Gut Microbial Metabolite Butyrate and Its Therapeutic Role in Inflammatory Bowel Disease: A Literature Review.
Study Design
- 研究タイプ
- Review
- 対象集団
- IBD patients (review)
- 介入
- Gut Microbial Metabolite Butyrate and Its Therapeutic Role in Inflammatory Bowel Disease: A Literature Review. Butyrate
- 比較対照
- None
- 主要アウトカム
- Intestinal inflammation and barrier function
- 効果の方向
- Positive
- バイアスリスク
- Unclear
Abstract
Background and objective: Inflammatory bowel disease (IBD), including Crohn's disease and ulcerative colitis, is a chronic inflammatory disorder characterized by aberrant immune responses and compromised barrier function in the gastrointestinal tract. IBD is associated with altered gut microbiota and their metabolites in the colon. Butyrate, a gut microbial metabolite, plays a crucial role in regulating immune function, epithelial barrier function, and intestinal homeostasis. In this review, we aim to present an overview of butyrate synthesis and metabolism and the mechanism of action of butyrate in maintaining intestinal homeostasis and to discuss the therapeutic implications of butyrate in IBD. Methods: We searched the literature up to March 2023 through PubMed, Web of Science, and other sources using search terms such as butyrate, inflammation, IBD, Crohn's disease, and ulcerative colitis. Clinical studies in patients and preclinical studies in rodent models of IBD were included in the summary of the therapeutic implications of butyrate. Results: Research in the last two decades has shown the beneficial effects of butyrate on gut immune function and epithelial barrier function. Most of the preclinical and clinical studies have shown the positive effect of butyrate oral supplements in reducing inflammation and maintaining remission in colitis animal models and IBD patients. However, butyrate enema showed mixed effects. Butyrogenic diets, including germinated barley foodstuff and oat bran, are found to increase fecal butyrate concentrations and reduce the disease activity index in both animal models and IBD patients. Conclusions: The current literature suggests that butyrate is a potential add-on therapy to reduce inflammation and maintain IBD remission. Further clinical studies are needed to determine if butyrate administration alone is an effective therapeutic treatment for IBD.
要約
The current literature suggests that butyrate is a potential add-on therapy to reduce inflammation and maintain IBD remission.
Full Text
nutrients
Review
Gut Microbial Metabolite Butyrate and Its Therapeutic Role in Inflammatory Bowel Disease: A Literature Review
Neeraja Recharla, Ramasatyaveni Geesala and Xuan-Zheng Shi *
Department of Internal Medicine, The University of Texas Medical Branch, 301 University Blvd, 4.106 Basic Science Building, Galveston, TX 77555-0655, USA; [email protected] (N.R.); [email protected] (R.G.)
* Correspondence: [email protected]
Citation: Recharla, N.; Geesala, R.; Shi, X.-Z. Gut Microbial Metabolite Butyrate and Its Therapeutic Role in Inflammatory Bowel Disease: A Literature Review. Nutrients 2023, 15,
2275. https://doi.org/10.3390/ nu15102275
Academic Editor: Rosa Casas
Copyright: © 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https:// creativecommons.org/licenses/by/ 4.0/).
Abstract: Background and objective: Inflammatory bowel disease (IBD), including Crohn’s disease and ulcerative colitis, is a chronic inflammatory disorder characterized by aberrant immune responses and compromised barrier function in the gastrointestinal tract. IBD is associated with altered gut microbiota and their metabolites in the colon. Butyrate, a gut microbial metabolite, plays a crucial role in regulating immune function, epithelial barrier function, and intestinal homeostasis. In this review, we aim to present an overview of butyrate synthesis and metabolism and the mechanism of action of butyrate in maintaining intestinal homeostasis and to discuss the therapeutic implications of butyrate in IBD. Methods: We searched the literature up to March 2023 through PubMed, Web of Science, and other sources using search terms such as butyrate, inflammation, IBD, Crohn’s disease, and ulcerative colitis. Clinical studies in patients and preclinical studies in rodent models of IBD were included in the summary of the therapeutic implications of butyrate. Results: Research in the last two decades has shown the beneficial effects of butyrate on gut immune function and epithelial barrier function. Most of the preclinical and clinical studies have shown the positive effect of butyrate oral supplements in reducing inflammation and maintaining remission in colitis animal models and IBD patients. However, butyrate enema showed mixed effects. Butyrogenic diets, including germinated barley foodstuff and oat bran, are found to increase fecal butyrate concentrations and reduce the disease activity index in both animal models and IBD patients. Conclusions: The current literature suggests that butyrate is a potential add-on therapy to reduce inflammation and maintain IBD remission. Further clinical studies are needed to determine if butyrate administration alone is an effective therapeutic treatment for IBD.
Keywords: butyrate; inflammatory bowel disease; gut microbiota; microbial metabolites; nutrients; gut homeostasis; immune responses; T-cells
1. Introduction
Inflammatory bowel disease (IBD) is characterized by chronic intestinal inflammation in the gastrointestinal (GI) tract and includes Crohn’s disease (CD) and ulcerative colitis (UC). Although both CD and UC present with chronic inflammation, they differ in many aspects such as location, distribution, and depth of inflammation, and complications, and rectal involvement (Table 1). The exact cause of IBD is still not well understood, but the pathogenesis is interlinked with genetic factors, abnormal immune reactivity, microbiota dysbiosis, diet, and environmental factors being involved. The dynamic balance between commensal microflora and host defensive responses in the intestine plays a key role in the initiation and chronic progression of IBD [1]. Disturbed immune function and epithelial barrier integrity are the major features of IBD.
