Pharmacological effects of different ginger juices on the concurrent symptoms in animal models of functional dyspepsia: A comparative study.
Study Design
- Type d'étude
- In Vitro
- Population
- None
- Intervention
- Pharmacological effects of different ginger juices on the concurrent symptoms in animal models of functional dyspepsia: A comparative study. None
- Comparateur
- Control
- Critère de jugement principal
- None
- Direction de l'effet
- Positive
- Risque de biais
- Unclear
Abstract
OBJECTIVE: Patients with gastrointestinal disorders commonly suffer from poor treatment outcomes and adverse effects of traditional pharmacological therapy. Herbal medicine is a favorable alternative due to the low risk of side effects. This study was performed to explore the antiemetic effects and the improvement effect on gastrointestinal function of components of three ginger juice excipients. METHODS: The compositions were analyzed by liquid chromatograph mass spectrometer (LC-MS), especially the gingerols of dried ginger juice (DGJ), fresh ginger juice (FGJ), and fresh ginger boiled juice (FGBJ). Furthermore, the respective gastrointestinal effects on rat models with functional dyspepsia (FD) were compared. RESULTS: The 6-keto-PGF1α levels in the serum of the treated groups were significantly reduced (p < 0.05), as compared with the control group. Compared with the cisplatin group, there was an apparent reduction in kaolin intake for DGJ, FGJ, and FGBJ (p < 0.01; p < 0.01; p < 0.05). The intestinal propulsive rate of the rats in the treated group was significantly higher than that in the control group (p < 0.05). Ginger juices significantly improved gastrointestinal function in rats. Eight common components were found in DGJ, FGJ, and FGBJ, among which 6-paradol, 10-gingerol, and 12-shogaol led to inhibited gastric mucosal damage function effect according to the Pearson correlation analysis. Only 6-shogaol was found to have a positive correlation with gastrointestinal function effect through Pearson correlation analysis. CONCLUSION: Ginger juice should be recommended for the medicinal materials used in the treatment of concurrent symptoms of FD.
En bref
Ginger juice should be recommended for the medicinal materials used in the treatment of concurrent symptoms of FD, as Ginger juices significantly improved gastrointestinal function in rats.
Full Text
Received:18September2018 | Revised:16January2019 | Accepted:16February2019
DOI: 10.1002/fsn3.990
O R I G I N A L R E S E A R C H
Pharmacological effects of different ginger juices on the concurrent symptoms in animal models of functional dyspepsia: A comparative study
Ling‐yun Zhong | Heng‐li Tong | Jing Zhu | