G.); [email protected] (P.K.)
Simple Summary: This study explores the immunomodulatory properties of bovine colostrum (COL), the initial milk produced by lactating cows, on cytokine production in vitro and in a novel murine calffecal microbiota transplantation (FMT) model. The researchers utilized a Caco-2/THP-1 macrophage co-culture model stimulated with Phorbol 12-myristate 13-acetate (PMA) to investigate the effects of COL on cytokine production. The findings indicate that COL pretreatment significantly reduced IL-6 production while enhancing IL-10 production. The IL-6 suppressive effect was heat-sensitive and associated with components of higher molecular mass (100 kDa). Colostrum demonstrated decreased intestinal permeability, reduced immune cell infiltration, and suppressed IL-6 production during S. typhimurium infection. These results highlight the immunomodulatory potential of bovine colostrum and its prospective therapeutic applications in inflammatory disorders. Further research is necessary to elucidate the underlying mechanisms and corroborate the findings in bovine models.
Abstract: Bovine colostrum (COL), the first milk secreted by lactating cows postpartum, is a rich source of bioactive compounds that exert a significant role in the survival, growth, and immune development of neonatal calves. This study investigated the immunomodulatory effects of COL on cytokine production in vitro using a Caco-2/THP-1 macrophage co-culture model stimulated with Phorbol 12-myristate 13-acetate (PMA). COL pretreatment significantly reduced IL-6 (241.3 pg/mL) production induced by PMA (p < 0.05), while increasing IL-10 production (45.3 pg/mL), in comparison to PMA control (441.1 and 12.5 pg/mL, respectively). Further investigations revealed that the IL6 suppressive effect of colostrum was heat-sensitive and associated with components of higher molecular mass (100 kDa). Moreover, colostrum primarily influenced THP-1 macrophages rather than Caco-2 epithelial cells. The effects of colostrum on IL-6 production were associated with reduced NF-κB activation in THP-1 macrophages. In calf-FMT transplanted C57BL/6 murine model, colostrum decreased intestinal permeability, reduced immune cell infiltration and intestinal score, and suppressed IL-6 (142.0 pg/mL) production during S. typhimurium infection, in comparison to control animals (215.2 pg/mL). These results suggest the immunomodulatory activity of bovine
Vet. Sci. 2023, 10, 519. https://doi.org/10.3390/vetsci10080519 https://www.mdpi.com/journal/vetsci
colostrum and its potential applications in inflammatory disorders. Further studies are needed to elucidate the underlying mechanisms and validate the findings in bovine models.
Keywords: bovine colostrum; cytokine production; immunomodulation; intestinal permeability; THP-1 macrophages; Caco-2 cells; NF-κB; S. typhimurium infection; inflammatory disorders
1. Introduction
Bovine colostrum (COL), the first milk produced by lactating cows postpartum, is a rich source of bioactive compounds that exert a significant role in the survival, growth, and immune development of neonatal calves [1–3]. Colostrum contains numerous bioactive constituents, including maternal immunoglobulins, growth factors, antimicrobial peptides, cytokines, and even immune cells [2–4]. These constituents collectively confer passive immunity against diverse pathogens, promoting intestinal microbiome development, maintaining anti-inflammatory homeostasis, and facilitating immune tolerance to microbiota [3,4].
Neonatal calves face notable challenges to their overall health, growth, and survival, particularly due to intestinal inflammation and infectious or non-infectious diarrheal diseases [5,6]. The gastrointestinal tract of neonates is highly susceptible to inflammatory and diarrheal conditions during this critical developmental period [6]. Early colonization of the intestinal tract by microbiota begins in the initial hours and continues through weeks and months of life [7,8]. The diversity of the microbial community is strongly influenced by the inflammatory status of the intestinal environment and could be further amplified by pathogenic microbiota and their secreted metabolites or factors. Additionally, several evidence suggests that dysregulation of innate immune responses in the gut, coupled with alterations in the intestinal microbiota composition, especially by the colonization of pathogenic flora, significantly contributes to the pathogenesis of inflammatory bowel diseases, leading to persistent diarrhea [8]. Therefore, gaining a comprehensive understanding of the molecular mechanisms underlying colostrum-mediated responses during inflammation and microbiota colonization holds substantial significance in improving calf health, preventing diseases, and developing novel host-directed therapeutic approaches.
Interleukin-6 (IL-6) represents a multifunctional pro-inflammatory cytokine that plays pivotal roles in numerous biological processes, including inflammation, immune regulation, and the interplay between innate and adaptive immune responses. Within the intestinal milieu, IL-6, previously shown to be crucial for the pathogenesis of inflammatory bowel diseases in humans, orchestrates acute inflammation, crucially contributes to immune cell recruitment, and modulates inflammatory processes [9]. Various cell types within the intestine, including macrophages, dendritic cells, T cells, and epithelial cells, produce IL-6 in response to diverse stimuli, such as infections, tissue damage, and inflammation [10]. Due to its highly deleterious nature, the production of IL-6 is tightly regulated; however, it can be chronically upregulated under inflammatory conditions, leading to tissue damage, compromised intestinal barrier function, and systemic infections [11].
Several studies have provided early evidence supporting the immunomodulatory activity of COL and milk derived from ruminant species. A study conducted by Kao et al. investigated the impact of goat milk supplementation in mice sensitized with ovalbumin to induce airway inflammation, revealing a significant increase in splenic CD3+ cell numbers [12]. The authors also showed that mice treated with goat milk demonstrated heightened production of interleukin-10 (IL-10) and interferon-gamma (IFNγ), suggesting an immune-modulating effect of goat milk on the cellular and cytokine responses [12]. Moreover, several distinct differences were observed in the composition of COL among different ruminant species [13]. Bovine COL exhibited higher levels of immunoglobulins IgG and IgA, while sheep colostrum displayed elevated IgG content compared to both goat and bovine counterparts [14,15]. Notably, goat COL demonstrated a unique composition
of growth factors, small regulatory RNA, and cytokines, highlighting potential variances in immune system development and maturation [16]. Despite early insights into the differences in colostrum among ruminants, the detailed composition of colostrum as well as extensive comparisons between various ruminants remains poorly described.
Recent studies have demonstrated the immunomodulatory activity of bovine COL within the intestinal environment. Bovine COL comprises cytokines, growth factors, and maternal-derived immunoglobulins (Ig’s), such as IgG and IgA, which act synergistically to regulate immune responses both on mucosal and systemic levels [2,17,18]. Additionally, COL is enriched with immunomodulatory oligosaccharides, sialyloligosaccharides, as well as various glycoproteins and glycolipids, which further modulate immune function [18,19]. Throughout the lactation period, the concentrations of bioactive molecules in COL undergo intricate and dynamic changes, possibly tailored to meet the various needs of the growing calf [20]. As lactation progresses into the transitional phase, the composition of colostrum undergoes changes. The levels of maternal immunoglobulins, particularly IgG, begin to decline gradually [14,17]. Moreover, during this phase, the concentration of lactoferrin, antimicrobial peptides, and defensins increases, enhancing calf defense against pathogens [17].