Since the pathogenic mechanisms of CD and UC remain unknown, IBD is not curable. Current therapies for IBD, including corticosteroids, immunomodulators, and biologics, are designed to induce remission [2]. However, patient response to the treatments decreases over time, and relapses occur frequently. Moreover, the side effects of these treatments
Nutrients 2023, 15, 2275. https://doi.org/10.3390/nu15102275 https://www.mdpi.com/journal/nutrients
are significant, and sometimes intolerable to patients. It is important to identify novel therapeutic targets and discover effective and safe treatments for IBD patients. Shortchain fatty acids (SCFAs) are the most abundant microbial metabolites in the intestine and provide 60–70% of the energy needs for colonocytes [3]. Specifically, butyrate is the major fuel source for the epithelial cells and has gained more attention than any other SCFAs as it regulates intestinal homeostasis and maintains epithelial integrity. A reduced number of butyrate-producing bacteria and lowered butyrate concentration have been found in IBD [4,5]. As butyrate is shown to modulate immune function and intestinal barrier function, it is considered a therapeutic target in the treatment of IBD. In this review, we discuss the production and metabolism of butyrate and the therapeutic implications of butyrate in IBD.
- Table 1. Comparison of Crohn’s disease and ulcerative colitis.
Features Crohn’s Disease Ulcerative Colitis Location Any part of the GI * tract Large intestine
Inflammation Transmural Superficial Complications
Fistula development, obstruction
No fistula, Hemorrhage Distribution Discontinuous and patchy Continuous
Rectal involvement Occasional Common
* GI: gastrointestinal.
The literature for this review was searched up to March 2023 from PubMed, Web of Science, and other sources using search terms such as butyrate, inflammation, IBD, Crohn’s disease, and ulcerative colitis. Studies on human trials and animal models were included to extract data for summarizing the therapeutic implications of butyrate. The relevant studies and their reported outcomes were analyzed and discussed with references to emphasize possible mechanisms of action. This review only includes papers published in English.
2. Gut Microbiota and Metabolites
The human gastrointestinal tract harbors a complex and diverse microbial population termed gut microbiota. The gut microbiota comprises trillions of microbes, including archaea, bacteria, fungi, and viruses. Many bacteria, particularly anaerobic bacteria, colonize the intestinal tract in a symbiotic relationship which plays a critical role in maintaining the intestinal homeostasis of the host. The high-throughput DNA sequencing technology has enhanced our understanding of gut microbiota without the need for microbial culturing. More than 1000 bacterial species colonized the human gastrointestinal tract, especially in the colon. Most of these bacterial species belong to two major phyla: Firmicutes and Bacteroidetes [6].
The gut microbiota produces a wide range of metabolites, including SCFAs, polyamines, vitamins, tryptophan-derived metabolites, and secondary bile acids, using exogenous undigested dietary substrates and endogenous compounds [7]. These metabolites can be classified into three types: (1) Metabolites produced by the microbial transformation of dietary components or drugs such as compound K; (2) Metabolites produced from host secretions that are modified by gut microbiota such as secondary bile acids; and
- (3) Metabolites synthesized by gut microbiota from diet components such as SCFAs [8]. These metabolites may also serve as nutrients or substrates for other bacterial species in the colon to further produce metabolites via interspecies cross-feeding interactions [9]. The microbial metabolites can be both beneficial and toxic to the host (Figure 1).
- Figure 1. Synthesis of microbial metabolites in the intestine. Commensal bacteria in the intestine utilize nondigested fermentable carbohydrates and proteins from the host-ingested diet and produce SCFAs and vitamins. Likewise, gut bacteria transform nonabsorbed primary bile salts into secondary bile acids. These microbial metabolites modulate the host physiological functions and provide health benefits.
Figure 1. Synthesis of microbial metabolites in the intestine. Commensal bacteria in the intestine utilize nondigested fermentable carbohydrates and proteins from the host-ingested diet and produce SCFAs and vitamins. Likewise, gut bacteria transform nonabsorbed primary bile salts into secondary bile acids. These microbial metabolites modulate the host physiological functions and pro-
The primary bile acids, cholic acid, and chenodeoxycholic acids are synthesized from cholesterol and conjugated to glycine or taurine in the liver then stored in the gallbladder and released into the intestine to facilitate dietary-fats emulsification, digestion, and absorption in the small intestine. The remaining bile acids are absorbed in the terminal ileum and reached the liver through enterohepatic circulation [10]. The escaped bile salts during enterohepatic circulation become substrates for gut microbial metabolism, including deconjugation, oxidation, epimerization, and dehydroxylation. The bacteria genera including Clostridium, Bifidobacterium, Bacteroides, Listeria, and Lactobacillus are involved in the deconjugation of bile acids. Bacteroides, Eggerthella, Escherichia, Clostridium, Ruminococcus, and Peptostreptococcus are involved in oxidation and epimerization [11]. The intestinal bacteria Clostridium and Eubacterium genera transform cholic acid and chenodeoxycholic acid into deoxycholic acid and lithocholic acid, respectively, by dihydroxylation using hydroxysteroid dehydrogenase enzymes [11–13]. Undigested dietary proteins enter the colon and serve as substrate for gut-microbial metabolism. Tryptophan is an essential amino acid consumed in the diet. Undigested or escaped tryptophan is fermented by colonic bacteria, producing various metabolites, indole, indoleacetic acid, indole-3-lactate, and indole-3propionate through direct tryptophan transformation pathway [14]. Indole-producing bacteria, such as Acinetobacter oleivorans, Vibrio cholera, Escherichia coli, Pseudomonas chlororaphis, and Synbiobacterium thermophilus, produce indole from tryptophan [15].
The primary bile acids, cholic acid, and chenodeoxycholic acids are synthesized from cholesterol and conjugated to glycine or taurine in the liver then stored in the gallbladder and released into the intestine to facilitate dietary-fats emulsification, digestion, and absorption in the small intestine. The remaining bile acids are absorbed in the terminal ileum and reached the liver through enterohepatic circulation [10]. The escaped bile salts during enterohepatic circulation become substrates for gut microbial metabolism, including deconjugation, oxidation, epimerization, and dehydroxylation. The bacteria genera including Clostridium, Bifidobacterium, Bacteroides, Listeria, and Lactobacillus are involved in the deconjugation of bile acids. Bacteroides, Eggerthella, Escherichia, Clostridium, Ruminococcus and Peptostreptococcus are involved in oxidation and epimerization [11]. The intestinal bacteria Clostridium and Eubacterium genera transform cholic acid and chenodeoxycholic acid into deoxycholic acid and lithocholic acid, respectively, by dihydroxylation using hydroxysteroid dehydrogenase enzymes [11–13]. Undigested dietary proteins enter the colon and serve as substrate for gut-microbial metabolism. Tryptophan is an essential amino acid consumed in the diet. Undigested or escaped tryptophan is fermented by colonic bacteria, producing various metabolites, indole, indoleacetic acid, indole-3-lactate, and indole-3-propionate through direct tryptophan transformation pathway [14]. Indole-producing bacteria, such as Acinetobacter oleivorans, Vibrio cholera, Escherichia coli, Pseudomonas chlororaphis, and Synbiobacterium thermophilus, produce indole from tryptophan [15].
SCFAs, including acetic, propionic, and butyric acids, are a group of carboxylic acids that consist of lesser than six carbon atoms. SCFAs are derived from the fermentation of nondigestible carbohydrates in the proximal colon and by proteolytic fermentation in the distal colon. SCFAs can be formed from fermentable carbohydrates through the glycolytic pathway and the pentose phosphate pathways by microbial fermentation [16]. Butyrate is mainly produced from species of the Firmicutes phylum, including Roseburia species,
SCFAs, including acetic, propionic, and butyric acids, are a group of carboxylic acids that consist of lesser than six carbon atoms. SCFAs are derived from the fermentation of nondigestible carbohydrates in the proximal colon and by proteolytic fermentation in the distal colon. SCFAs can be formed from fermentable carbohydrates through the glycolytic pathway and the pentose phosphate pathways by microbial fermentation [16]. Butyrate is
Faecalibacterium prausnitzii, and Eubacterium rectale, whereas acetate and propionates are produced from the species of the Bacteroidetes phylum [17,18]. The production of SCFAs in the intestine is substrate dependent. About 300 to 600 mmol of SCFAs are produced in the human intestine per day and only a small amount of SCFAs (~10 mmol) are excreted through fecal excretion. The remaining SCFAs are rapidly absorbed by the host epithelial cells via passive diffusion or active transport [19,20].
3. Butyrate Production, Absorption, and Metabolism
Gut microbiota produces butyrate from acetyl-CoA, lysine, glutarate, or succinate pathways in the colon [21]. Various bacterial species in the human intestine generate enzymes that can synthesize butyrate from complex fermentable substrates. The predominant butyrogenic bacterial species, including Faecalibacterium prausnitzii, Clostridium spp., Eubacterium spp., and Roseburia spp., are from two clusters (Clostridium clusters IV and XIVa) in the Firmicutes phylum and the Clostridiales order [22,23]. Most luminal butyrate is synthesized from nondigestible carbohydrates via the acetyl-CoA pathway (Figure 2). In the first step, nondigestible carbohydrates are catabolized into pyruvate through the pentose phosphate pathway or Embden–Meyerhof–Parnas pathway. Pyruvate can be converted into acetyl-CoA, which is further broken down into butyryl-CoA. In the final step, butyryl-CoA can be converted into butyrate by butyryl-CoA: acetyl-CoA transferase or phosphorylated to butyryl-phosphate through phospho-transbutyrylase and then subsequently converted to butyrate through butyrate kinase [24–26]. Acetate is required to produce butyrate via butyryl-CoA: acetyl-CoA transferase through cross-feeding microbial reactions. Butyrate is produced by cross-feeding interactions between acetate-producing Bifidobacterium spp. and acetate-utilizing Faecalibacterium prausnitzii [23]. Moreover, the metabolite cross-feeding within the microbial community plays a key role in maintaining the diversity of the gutmicrobial ecosystem [27]. In the succinate pathway, butyrogenic bacteria convert succinate to crotonyl-CoA, which is subsequently converted into butyrate. Crotonyl-CoA is the common butyrate precursor in L-lysine and glutarate pathways (Figure 2).
Gut-microbiome-derived butyrate is taken up rapidly by colonocytes through passive nonionic diffusion or active carrier-mediated transport [28]. The ionized form of butyrate is transported across the apical surface of intestinal epithelial cells through active transport mediated by H+-monocarboxylate transporter-1 (MCT1) and Na+-coupled monocarboxylate transporter-1 (SMCT1). Solute carrier family 5 member 8 (SLC5A8) is one of the major SMCT1 transporters of butyrate across the colonocytes [29]. The gene expression levels of SLC5A8 are abundant in the apical membrane of the colon and ileum. On the basolateral membrane, butyrate is transported through the carrier-mediated bicarbonate exchange system [30]. Butyrate predominantly presents in the anionic form in the colon due to colonic luminal pH conditions. Thus, it requires carrier-mediated transportation for cellular entry.
The absorbed butyrate is metabolized in the intestinal epithelial cells, liver cells, and other tissues and cells [31]. In the epithelial cells, butyrate is transformed into acetyl-CoA and enters the tricarboxylic acid (TCA) cycle in the mitochondria to produce ATP, which is consumed by the colon epithelial cells. The portion of butyrate which is not utilized by epithelial cells can reach the liver via portal circulation, where it is metabolized into acetyl-CoA and becomes a substrate for fatty acids, cholesterol, and ketone bodies by hepatocytes [22,32]. The plasma concentration of butyrate is very low compared to colonic levels, only 2% of butyrate enters systemic circulation, being utilized by other tissues and cells [32]. The remaining SCFAs, including butyrate, are excreted through the lungs and urine.
- Figure 2. Schematic representation of pathways involved in butyrate production, absorption, and metabolism. Butyrate is synthesized by intestinal bacteria via four pathways from nondigestible carbohydrates, succinate, L-lysate, and glutarate. It is taken-up and metabolized by the colonic epithelial cells. Low levels of butyrate enter into the liver and regulate fatty acid metabolism. Small amounts of butyrate enter into the systemic circulation and may reach other tissues.
Figure 2. Schematic representation of pathways involved in butyrate production, absorption, and metabolism. Butyrate is synthesized by intestinal bacteria via four pathways from nondigestible carbohydrates, succinate, L-lysate, and glutarate. It is taken-up and metabolized by the colonic epithelial cells. Low levels of butyrate enter into the liver and regulate fatty acid metabolism. Small amounts of butyrate enter into the systemic circulation and may reach other tissues.
4. Role and Mechanisms of Butyrate in the Regulation of Barrier Function andImmune Response
The single layer of intestinal epithelium serves as a barrier between the host and its external environment that controls the interaction between the luminal contents and the internal milieu of the body. The intestinal epithelial monolayer contains several types of specialized cells: (1) enterocytes, for absorption of nutrients; (2) goblet cells, producing secretory and gel-forming mucins which are glycosylated proteins that form polymeric nets called mucus layer, a physical barrier between intestinal bacteria and epithelial cells; (3) enteroendocrine cells, secreting various hormones regulating digestive function;
- (4) Paneth cells, residing at crypt base and secreting antimicrobial peptides such as lysozyme, defensins, and cryptidins; (5) microfold cells (M cells), sampling antigens from the lumen to subepithelium; and (6) tuft cells, for chemosensing function in the epithelium [33,34]. These epithelial cells are connected by intercellular desmosomes, tight junctions (TJs), and adherent junctions (AJs), which create a physical barrier for luminal contents of the gut and regulate epithelial permeability. TJs are a complex network formed by transmembrane proteins such as claudins, occludin, tricellulin, and junctional adhesion molecules and cytosolic scaffold proteins such as zonulae occludens (ZO) and cingulin [35,36]. Both TJs and AJs are connected to the actin cytoskeleton and form an apical junction complex. On the basal side, epithelial cells are connected by hemidesmosomes.
The intestinal epithelium lies between the commensal organisms in the gut lumen and the immune cells in lamina propria. The complex immune interactions between commensal microflora, the epithelial layer, and the subepithelial immune cells maintain homeostasis under normal conditions. Lamina propria contains the gut-associated lymphoid tissue (GALT) which is comprised of Peyer’s patches, a group of lymphoid follicles containing several immune cells, such as specialized M cells, dendritic cells, T cells, B cells, intraepithelial lymphocytes, and macrophages [37]. The dendritic cells (DCs) from lamina propria sample the luminal food and microbial antigens by extending their dendrites between epithelial cells and transport to antigen-presenting cells (APCs) in GALT [38,39]. Upon activation, GALT performs effector immune functions by activating immune cells to produce specific cytokines from T cells and immunoglobulins from B cells. Antigens in the gut lumen can be taken up by specialized M cells and delivered to DCs for effector functions in the Peyer’s patches [40]. Intestinal epithelial cells themselves can also act as dynamic sensors by pattern recognition molecule receptors (PRRs) such as toll-like receptors (TLRs) and nucleotide-binding oligomerization domain (NOD)-like receptors (NLRs) to sense pathogen-associated molecular patterns.
