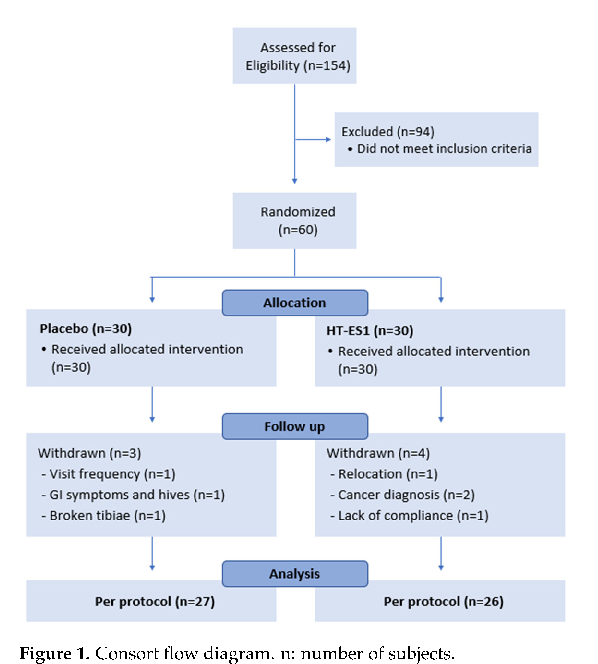
Deskripsi
Study product composition and dosing protocol for heat-treated Bifidobacterium longum CECT 7347 are described, detailing the intervention design for this gastrointestinal symptom trial.
More Figures from This Paper
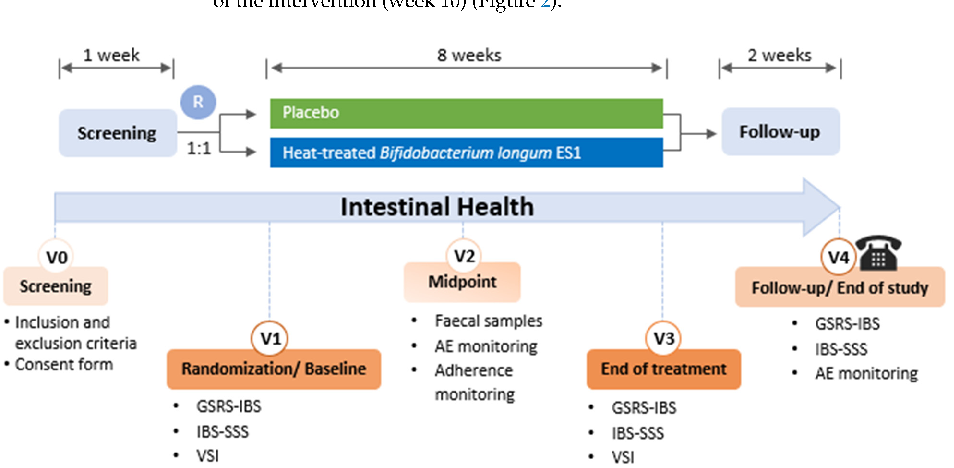
Figure 3
Gastrointestinal symptom scores at baseline and follow-up are compared between the postbiotic HT-ES1 and placebo groups, showing changes in digestive comfort over the intervention period.
chart
Figure 4
Individual gastrointestinal symptom subscores including bloating, abdominal pain, and flatulence are presented for postbiotic and placebo groups across the study timepoints.
chartFigure 2
Diagram
598 × 664px
· 103,5 KB
Source Paper
Effect of Postbiotic Bifidobacterium longum CECT 7347 on Gastrointestinal Symptoms, Serum Biochemistry, and Intestinal Microbiota in Healthy Adults: A Randomised, …Cite This Figure
 > Source: Malwina Naghibi et al. "Effect of Postbiotic Bifidobacterium longum CECT 7347 on Gastrointestinal Sympto." *Nutrients*, 2024. PMID: [39599737](https://pubmed.ncbi.nlm.nih.gov/39599737/)
<figure> <img src="https://pdfs.citedhealth.com/figures/39599737/166.png" alt="Study product composition and dosing protocol for heat-treated Bifidobacterium longum CECT 7347 are described, detailing the intervention design for this gastrointestinal symptom trial." /> <figcaption>Figure 2. Study product composition and dosing protocol for heat-treated Bifidobacterium longum CECT 7347 are described, detailing the intervention design for this gastrointestinal symptom trial.<br> Source: Malwina Naghibi et al. "Effect of Postbiotic Bifidobacterium longum CECT 7347 on Gastrointestinal Sympto." <em>Nutrients</em>, 2024. PMID: <a href="https://pubmed.ncbi.nlm.nih.gov/39599737/">39599737</a></figcaption> </figure>