It has been established that COL can augment the production of anti-inflammatory interleukin-10 (IL-10), thereby favorably shifting immune responses from pro-inflammatory Th1 responses towards anti-inflammatory Th2 responses [21]. Notably, administration of COL in a dextran sulfate-induced colitis model resulted in increased numbers of CD11bpositive leukocytes producing IL-10, ultimately leading to diminished intestinal inflammation via the Src–Akt pathway [22]. Furthermore, IL-10 can negatively regulate IL-6 production by potentially modulating nuclear factor-kappa B (NF-κB) and signal transducer and activator of transcription 3 (STAT3) signaling pathways. However, the precise regulatory mechanisms underlying the interplay between IL-6 and IL-10 in response to colostrum within the intestine remain incompletely characterized.
Our previous studies have elucidated the critical impact of COL administration on the health and development of neonatal Holstein calves [23]. Notably, we observed heightened leukocyte numbers in colostrum-fed calves compared to the control group, providing preliminary evidence of the potential immunomodulatory activity of colostrum [23]. Building upon this, with our interest in comprehensively deciphering the molecular mechanisms underlying COL’s effects in calves, we aimed to employ advanced immunological, cellular biology, and in vivo animal models to delineate the immunomodulatory properties of COL. In this study, we present the development of novel calf-fecal microbiota transplantation (FMT) into convenient C57BL/6 mice models, in which mice harbor engrafted calf microbiota, as an approach to investigate the alterations induced by colostrum within the intestinal environment. Our findings provide experimental evidence regarding the interplay between IL-6 and IL-10 mediated by colostrum in both in vitro and in vivo models and propose the utility of colostrum as a preventative or therapeutic tool to mitigate intestinal inflammation in calves.
2. Materials and Methods
- 2.1. Ethics
- 2.2. Collection and Processing of Bovine Colostrum
Bovine colostrum (COL) was collected from randomly selected 10 healthy Holstein Friesian cows in their first or second parity from a farm (Lytagra Agriculture company, Naujieji Bernatoniai, Lithuania). Before COL collection, cows were evaluated by a veterinary doctor to ensure that there is no evidence of mastitis and that the animals are healthy.
To ensure consistent experimental conditions and eliminate the potential influence of diet or animal management on the results, the colostrum was collected from the same farm where all the animals were managed in a uniform manner, including diet and circadian rhythm. The calves were born within a similar time frame (between 24–48 h). Right after the cows gave birth, they were milked every hour for 4 h to collect early-produced colostrum (10–25 mL). The colostrum samples obtained from different cows were promptly cooled on ice, pooled together, aliquoted into 10 mL portions, and then frozen at −80 ◦C until the day of the experiments.
- 2.2.1. Colostrum Serum Preparation
- 2.2.2. Colostrum Serum Protein Quantification
- 2.2.3. Colostrum Inactivation by Heat
- 2.2.4. Ultrafiltration of Colostrum
To generate 10, 50, and 100 kDa fractions of colostrum, Amicon-Ultra 15 filters (SigmaAldrich, #UFC9010, UFC9050, UFC90100, Schnelldorf, Germany) were filled with colostrum and subjected to centrifugation for 1 h at 7500× g, 4 ◦C. This centrifugation step allowed the separation of colostrum based on molecular size. The flow-through compartment of the filters flows through compartments containing defined molecular size fractions (10, 50, and 100 kDa) of colostrum. The total protein concentration in these fractions was quantified using the Bradford assay (ThermoFisher Scientific, #23236, Waltham, MA, USA). The protein content in each fraction was then used for the cell culture experiments.
- 2.3. Cells and Culture Conditions
Human Caco-2 intestinal epithelial cells (ATCC HTB-37) were obtained from American Type Culture Collection (ATCC) (https://www.atcc.org/products/htb-37, accessed on
- 9 September 2022) were cultured in DMEM/F12 supplemented with GlutaMAX (Gibco, #10565018, ThermoFisher Scientific, Waltham, MA, USA), PenStrep (Gibco, #10378016, ThermoFisher Scientific, Waltham, MA, USA), and 10% heat-inactivated FBS (Gibco, #A3160501, ThermoFisher Scientific, Waltham, MA, USA). Human THP-1 monocytes (ATCC TIB-202) were obtained from the ATCC (https://www.atcc.org/products/tib-202, accessed on 9
September 2022) and maintained in complete RPMI media (Gibco, #11875101, ThermoFisher Scientific, Waltham, MA, USA) supplemented with GlutaMAX (Gibco, #35050061, ThermoFisher Scientific, Waltham, MA, USA) and 10% heat-inactivated FBS (Gibco, #A3160501, ThermoFisher Scientific, Waltham, MA, USA). Both cell lines were cultured at 37 ◦C with 5% CO2. When differentiation of THP-1 cells into macrophages was required, the cells were incubated in complete RPMI media containing 200 ng/mL of Phorbol 12-myristate 13-acetate (PMA) (Sigma-Aldrich, #P8139, Schnelldorf, Germany) for 48 h. Subsequently, the cells were rested in PMA-free RPMI media for an additional 48 h before being used in experiments.
- 2.4. Establishment of Caco-2/THP-1 Co-Culture System
- 2.5. Stimulation of Caco-2/THP-1 Co-Culture with Colostrum
- 2.6. LDH Release Assay
- 2.7. Immunoblot of NFκB Phosphorylation In Vitro
The THP-1 derived macrophages or Caco-2/THP-1 co-culture was treated with bovine colostrum and stimulated with PMA as described above. After the treatments, the cells were rinsed three times with ice-cold DPBS (Gibco, #10010023, ThermoFisher Scientific, Waltham, MA, USA) containing 1 mM sodium orthovanadate (NEB, # P0758S, New Engalnd Biolabs,
Ipswich, England). The membrane from the transwell inserts was cut using forceps and placed in 300 µL of 1× cell lysis buffer (Invitrogen, # FNN0011, ThermoFisher Scientific, Waltham, MA, USA) for 10 min on ice. The membranes were sonicated, and samples were centrifuged at 5000× g for 10 min at 4 ◦C. The proteins were quantified using the Pierce BCA protein assay kit (ThermoFisher Scientific, #23227, Waltham, MA, USA) as described by the manufacturer. The protein samples were incubated at 70 ◦C in 1× NuPAGE LDS loading dye (Invitrogen, # NP0008, ThermoFisher Scientific, Waltham, MA, USA) with 1× Bolt Reducing agent (Invitrogen, # B0009, ThermoFisher Scientific, Waltham, MA, USA) for
- 10 min. The lysates were separated by SDS-PAGE, electrotransferred onto PVDF membrane, and blocked using Intercept TSB blocking buffer (Licor, #927-60001, LI-COR Biosciences, Lincoln, USA). Membranes were probed with primary rabbit anti-NF-κB p65 (D14E12) (Cell Signaling, #8242, ThermoFisher Scientific, Waltham, MA, USA) or rabbit-phosphor NF-κB p65 (93H1) (Ser 536) (Cell Signaling, #3033) antibodies, and mouse β-Actin (8H10D10) (Cell Signaling, #3700, ThermoFisher Scientific, Waltham, MA, USA). Membranes were then probed using secondary IRDye® 800CW Goat anti-Rabbit IgG Secondary Antibody (Licor, #926-32211, LI-COR Biosciences, Lincoln, USA) for NF-κB or IRDye® 800CW Goat antiMouse IgG (Licor, #926-32210) for β-Actin. Blots were visualized using the Licor system.
- 2.8. Fecal Microbiome Transplantation and Generation of Calf-FMT C57BL/6 Mice
- 2.9. Depletion of C56BL/6 Mice Intestinal Microbiota
- 2.10. Calve Fecal Microbiota Transplant (FMT) Preparation
Fecal material was collected from randomly selected 7-day-old 3 male and 3 female calves, with a total of 6 calves included in the study. The calves were inspected for diarrhea and health status by a veterinary doctor. The collected fecal samples were kept refrigerated on ice during transportation to the laboratory to maintain sample integrity.