Gut microflora and their metabolites play a major role in maintaining epithelial barrier function and immune homeostasis. Among the microbial metabolites, butyrate involves a number of signaling pathways in the gut immune cells and epithelial cells for the restoration of impaired colonic barrier function and gut homeostasis (Figure 3). The pathophysiology of IBD involves both epithelial barrier dysfunction and abnormal immune-cell activation. Changes in TJs structure, downregulation of claudin proteins, and upregulation of pore-forming claudin-2 were observed in both CD and UC conditions [34]. Since 2007, butyrate was found to enhance the intestinal barrier function by facilitating tight junction assembly via activation of AMPK, Akt, and other signaling pathways in a dose-dependent manner as shown in studies with transepithelial electrical resistance (TEER) and fluorescein isothiocyanate-dextran (FITC-dextran) permeability assays in in vitro settings [41–43]. Marinelli et al. [44] demonstrated that butyrate regulates the epithelial barrier function by acting as a signaling molecule for cell-surface G-protein-coupled receptors (GPRs) and nuclear factors (NFs). Indeed, butyrate was found to induce T cell-independent IgA secretion in the colon via activation of GPR41 (free fatty acid receptor 3, FFAR3) and GPR109A (hydrocarboxylic acid receptor 2, HCAR2), and inhibition of histone deacetylase (HDAC) to restore epithelial barrier function under inflammatory conditions [45]. Studies also explored the effect of butyrate on claudins expression. Zheng et al. [46] reported that butyrate promotes epithelial barrier function through interleukin-10 receptor α-subunit (IL-10RA)-dependent
receptor α-subunit (IL-10RA)-dependent repression of claudin-2 TJ protein. Wang et al.
repression of claudin-2 TJ protein. Wang et al. [47] demonstrated that butyrate treatment improved epithelial barrier function via the upregulation of claudin-1 transcription by facilitating the interaction between specific motifs in the claudin-1 promoter region and SP1 transcription factor. Moreover, butyrate enhances mucin secretion and protects epithelial cells by inducing MUC2 gene expression via AP-1 and acetylation/methylation of histones at the MUC2 promoter in intestinal epithelial goblet cells [48]. Hypoxia-inducible factor 1 (HIF-1)-dependent mechanism may also contribute to butyrate-enhanced epithelial barrier function [49].
tifs in the claudin-1 promoter region and SP1 transcription factor. Moreover, butyrate enhances mucin secretion and protects epithelial cells by inducing MUC2 gene expression via AP-1 and acetylation/methylation of histones at the MUC2 promoter in intestinal epithelial goblet cells [48]. Hypoxia-inducible factor 1 (HIF-1)-dependent mechanism may also contribute to butyrate-enhanced epithelial barrier function [49].
- Figure 3. Schematic overview of butyrate transport and cellular mechanisms. Butyrate is absorbed by intestinal epithelial cells via active transport mediated by MCT1 and SMCT1 transporters or via passive diffusion. Butyrate activates GPRs and couples to G proteins to interact with downstream effectors such as HDACs to reduce inflammation.
- Figure 3. Schematic overview of butyrate transport and cellular mechanisms. Butyrate is absorbed by intestinal epithelial cells via active transport mediated by MCT1 and SMCT1 transporters or via passive diffusion. Butyrate activates GPRs and couples to G proteins to interact with downstream effectors such as HDACs to reduce inflammation.
An inappropriate immune response to antigens derived from intestinal components is a key feature in IBD, leading to an imbalance of inflammatory cytokines, tissue damage, and disease progression [50,51]. Increased phagocytic activity of macrophages and cytokines’ secretion (for example, IL-1, IL-6, IL-17, and TNF) has been found in IBD patients [52]. T lymphocytes (T-cells) play a crucial role in maintaining immune homeostasis by regulating innate and adaptive immune responses. Upon specific antigen stimulation, naïve CD4+ T-cells differentiate into effector T helper (Th) cells, including Th1, Th2, T regulatory (Treg), and Th17 cells [53]. Each Th type secretes specific cytokines to perform protective or pathogenic roles. Treg cells have immunosuppressive properties that help to maintain immune homeostasis by secreting anti-inflammatory cytokines, including IL-10 [54]. IBD is associated with dysregulated T-cell immune responses such as increased Th1, Th2, and Th17 cell function and decreased Treg cells function [55]. Th17 produces inflammatory cytokines such as IL-17A, IL-17F, and IL-21 which are involved in the pathogenesis of IBD. Gut microbial metabolite butyrate regulates the differentiation and proliferation of T cells (Figure 4). Butyrate administration enhanced Treg cell function and suppressed IL-17 levels
An inappropriate immune response to antigens derived from intestinal components is a key feature in IBD, leading to an imbalance of inflammatory cytokines, tissue damage, and disease progression [50,51]. Increased phagocytic activity of macrophages and cytokines’ secretion (for example, IL-1, IL-6, IL-17, and TNF) has been found in IBD patients [52]. T lymphocytes (T-cells) play a crucial role in maintaining immune homeostasis by regulating innate and adaptive immune responses. Upon specific antigen stimulation, naïve CD4+ T-cells differentiate into effector T helper (Th) cells, including Th1, Th2, T regulatory (Treg), and Th17 cells [53]. Each Th type secretes specific cytokines to perform protective or pathogenic roles. Treg cells have immunosuppressive properties that help to maintain immune homeostasis by secreting anti-inflammatory cytokines, including IL-10 [54]. IBD is associated with dysregulated T-cell immune responses such as increased Th1, Th2, and Th17 cell function and decreased Treg cells function [55]. Th17 produces inflammatory cytokines such as IL-17A, IL-17F, and IL-21 which are involved in the pathogenesis of IBD. Gut microbial metabolite butyrate regulates the differentiation and proliferation
- as well as Th17 cells in the peripheral blood and colon tissues of TNBS-induced colitis rats compared to a control group [54]. Zimmerman et al. [56] have demonstrated that