Upon arrival at the laboratory, the individual fecal samples obtained from different animals were combined and pooled together. The pooled fecal sample was then diluted in a preservation media (Brain-Heart Infusion broth (Sigma-Aldrich, Taufkirchen, Germany), 20% skim milk, 1.25 g L-cysteine (Sigma-Aldrich, Taufkirchen, Germany) preservation media medium) to achieve a concentration of 1 g/mL of FMT. To ensure homogeneity,
the fecal sample was homogenized using the Stomacher 450 Bam homogenization system (Thomas Scientific, #1185V52) for 1 min at room temperature.
Following homogenization, the fecal microbial transplant (FMT) was divided into aliquots and transferred to sterile polypropylene screw-cap tubes (JSHD, Jiangsu, China). These tubes were then stored at −80 ◦C until the experiment to preserve the viability and stability of the microbial content within the FMT samples.
- 2.11. Calve-FMT Transplantation to C57BL/6 Mice
- 2.12. Calf-FMT C57BL/6 Mice Feeding Studies
- 2.13. Lethal Challenge with S. typhimurium ATCC 14028 Strain
The Salmonella typhimurium ATCC 14028 (S. typhimurium ATCC 14028) strain was cultured on Tryptic-Soy agar (Sigma-Aldrich, Schnelldorf, Germany) plates overnight to obtain single colonies. The bacterial mass was then suspended in sterile saline, and the inoculum size was quantified spectrophotometrically to achieve approximately 1.5 × 108 CFU/mL. Each animal received a volume of 200 µL of the S. typhimurium ATCC 14028 suspension, resulting in an infectious dose of approximately 3.0 × 107 CFU/animal. The accuracy of the inoculum size was confirmed by performing serial dilutions and plating on Tryptic-Soy agar (Sigma-Aldrich, Schnelldorf, Germany) plates (BD Difco, #236920, ThermoFisher Scientific, Waltham, MA, USA).
After 24 h after infection, the calf-FMT C57BL/6 mice received either colostrum (20 mL/kg) or normal saline (20 mL/kg) at regular intervals of 8 h twice a day for 5 days. The dosage of COL or NS was selected based on the maximum tolerable quantity that could be delivered to C57BL/6 mice via oral gavage. In the survival studies, the administration of colostrum or normal saline continued until the experimental endpoint was reached, which was defined as a 20% loss of body weight or a morbid appearance. The animals were euthanized by cervical dislocation. Blood samples were collected via cardiac puncture, and the blood serum was prepared by centrifugation. The bacterial burden, expressed as log10 CFU/mL, was determined by performing serial dilutions and plating on MacConkey agar plates (BD Difco, #212123, ThermoFisher Scientific, Waltham, MA, USA). Additionally,
the fecal burden of S. typhimurium ATCC 14028 was calculated by serially diluting fecal material collected from the cecum and plating it on Xylose Lysine Deoxycholate agar (XLD) (Biomerieux, # CM0469B, ThermoFisher Scientific, Waltham, MA, USA) to differentiate from fecal flora. Sections of the intestine were collected for histological analysis.
- 2.14. Cytokine Quantification by ELISA
- 2.15. Evans Blue Intestinal Permeability Assay
- 2.16. Histological Analysis
The calf-FMT C57BL/6 mouse intestines were processed by using the Swiss roll technique as described by [27]. Right after euthanasia, the intestines were dissected and placed in ice-cold PBS (Gibco, # 10010031). The cecum was dissected and discarded, and the small intestine was flushed by ice-cold PBS, following cold modified Bouin fixative (5% acetic acid, 50% ethanol in deionized water) [23,27]. The intestinal rolls were then formed
- as described by Grigaleviciute et al., and tissues were placed in tissue processing cassettes, fixed overnight in 10% neutral formaldehyde (Fisher Scientific, # SF98-4, Waltham, MA, USA), processed and embedded by an automatic tissue processor. Tissue sections were
then stained with Hematoxylin-Eosin (HE) stain to characterize tissue architecture and Alcian blue (AB) staining to visualize mucins [23]. Tissue sections were then evaluated, and the inflammation as well as tissue architecture was scored by a pathologist using the method described by Erben et al. [28].
- 2.17. Statistical Analysis
The collected experimental data were analyzed using GraphPad Prism (GraphPad Software 8.0.1, San Diego, CA, USA). The in vitro cell culture experiments consisted of
- at least three experimental replicas, and data are shown as mean ± standard deviation (SD). The statistical significance was analyzed Mann–Whitney U test, where the mean of experimental values was compared with the mean of control values-tests. Data were considered significant when p < 0.05.
3. Results
- 3.1. Effects of Bovine Colostrum on PMA-Induced Cytokine Production and Caco-2/THP-1-Derived Macrophage Co-Culture Model
To investigate the immunomodulatory effect of COL on cytokine production in vitro, we utilized a well-established, polarized Caco-2/THP-1 macrophage co-culture model. The cells were stimulated with phorbol 12-myristate 13-acetate (PMA) that acted as broadspectrum mitogen inducing various cytokines. Initially, the Caco-2/THP-1 system was pretreated with COL at a fixed concentration of 250 µg/mL for 1 h. Subsequently, PMA was added at a concentration of 10 µg/mL in the presence of COL, and cell cultures were then incubated for 24 h to induce cytokine secretion to the culture media without noticeable phenotype change in cells (Figure 1A).
Our results demonstrated that PMA stimulation significantly increased the production of cytokines, such as IL-1b, IL-6, IL-8, IL-12, TNFα, and INFγ, compared to the unstimulated control (Figure 1B). However, pretreatment of the Caco-2/THP-1 cells with COL resulted in a significant decrease in IL-6 production compared to the cells treated with PMA alone (p < 0.05). It suggests that COL exerts an inhibitory effect on IL-6 production in response to PMA stimulation (Figure 1B). Furthermore, our study revealed that pretreatment with COL prior to PMA stimulation enhanced the production of IL-10 compared to both the control group and the cells stimulated with PMA alone. These findings indicate that COL has the ability to augment the production of IL-10, an anti-inflammatory cytokine (Figure 1B).
In addition to assessing cytokine production, we evaluated the effects of COL on Caco2/THP-1 viability by measuring the release of lactate dehydrogenase (LDH) (Figure 1C). Interestingly, we observed that pretreatment with COL prior to PMA stimulation significantly reduced the release of LDH compared to PMA alone. Moreover, PMA stimulation alone or in combination with COL pretreatment reduced the transepithelial electrical resistance (TEER), indicating increased permeability of the Caco-2/THP-1 epithelium (Figure 1C). However, COL pretreatment failed to significantly restore the loss of TEER induced by PMA.
After demonstrating the immunomodulatory activity of COL on IL-6 production using a PMA-stimulated Caco-2/THP-1 cell culture model, we aimed to characterize the types of colostrum constituents responsible for IL-6 inhibition COL (Figure 1D). To do so, we performed a series of experiments where we used a heat inactivation or ultrafiltration, to prepare different types of colostrum for the cell culture experiments. Our findings indicated that the IL-6 suppressive effect of COL is influenced by temperature-sensitive factors. Heat inactivation significantly attenuated the colostrum’s ability to inhibit IL-6 production in the Caco-2/THP-1 cell culture model, suggesting the involvement of heat-sensitive components (Figure 1D). To further investigate the factors responsible for the observed IL-6 suppression, we fractionated COL based on molecular size. The fractions with molecular weights of 10 kDa and 50 kDa did not show significant IL-6 inhibitory activity. However, the 100 kDa fraction of COL restored the IL-6 suppressive effect, indicating the presence of immunomodulatory factors with a higher molecular mass (Figure 1D).
Vet. Sci. 2023, 10, 519 epithelium (Figure 1C). However, COL pretreatment failed to significantly restore the loss10 of 20 of TEER induced by PMA.
- Figure 1. Bovine colostrum exerts immunomodulatory and chemoprotective activity on the Caco1/THP-1 macrophage co-culture system. Panel (A) shows the morphological changes of Caco-2, THP-1, and co-cultures during the stimulation with Phorbol 12-myristate 13-acetate (PMA) and treatment with bovine colostrum (COL). Scale bar represents 100 µm. Panel (B) shows the effects of bovine colostrum on PMA-induced cytokine production by Caco-2/THP-1 macrophage co-culture. Panel (C) shows the effects of bovine colostrum on PMA-induced LDH release and transepithelial electric resistance (TEER) changes. Panel (D) demonstrates the effect of various treatments of bovine colostrum on the ability to suppress IL-6 production by Caco-2/THP-1 cells. Data are shown as mean ± standard deviation (SD). ns—non-significant.
Figure 1. Bovine colostrum exerts immunomodulatory and chemoprotective activity on the Caco1/THP-1 macrophage co-culture system. Panel (A) shows the morphological changes of Caco-2, THP-1, and co-cultures during the stimulation with Phorbol 12-myristate 13-acetate (PMA) and treatment with bovine colostrum (COL). Scale bar represents 100 µm. Panel (B) shows the effects of bovine colostrum on PMA-induced cytokine production by Caco-2/THP-1 macrophage co-culture. Panel (C) shows the effects of bovine colostrum on PMA-induced LDH release and transepithelial electric resistance (TEER) changes. Panel (D) demonstrates the effect of various treatments of bovine colostrum on the ability to suppress IL-6 production by Caco-2/THP-1 cells. Data are shown as mean ± standard deviation (SD). ns—non-significant.
After demonstrating the immunomodulatory activity of COL on IL-6 production using a PMA-stimulated Caco-2/THP-1 cell culture model, we aimed to characterize the types of colostrum constituents responsible for IL-6 inhibition COL (Figure 1D). To do so, we performed a series of experiments where we used a heat inactivation or ultrafiltration, to prepare different types of colostrum for the cell culture experiments. Our findings indicated that the IL-6 suppressive effect of COL is influenced by temperature-sensitive factors. Heat inactivation significantly attenuated the colostrum’s ability to inhibit IL-6 production in the Caco-2/THP-1 cell culture model, suggesting the involvement of heatsensitive components (Figure 1D). To further investigate the factors responsible for the observed IL-6 suppression, we fractionated COL based on molecular size. The fractions with molecular weights of 10 kDa and 50 kDa did not show significant IL-6 inhibitory activity. However, the 100 kDa fraction of COL restored the IL-6 suppressive effect, indicating the presence of immunomodulatory factors with a higher molecular mass (Figure 1D).
These data suggest that COL possesses chemoprotective and immunomodulatory activity by suppressing the PMA-induced IL-6 production and cell death by Caco-2/THP-1 derived macrophage model.
3.2. Bovine Colostrum Is Able to Suppress PMA-Induced IL-6 Production in THP-1-Derived Macrophages via NF-κB-Dependent and Independent Manner
After demonstrating the suppressive effect of COL on IL-6 production and its chemoprotective activity, our next objective was to identify whether Caco-2 epithelial cells or THP-1 derived macrophages are mediating the IL-6 supresory phenotype in response to COL. To investigate this, we individually pre-treated Caco-2 cells and THP-1 macrophages with colostrum and performed co-culture experiments, followed by stimulation with PMA to induce cytokine production, and compared the findings to the co-culture experiment.
Interestingly, we observed that COL did not significantly decrease IL-6 production in Caco-2 intestinal epithelial cells alone. However, when THP-1 macrophages treated with COL were stimulated with PMA, a significant reduction in IL-6 was observed. Similar results were obtained when cells in the co-culture system were treated with colostrum and then stimulated with PMA (Figure 2). These findings suggest that the IL-6 suppressive
with COL were stimulated with PMA, a significant reduction in IL-6 was observed. Similar results were obtained when cells in the co-culture system were treated with
suppressive activity of colostrum may be primarily mediated through the THP-1 macrophage axis rather than the Caco-2 epithelial cells (Figure 2A).
activity of colostrum may be primarily mediated through the THP-1 macrophage axis rather than the Caco-2 epithelial cells (Figure 2A).
- Figure 2. Bovine colostrum is able to reduce IL-6 production via THP-1-derived macrophage modulation. Panel (A) shows the effects of COL treatment on PMA-induced IL-6 production by Caco-2, THP-1 derived macrophages or their co-cultures. Panel (B) shows Western blots for NF-κB from THP1-derived macrophages and Caco-2 co-culture. Data are shown as mean ± standard deviation (SD).
- 3.3. Colostrum Reduces the Intestinal Permeability in Calf-FMT Mice
Figure 2. Bovine colostrum is able to reduce IL-6 production via THP-1-derived macrophage modulation. Panel (A) shows the effects of COL treatment on PMA-induced IL-6 production by Caco-2, THP-1 derived macrophages or their co-cultures. Panel (B) shows Western blots for NF-κB from
(SD).
To further support our observations, we investigated the activation of the NF-κB signaling cascade by measuring the phosphorylation of p65 at Ser536 in THP-1 macrophages and the Caco-2/THP-1 macrophage co-culture after pre-treatment with colostrum followed by PMA stimulation. PMA stimulation, both in monoculture and co-
It is widely accepted that the interplay between gut microbiota and the host plays a crucial role in mucosal immune responses and intestinal homeostasis. We first sought to investigate further the histological changes and intestinal inflammation associated with colostrum feeding in calf-FMT transplanted C57BL6 mice. Due to a lack of bovine experimental models and well-established murine models, we conducted experiments
using calf fecal microbiota transplantation (FMT) in conventional C57BL/6 mice (calf-FMT C57BL/6).
The C57BL/6 mice were initially treated with a combination of antibiotics and antifungal drugs, followed by PEG 4000 cleansing to deplete the intestinal microbiota. The calf FMT was then engrafted into the conditioned mice via oral gavage. The animals were divided into two groups: one receiving colostrum (COL) (20 mL/kg, q8 h two times a day) and the other receiving normal saline (20 mL/kg, q8 h two times a day) for 5 days. To quantify the feeding-associated changes, we performed experiments using COL and normal saline (NS) feeding and employed the Evans blue assay to assess tissue permeability, which indicates cellular junction integrity. Our findings revealed that COL significantly (p = 0.0395) reduced the accumulation of Evans blue as a marker of intestinal permeability in the jejunum and ileum of the calf-FMT C57BL/6 mouse model, as compared to the untreated control (UC) (49.2 and 62.3 µg/g of tissue) or NS feeding (31.2 and 82.1 µg/g of tissue) (Figure 3A). Moreover, feeding with COL was associated with a lower pathological score of inflammation (2 severity score) and noticeable changes in tissue architecture (Figure 3C). Specifically, mice that received colostrum exhibited reduced numbers of tissueinfiltrating neutrophils and macrophages, as compared to those in the UC or NS groups, while mucus production and the localization of Goblet cells were similar (Figure 3B).