duced colitis rats compared to a control group [54]. Zimmerman et al. [56] have demonstrated that butyrate inhibits proliferation of both CD4+ and CD8+ T cells in a dose-de-
butyrate inhibits proliferation of both CD4 and CD8+ T cells in a dose-dependent manner and it induces apoptosis in T cells through the Fas-mediated apoptosis pathway. Butyrate facilitates Treg cell differentiation by increasing histone H3 acetylation at the promoter and CNS3 region of the FOX3 gene locus [57]. Chen et al. [58] found that butyrate enhanced Th1 differentiation by promoting IFN-γ levels and T-bet expression in healthy conditions, but inhibited Th1 differentiation through IL-10 production and T-bet expression in colonic inflammation. In addition, butyrate has been shown to regulate inflammatory response by influencing NF-κB activity. NF-κB is a transcription factor involved in the regulation of various inflammatory mediators and cytokines expression including, TNF-α and IL-6 [59]. Butyrate is shown to reduce inflammatory response by suppressing NF-κB activity. Several studies have demonstrated the ability of butyrate to reduce NF-κB activity in human colon-cell lines and in lamina propria mononuclear cells isolated from CD patients [60–62]. Butyrate activates transmembrane GPRs and nuclear receptors such as aryl hydrocarbon receptor (AhR) in the intestinal epithelial cells. AhR is a ligand-activated transcription factor that resides in the cytosol in activated form, and translocates to the nucleus upon activation, thereby regulating AhR-dependent gene expression [63,64]. SCFAs, including butyrate, are shown to enhance AhR ligand interactions in mouse and human colon cells [44,65].
pathway. Butyrate facilitates Treg cell differentiation by increasing histone H3 acetylation
- at the promoter and CNS3 region of the FOX3 gene locus [57]. Chen et al. [58] found that butyrate enhanced Th1 differentiation by promoting IFN-γ levels and T-bet expression in healthy conditions, but inhibited Th1 differentiation through IL-10 production and T-bet expression in colonic inflammation. In addition, butyrate has been shown to regulate inflammatory response by influencing NF-κB activity. NF-κB is a transcription factor involved in the regulation of various inflammatory mediators and cytokines expression including, TNF-α and IL-6 [59]. Butyrate is shown to reduce inflammatory response by suppressing NF-κB activity. Several studies have demonstrated the ability of butyrate to reduce NF-κB activity in human colon-cell lines and in lamina propria mononuclear cells isolated from CD patients [60–62]. Butyrate activates transmembrane GPRs and nuclear receptors such as aryl hydrocarbon receptor (AhR) in the intestinal epithelial cells. AhR is a ligand-activated transcription factor that resides in the cytosol in activated form, and translocates to the nucleus upon activation, thereby regulating AhR-dependent gene expression [63,64]. SCFAs, including butyrate, are shown to enhance AhR ligand interactions in mouse and human colon cells [44,65].
- Figure 4. Microbial metabolite butyrate in the regulation of host immune function. Butyrate plays a key role in the maintenance of intestinal immune homeostasis. It promotes Treg differentiation and secretion of IL-10 and secretary IgA but inhibits Th17 cell differentiation.
5. Therapeutic Implications of Butyrate for IBD
IBD is characterized by aberrant immune response and barrier dysfunction and is associated with a reduced number of butyrate-producing bacteria in the gut. As butyrate was found to not only provide energy to colonic epithelial cells but also help maintain intestinal integrity and modulate immune responses [43]; numerous studies have investigated the role of various forms of butyrate in reducing gut inflammation [66–68]. Many of the studies have demonstrated the efficacy of oral butyrate supplements, butyrate enema, butyrogenic diet, and bacterial supplements in the treatment of IBD.
- 5.1. Butyrate Supplements 5.1.1. Oral Administration
Dysbiosis of gut microbiota leads to decreased butyrate synthesis and impaired butyrate metabolism as observed in IBD [69]. Although a low concentration of butyric acid is commonly present in our daily regular diet, it may not be sufficient to restore the epithelial function in inflammation in the colon. Many studies have investigated the therapeutic potential of butyrate oral supplements in gut inflammation in both preclinical studies and clinical trials. Table 2 summarizes these results (Table 2). Butyrate has been shown to reduce gut inflammation and ameliorate symptoms in a dose-dependent manner. Butyrate at 20 mg/kg/day or lower doses was found to have no significant effect, while at 100 mg/kg, it was effective against inflammation in mice [68,70]. Lee et al. [68] reported that the oral supplementation of sodium butyrate at 100 mg/kg of body weight daily decreased colitis scores, prevented body weight loss, and induced histone H3 acetylation in colonic mucosa in mouse models of acute and chronic colitis. Moreover, butyrate treatment restored the microbial community diversity and reduced microbiota dysbiosis in gut inflammation [71].
As orally supplemented butyrate is rapidly absorbed in the duodenum, the majority of the orally administered butyrate would not reach the colon. Moreover, the clinical application of oral butyrate is limited due to its unpleasant taste and odor. To address these issues, some studies used colon-targeted formulations and encapsulated butyrate to test if butyrate in such formulations has better effects in IBD patients, especially UC patients [67,72,73]. Sabatino et al. [72] demonstrated that enteric-coated butyrate tablets administration effectively reduced ileocaecal inflammation and maintained clinical remission in Crohn’s disease patients. Lipophilic microencapsulated sodium butyrate treatment showed enrichment of butyrogenic colonic bacteria in IBD patients [67]. Wang et al. [74] developed butyrate micelles so that butyrate is released in the lower gastrointestinal tract. They found that butyrate micelles significantly improved intestinal barrier function and reduced disease severity in DSS-induced colitis and CD45RBhiT-cell transfer colitis in mice.