, x FOR PEER REVIEW 13 of 21
Figure 3. Bovine colostrum decreases intestinal permeability and infiltration with immune cells in calfFMT C57BL/6 mice. Panel (A) demonstrates the intestinal permeability and inflammation measured by Evans blue assay. Panel (B) shows representative photomicrographs of the mouse intestine stained with hematoxylin/eosin (HE) and Alcian blue (AB). Panel (C) shows the pathological scoring of calf-FMT C57BL6 mice undergoing feeding with normal saline (NS) and colostrum (COL). Data are shown as mean ± standard deviation (SD).
Figure 3. Bovine colostrum decreases intestinal permeability and infiltration with immune cells in calf-FMT C57BL/6 mice. Panel (A) demonstrates the intestinal permeability and inflammation measured by Evans blue assay. Panel (B) shows representative photomicrographs of the mouse intestine stained with hematoxylin/eosin (HE) and Alcian blue (AB). Panel (C) shows the pathological scoring of calf-FMT C57BL6 mice undergoing feeding with normal saline (NS) and colostrum (COL). Data are shown as mean ± standard deviation (SD).
Additionally, when the mice were challenged with S. typhimurium, a significant (p = 0.0113) reduction in Evans blue accumulation was observed in the ileum of COLtreated animals in comparison to the UC or NS groups. However, no significant differences were observed in the jejunum, suggesting that the anti-inflammatory and modulatory effects of COL are primarily directed in the ileum. Furthermore, the administration of COL
- Vet. Sci. 2023, 10, 519 13 of 20
- Figure 3. Bovine colostrum decreases intestinal permeability and infiltration with immune cells in
- Figure 4. Bovine colostrum reduces intestinal inflammation in calf-FMT C57BL/6 mice lethally challenged with S. typhimurium ATCC 14028 strain. Panel (A) demonstrates the intestinal permeability and inflammation measured by Evans blue assay followed by lethal challenge with S. typhimurium ATCC 14028. Panel (B) shows representative photomicrographs of S. typhimurium infected calf-FMT C57BL/6 mouse intestine stained with hematoxilin/eosin (HE) and Alcian blue (AB). Panel (C) shows the pathological scoring of Salmonella-infected calf-FMT C57BL6 mice undergoing feeding with normal saline (NS) and colostrum (COL). Data are shown as mean ± standard deviation (SD).
in S. typhimurium-infected mice resulted in a decreased histological score of inflammation and an enhanced epithelial changes score, suggesting possible pathogen-mediated tissue damage (Figure 4A–C).
Collectively, these data suggest that COL modulates the infiltration of immune cells into the gut environment as well as reduces the inflammation-mediated damage during lethal S. typhimurium challenge.
3.4. Colostrum Exerts Immunomodulatory Activity in Calf FMT-Transplanted Mice
Surprisingly, the mice fed with COL exhibited significantly decreased production of IL6 (42.3 pg/mL) compared to the untreated control (UC) or the group receiving normal saline (NS) (Figure 5A). Additionally, we observed an enhanced production of IL-10, indicating that colostrum can modulate the production of cytokines in our calf-FMT C57BL/6 mice (Figure 5A). Building upon this observation, we investigated whether colostrum feeding could modulate inflammatory immune responses in the gut during the challenge with S. typhimurium ATCC 14028 (Figure 5B).
To examine this, we infected calf-FMT C57BL/6 mice with S. typhimurium ATCC 14028 12 h before administration of colostrum. The administration of colostrum (COL) (20 mL/kg, q8 h two times a day) or normal saline (NS) (20 mL/kg, q8 h two times a day) was continued during the experiment for 5 days. We found that colostrum administration during S. typhimurium infection resulted in reduced production of IL-6 and INFγ compared to the untreated control as well as increased production of IL-10 (Figure 5B). However, when a survival study was conducted, the colostrum-treated animals showed decreased
Surprisingly, the mice fed with COL exhibited significantly decreased production of IL-6 (42.3 pg/mL) compared to the untreated control (UC) or the group receiving normal
- Vet. Sci. 2023, 10, 519 14 of 20
indicating that colostrum can modulate the production of cytokines in our calf-FMT C57BL/6 mice (Figure 5A). Building upon this observation, we investigated whether colostrum feeding could modulate inflammatory immune responses in the gut during the challenge with S. typhimurium ATCC 14028 (Figure 5B).
survival percentage during the lethal challenge, suggesting an altered intestinal immune defense against S. typhimurium and subsequent systemic dissemination (Figure 5C). To confirm this hypothesis, we measured the bacterial burden in the feces (cecum), blood, and spleen (Figure 5D).
Figure 5. Bovine colostrum exhibits IL-6-targeted anti-inflammatory effects and mitigates S. typhimurium ATCC 14028 infection in calf-FMT C57BL/6 mice. Panel (A) illustrates cytokine production in calf-FMT C57BL/6 mice subjected to colostrum feeding. Panel (B) displays cytokine production in response to a lethal challenge with the S. typhimurium ATCC 14028 strain. Panel (C) presents survival data of calf-FMT C57BL/6 mice following a lethal challenge with S. typhimurium ATCC 14028, with interventions involving normal saline (NS), colostrum (COL), untreated Salmonella-challenged control, or calf-FMT transplanted and non-infected mice. Panel (D) demonstrates the Salmonella bacterial burden subsequent to the lethal challenge and feeding with NS or COL. Data are shown as mean ± standard deviation (SD). * p < 0.05; ** p < 0.001.
Figure 5. Bovine colostrum exhibits IL-6-targeted anti-inflammatory effects and mitigates S. typhimurium ATCC 14028 infection in calf-FMT C57BL/6 mice. Panel (A) illustrates cytokine production in calf-FMT C57BL/6 mice subjected to colostrum feeding. Panel (B) displays cytokine production in response to a lethal challenge with the S. typhimurium ATCC 14028 strain. Panel (C) presents survival data of calf-FMT C57BL/6 mice following a lethal challenge with S. typhimurium ATCC 14028, with interventions involving normal saline (NS), colostrum (COL), untreated Salmonella-challenged control, or calf-FMT transplanted and non-infected mice. Panel (D) demonstrates the Salmonella bacterial burden subsequent to the lethal challenge and feeding with NS or COL. Data are shown as mean ± standard deviation (SD). * p < 0.05; ** p < 0.001.
Notably, there were no significant changes in cecal S. typhimurium burden between infected and treated animals, indicating that COL or NS feeding was not associated with increased bacterial proliferation in the cecum (Figure 5D). Conversely, when assessing systemic dissemination, the bacterial burden in the blood was significantly higher in COLtreated animals (3.4 log10 CFU/mL) compared to the UC (2.3 log10 CFU/mL) or NS group (2.7 log10 CFU/mL). Similar results were observed when quantifying S. typhimurium in the spleen of COL-treated animals (5.5 log10 CFU/mL), suggesting that colostrum feeding provides immunomodulatory activity in the calf-FMT-transplanted C57BL/6 mice by modulating IL-6 and IL-10 responses (Figure 5D).
14028 12 h before administration of colostrum. The administration of colostrum (COL) (20 mL/kg, q8 h two times a day) or normal saline (NS) (20 mL/kg, q8 h two times a day) was continued during the experiment for 5 days. We found that colostrum administration during S. typhimurium infection resulted in reduced production of IL-6 and INFγ
4. Discussion
Colostrum (COL), as the initial milk produced by cows after parturition, plays a pivotal role in the systemic and gut immune development of neonate calves, contributing to their enhanced survival and optimal growth during this critical period [29,30]. The rich composition of biologically active substances in COL, including vital nutrients, growth factors, and immunoglobulins, orchestrates the maturation of the intestinal mucosa and shapes the immunological milieu at both local and systemic levels [30]. Notably, COL
has been found to influence the microbiome of calves, suggesting a potential interplay between colostrum, the microbiome, and mucosal immune responses that contribute to intestinal homeostasis, tolerance, and protection against diarrheal diseases [31]. Despite the well-documented observations regarding the beneficial effects of COL on calf health and development, there remains a significant knowledge gap concerning the specific molecular pathways regulated by colostrum that underlie its beneficial effects on the intestinal environment and innate immune responses [32–34].
Intestinal inflammation and diarrhea are prevalent and severe conditions in neonate calves, contributing to high morbidity and mortality rates [35]. Various infectious and non-infectious factors can trigger diarrhea, leading to intestinal and systemic inflammation, compromised intestinal barrier integrity, dysbiosis of the intestinal microbiota, impaired growth patterns, and increased mortality [36,37]. The dysregulated production of pro-inflammatory cytokines in the gut is responsible for initiating and perpetuating the inflammatory processes within the gut leading to systemic inflammation and potential autoimmune disorders [38–40]. A study conducted by Fisher et al. highlighted the critical role of interleukin-6 (IL-6) in bovine diarrhetic diseases, suggesting its potential as a prognostic marker [41]. In addition to IL-6, interleukin-1β (IL-1β) and tumor necrosis factor-alpha (TNFα) have been implicated in the acute phase responses observed in neonate calves, underscoring the central role of these cytokines in intestinal inflammation [39].
However, due to the limited availability of molecular reagents and established bovine cell lines and intestinal models, it is difficult to accurately recapitulate the bovine intestinal environment in vitro. Therefore, we employed a well-established Caco-2/THP-1 intestinal model to investigate the innate immune responses to bovine colostrum [42]. To induce inflammatory processes, we utilized phorbol 12-myristate 13-acetate (PMA), a non-specific mitogen that acts through protein kinase C (PKC), nuclear factor kappa-lightchain-enhancer of activated B cells (NF-κB), and mito-gen-activated protein kinase (MAPK) signaling pathways, ultimately resulting in the production of pro-inflammatory cytokines across various cell lines, including Caco-2/THP-1 co-culture [30,43,44].
In our experimental in vitro model, pretreatment with COL before PMA stimulation resulted in a decreased production of the pro-inflammatory cytokine IL-6. Interestingly, we observed that COL also exhibited immunomodulatory properties by inducing the production of the anti-inflammatory cytokine IL-10 in response to PMA stimulation, suggesting its potential to regulate the inflammatory response in Caco-2/THP-1 cells. Furthermore, colostrum demonstrated the ability to restore cellular viability by reducing lactate dehydrogenase (LDH) production induced by PMA, indicating its potential cytoprotective effects [45].
Early investigations have provided valuable insights into the potential immunomodulatory activity of COL. For instance, Biswas et al. demonstrated that exposure of human peripheral blood mononuclear cells (PBMCs) to bovine colostrum led to the polarization of immune responses towards Th1 responses [46]. This suggests that COL has the ability to modulate the balance of immune cell subsets and their associated cytokine profiles. A comprehensive review conducted by Menchetti highlighted the diverse activities of COL on human innate and adaptive immune responses, indicating its potential therapeutic applications. Colostrum contains high quantities of maternal immunoglobulins, cytokines, growth factors, and immunomodulatory milk oligosaccharides [47]. These components have been shown to regulate intestinal mucosal responses through interactions with cytokine and growth factor receptors, as well as with various pathogen-associated molecular patterns (PAMPs) or damage-associated molecular patterns (DAMPs) [48]. This suggests that COL can exert its immunomodulatory effects by influencing the signaling pathways involved in immune cell activation and regulation. The presence of maternal immunoglobulins in colostrum provides passive immunity to newborns, offering protection against various pathogens [1,49,50]. As non-protein-derived constituents, immunomodulatory milk oligosaccharides found in colostrum have been shown to interact with the gut microbiota,
influencing its composition and function, which in turn can impact immune responses and overall gut health [51].
In our study, we observed that the ability of COL to suppress PMA-induced IL-6 production is influenced by heat, indicating a temperature sensitivity, which supports the hypothesis of the possible involvement of proteinaceous matter. Furthermore, our subsequent experiments involving ultrafiltration demonstrated that the constituents of COL, capable of passing through 10 kDa molecular cutoff filters, possess the ability to mediate this activity. In contrast to the findings reported by Ann et al., who demonstrated that the inhibition of pro-inflammatory cytokine expression by colostrum occurs through the regulation of epithelial NF-κB, our study reveals that the production of IL-6 induced by PMA in a co-cultured Caco-2 and THP-1 cells probably does not fully rely on the activation of NF-κB [52].
Intestinal inflammation often leads to impaired intestinal barrier function and increased gut permeability, which can exacerbate immune responses, disrupt the gut microbiota, and potentially result in systemic infections and mortality, particularly in neonate calves [53,54]. Additionally, colostrum is rich in beneficial probiotic bacteria, such as Lactobacillus and Bifidobacterium, which have been shown to, directly and indirectly, impact intestinal health [55,56]. These probiotics can modulate mucus production, maintain tight junctions between epithelial cells, and influence immunological mucosal tolerance through the IL-10/regulatory T cell axis [57]. Thus, the interplay between colostrum and the intestinal microbiota emerges as a critical factor in maintaining intestinal homeostasis.
To validate our in vitro findings regarding the IL-6/IL-10-directed immunomodulatory activity of colostrum, we utilized murine models with engrafted calf fecal microbiota to replicate the intestinal environment of calves in a well-established organism with defined immunological and histological properties. We chose the C57BL/6 background mice due to their wild-type Th1 responses, both at mucosal and systemic levels, and the similar strategy described by Wrzosek et al. [26]. To simulate acutely inflamed gastrointestinal conditions, we employed a lethal challenge with Salmonella typhimurium ATCC 14028 to investigate the effects of colostrum on the host’s innate immune responses to this intestinal pathogen [58]. As S. typhimurium induces robust Th1 inflammatory responses in the intestine, characterized by immune cell recruitment, cytokine production, and increased gut permeability, we hypothesized that the immunomodulatory and anti-inflammatory effects of COL would enhance the S. typhimurium infection, resulting in enhanced lethality due to an inability to mount pro-inflammatory cytokine responses necessary for immune cell recruitment [59].