- Table 2. Impact of oral butyrate supplements on IBD.
Treatment Name Concentration Colitis Model Effects Authors Mice Sodium butyrate 0.5% of sodium
butyrate DSS-induced colitis Decreased mucosal
inflammation Vieira et al. [75]
Reduced disease activity index, rebalanced gut microbiota, and reversed the imbalance between pro- and anti-inflammatory cytokines
Butyrate-releasing polysaccharide derivative
Mice
200 mg/kg DSS-induced colitis
Zha et al. [66]
Balatable butyrate-releasing derivative, N-(1-carbamoyl-2phenylethyl) butyramide (FBA)
Mice
42.5 mg/kg DSS-induced colitis Reduced disease activity index Simeoli et al. [76]
Prevented mice from weight loss and suppressed intestinal inflammation
Citrobacter rodentium infection model
Mice Sodium butyrate 200 mM
Zhou et al. [77]
Table 2. Cont.
6. Discussion and Conclusions
Studies in the last two decades or so have shown that butyrate plays a critical role in the regulation of gut immune function and maintenance of barrier function and intestinal homeostasis. Butyrate regulates these functions by distinct transcriptional regulatory mechanisms, including inhibition of NF-κB and HDACs activation. The effects of butyrate on intestinal barrier function are in a dose-dependent manner, as high concentrations may induce apoptosis of epithelial cells and interrupt barrier function [41]. Most of the animal and human studies showed the positive effects of butyrate as a potential therapeutic agent to prevent inflammation and maintain remission in IBD. Butyrate oral supplements and butyrogenic diets are found to be effective in decreasing disease activity index and reducing inflammation. However, among nine preclinical studies on the effect of oral butyrate supplements in mouse models of colitis, one study showed no significant reduction in colon inflammation by butyrate supplement [79]. In that study, sodium butyrate was shown to modulate gut microbial composition compared to the control and colitis groups [79]. Similarly, in all four clinical studies, only one study reported no significant difference between sodium butyrate supplements [81]. Pietrzak et al. [81] assessed the effect of oral sodium butyrate along with standard therapy in newly diagnosed IBD children and adolescents and reported no significant effects comparing the sodium butyrate group with the placebo group. Butyrate administration is largely safe, though a few adverse effects have been noted [107]. Lin et al. [107] noticed that butyrate at excessive doses (more than 150 mmol/L) induced minimal mucosal damage in the colon and distal ileum in newborn rats.
A limitation of this review is that we did not perform any statistical analysis such as meta-analysis for the included studies. This is mainly due to the scarcity of data and heterogeneity of the studies, as various doses, forms, and administration routes of butyrate have been used in these studies. More clinical trials are required to determine the effective doses and forms of butyrate supplements for IBD patients.
In conclusion, butyrate at appropriate concentrations helps to maintain intestinal barrier function and regulate the immune response in the gut. Clinical trials and animal studies have shown that butyrate can reduce mucosal inflammation and improve barrier function in UC and CD. Butyrate formulations and butyrogenic compounds may represent alternative therapeutic approaches for IBD. Combination therapies with butyrate and other SCFAs may further increase the efficacy of butyrate in the treatment of IBD. Although most of the studies have shown the beneficial effects of butyrate in colitis models and IBD patients, more clinical studies are needed to understand the impact of butyrate administration alone or with standard therapy in the management of IBD.
Author Contributions: N.R. searched the literature and drafted the manuscript. X.-Z.S. and R.G. reviewed and revised it. X.-Z.S. obtained the funds and helped to organize the review. All authors have read and agreed to the published version of the manuscript.
Funding: Thisstudywas supported inpartby the United States Department of Defense (W81XWH2010681 to X.-Z.S.) and the National Institute of Health (R01 DK124611 to X.-Z.S.).
Institutional Review Board Statement: Not applicable. Informed Consent Statement: Not applicable. Data Availability Statement: Available upon request. Acknowledgments: The authors wish to thank Yingzi Cong for beneficial discussions. All graphical images were created using BioRender. Conflicts of Interest: The authors declare that this work was completed in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
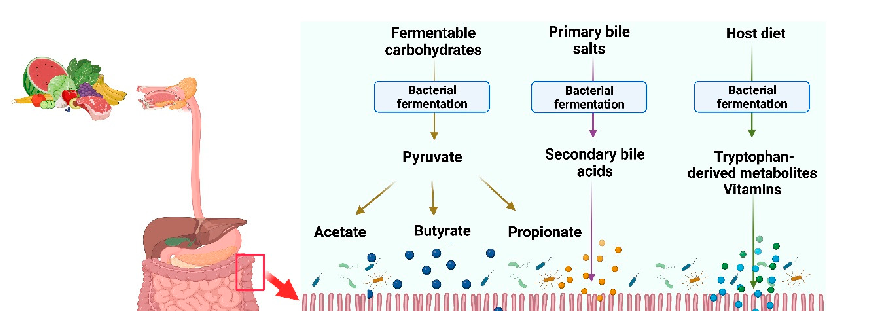
Figure 6
Butyrate modulates immune cell function in the gut mucosa, suppressing pro-inflammatory macrophage and dendritic cell activation while promoting regulatory T-cell differentiation. These immunomodulatory effects contribute to intestinal homeostasis.
diagram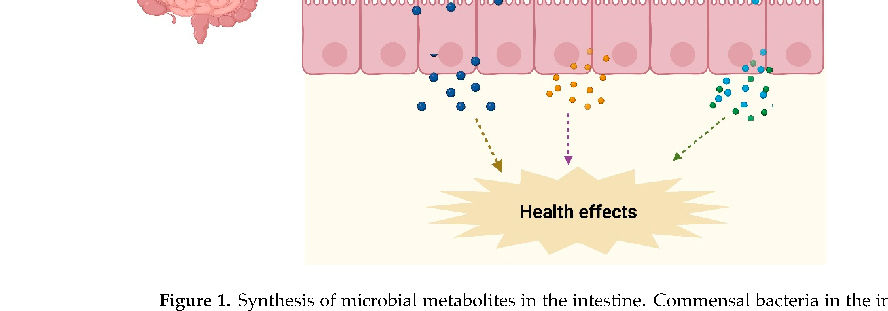
Figure 7
Dietary fiber fermentation pathways in the colon illustrate how complex carbohydrates are sequentially broken down by microbial consortia, ultimately yielding butyrate, propionate, and acetate as primary end products.
diagram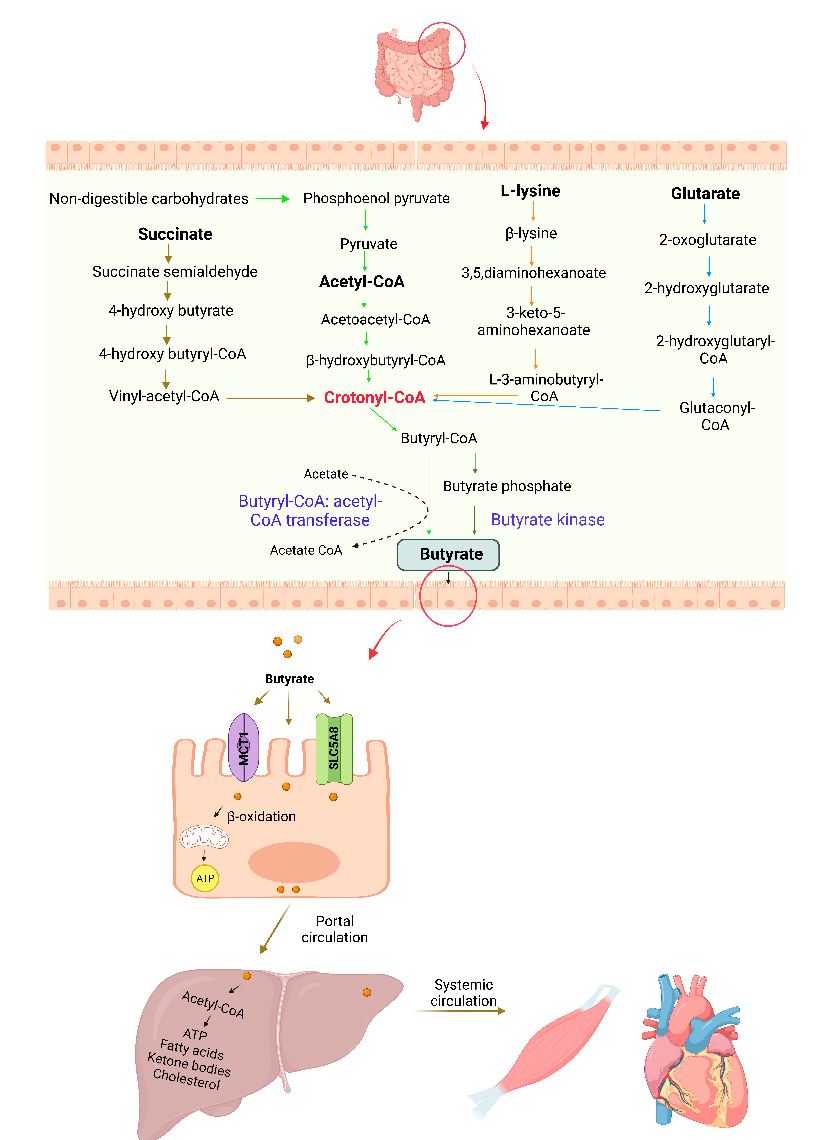
Figure 8
Clinical evidence from intervention studies suggests that butyrate supplementation, either orally or via enema, may reduce disease activity scores in ulcerative colitis patients. Response rates vary across delivery methods and formulations.
chartUsed In Evidence Reviews
Similar Papers
Frontiers in immunology · 2019
Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases.
Proceedings of the National Academy of Sciences of the United States of America · 2014
The microbial metabolite butyrate regulates intestinal macrophage function via histone deacetylase inhibition.
Gut · 2014
A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis.
Gut · 2011
Dysbiosis of the faecal microbiota in patients with Crohn's disease and their unaffected relatives.
World journal of gastroenterology · 2018
Relationship between intestinal microbiota and ulcerative colitis: Mechanisms and clinical application of probiotics and fecal microbiota transplantation.
Digestion · 2016