In our FMT murine model, we observed that COL significantly reduced intestinal permeability in the duodenum, jejunum, and ileum, while having no effect on the colon, as evidenced by reduced Evans blue accumulation in calf-FMT C57BL/6 mice. Surprisingly, we demonstrated decreased intestinal infiltration of neutrophils and macrophages in colostrum-treated animals compared to untreated controls or those receiving normal saline. Following the S. typhimurium ATCC 14028 challenge, we observed decreased intestinal inflammation and leakiness in the ileum of colostrum-fed calf-FMT C57BL/6 mice, along with reduced tissue-infiltrating immune cells. These findings suggest the immunomodulatory and anti-inflammatory activity of COL in the calf-FMT C57BL/6 mouse model. Similar insights into the anti-inflammatory properties of colostrum were observed by Menchetti et al., although the exact mechanism was not proposed [47]. Furthermore, we unexpectedly observed that calf-FMT C57BL/6 mice undergoing colostrum feeding exhibited decreased IL-6 production and enhanced IL-10 and IL-12 production compared to untreated calf-FMT C57BL/6 mice or those receiving saline feeding. This finding demonstrates the in vivo relevant immunomodulatory and anti-inflammatory activity of COL. When mice were challenged with S. typhimurium, colostrum-treated mice displayed an IL-6 suppressive phenotype and increased IL-10 production. This indicates that the anti-inflammatory activity of COL is directed toward the IL-6/IL-10 axis, even in a highly inflammatory environment, as anticipated, leading to early mortality and systemic bacterial dissemi-
nation, through regulation of IL-6/IL-10 production without significant activity on other inflammatory cytokines.
Despite using multiple in vitro approaches and validating our findings in novel murine models, our study has several limitations. Firstly, the use of the Caco-2/THP-1 model cannot guarantee that similar phenotypes would be exhibited by bovine epithelium. Moreover, in vitro model using Caco-2/THP-1 cell co-culture resulted in a high variability of cytokine production, suggesting that the effects observed in this model could differ if other epithelial cells or macrophages would be used. However, due to the lack of established bovine cell lines and intestinal models, as well as the limited availability of immunological reagents, conducting comprehensive in vitro studies to evaluate the effects on bovine epithelial models would be challenging. Secondly, calves possess a unique ruminant intestinal system with potentially bovine-specific enzymatic and metabolic properties that differ significantly from rodents. Since the intestinal microbiota is known to influence mucosal immune responses, we performed bovine fecal microbiota transplantation (FMT) into mice models with wild-type responses to generate an intestinal environment similar to bovines. Lastly, we understand that our findings in cell culture models and calf-FMT C57BL/6 mice need further validation using bovine models to fully confirm the colostrum-mediated immunomodulatory effects.
5. Conclusions
Our study provided an important insight into the immunomodulatory activity of COL in the intestinal environment. The findings described in this study suggest that COL has the capacity to modulate the inflammatory response by potentially acting on the IL-6 and IL-10 axis in cell culture-based models. Furthermore, in our in vivo model using calfFMT C57BL/6 mice, COL feeding further confirmed effects observed in in vitro models, where COL feeding resulted in decreased production of IL-6 and enhanced production of IL-10 and IL-12 compared to untreated mice. Moreover, feeding with COL was able to suppress IL-6 production in S. typhimurium-infected animals, demonstrating powerful immunomodulatory activity during acute inflammation and infection. This confirms the in vivo relevance of the immunomodulatory activity of COL and its potential ability to shift the cytokine profile towards anti-inflammatory responses both in tissue culture models and mice. Taken together, our data provide valuable insights into the potential antiinflammatory applications of COL via interaction with the IL-6/IL-10 axis. Further studies are needed to fully elucidate the underlying molecular mechanisms and validate these findings in bovine models, with the long-term aim of potentially developing COL-based interventions for modulating intestinal inflammatory conditions.
Supplementary Materials: The following supporting information can be downloaded at: https:// www.mdpi.com/article/10.3390/vetsci10080519/s1, Figure S1. The full-length Western blot image demonstrating human total NF-κB and beta-actin in THP-1-derived macrophages as described in the materials and methods section of the manuscript. Figure S2. The full-length Western blot image demonstrating human total NF-κB and beta-actin in Caco-2/THP-1 co-culture as described in the materials and methods section of the manuscript. Figure S3. The full-length Western blot image demonstrates human phosphor-NF-κB (Ser536) and beta-actin in THP-1 macrophages as described in the materials and methods section of the manuscript. Figure S4. The full-length Western blot image demonstrating human phosphor-NF-κB (Ser536) and beta-actin in Caco-2/THP-1 co-culture as described in the materials and methods section of the manuscript.
Author Contributions: Conceptualization, R.G. and P.K.; methodology, P.K. and R.G.; software, R.G. and P.K.; validation, R.G. and P.K.; formal analysis, R.G. and P.K.; investigation, R.G. and P.K.; resources, R.G. and P.K.; data curation, R.G. and P.K.; writing—original draft preparation, P.K. and R.G.; writing—review and editing, R.P., E.R.-V., A.B., A.Ž., A.Z., V.Z., A.K., P.M. and R.S.; visualization, P.K. and R.G.; supervision, P.K. and P.M. All authors have read and agreed to the published version of the manuscript.
Funding: The research was partly funded by the Science Foundation of the Lithuanian University of Health Sciences (No. 119-05).
Institutional Review Board Statement: The animal study protocol was approved by the State Food and Veterinary Service (SFVS) animal care and welfare committee (approval number: No G2-164).
Informed Consent Statement: Informed consent to collect samples from farm animals were obtained from the representative of the farm. All experiments involving animal subjects were approved State Food and Veterinary Service (SFVS) animal care and welfare committee (approval number: No G2-164).
Data Availability Statement: All data generated during this study is disclosed within the manuscript. The biological samples obtained during this study are available upon reasonable request from the corresponding author.
Conflicts of Interest: The authors declare no conflict of interest.
Figures
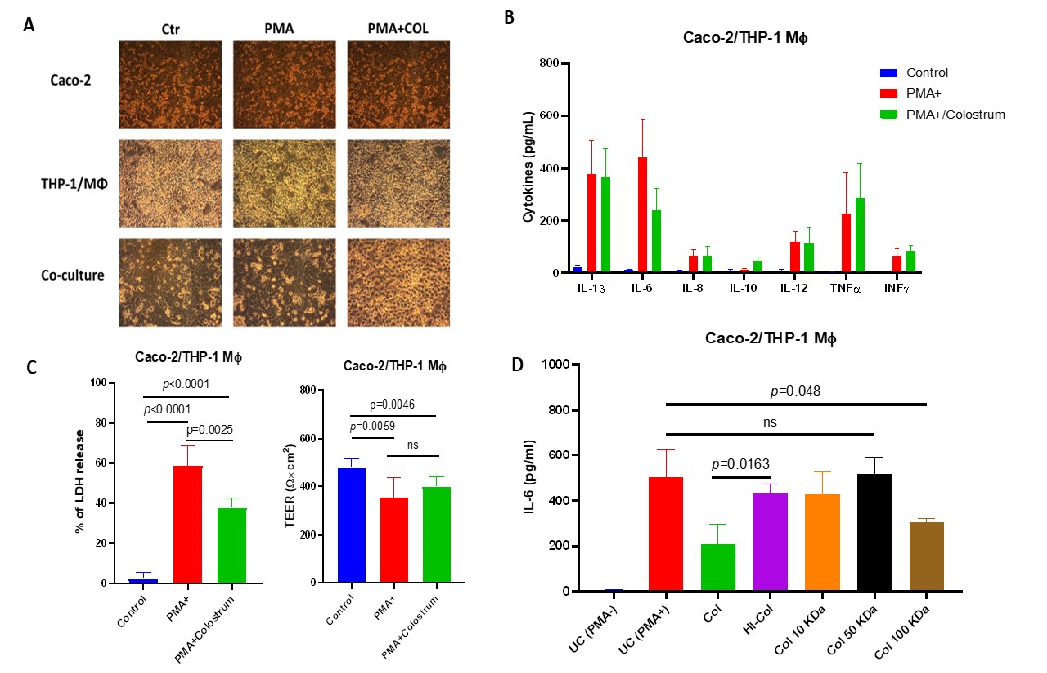
Figure 6
Extended analysis of bovine colostrum's effects on inflammatory cytokine profiles in the Caco-2/THP-1 co-culture system, demonstrating dose-dependent immunomodulatory activity.
chart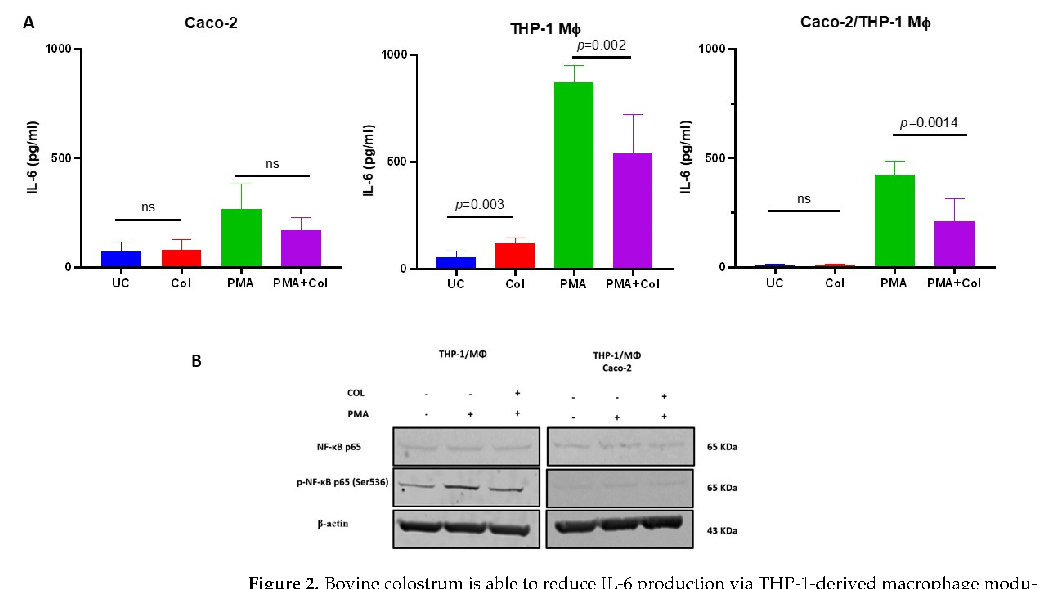
Figure 7
Supplementary immunological data showing bovine colostrum's influence on macrophage-mediated inflammation, with particular focus on the balance between pro- and anti-inflammatory responses.
chart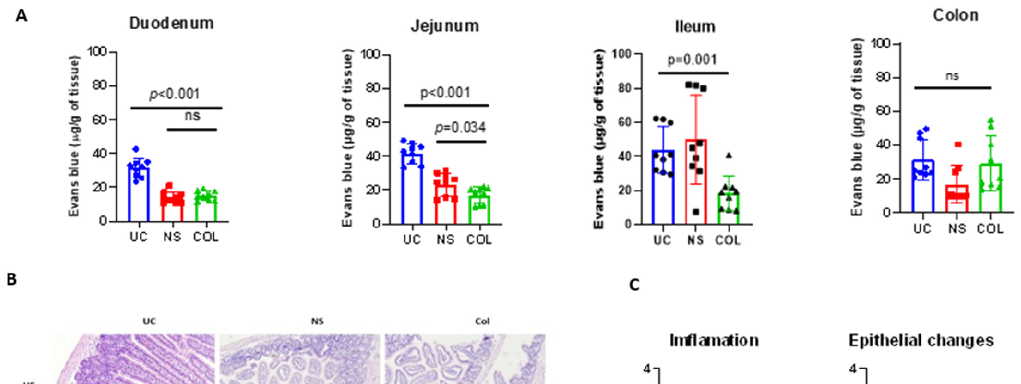
Figure 8
Additional cytokine measurement data from bovine colostrum-treated cell cultures, comparing IL-6 and IL-10 levels across different treatment conditions.
chart
Figure 9
Further in vitro data demonstrating bovine colostrum's immunomodulatory capacity, including its effects on PMA-stimulated inflammatory signaling pathways.
chart
Figure 10
Gene expression or protein analysis data related to the IL-6/IL-10 signaling axis in bovine colostrum-treated macrophage co-cultures.
chart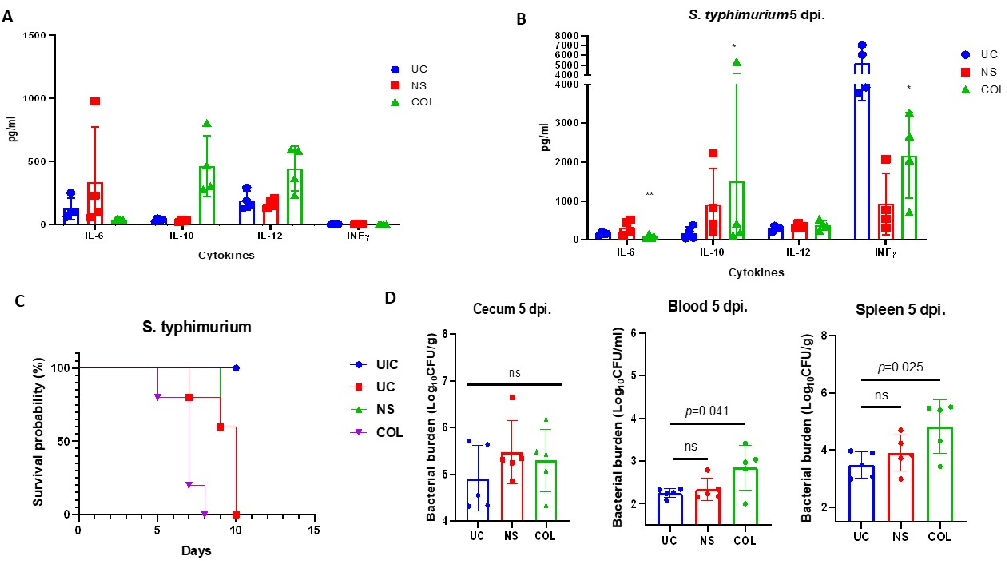
Figure 11
Supplementary experimental data from the bovine colostrum immunomodulation study, supporting the role of colostrum bioactive compounds in inflammatory control.
chartUsed In Evidence Reviews
Similar Papers
Alimentary pharmacology & therapeutics · 2006
Review article: complementary and alternative therapies for inflammatory bowel disease.
American journal of physiology. Gastrointestinal and liver physiology · 2011
The nutriceutical bovine colostrum truncates the increase in gut permeability caused by heavy exercise in athletes.
European journal of applied physiology · 2017
Intestinal fatty acid-binding protein and gut permeability responses to exercise.
American journal of physiology. Gastrointestinal and liver physiology · 2015
Early gradual feeding with bovine colostrum improves gut function and NEC resistance relative to infant formula in preterm pigs.
Scandinavian journal of gastroenterology · 2011
Randomized control trials using a tablet formulation of hyperimmune bovine colostrum to prevent diarrhea caused by enterotoxigenic Escherichia coli in volunteers.
The American journal of clinical nutrition · 2016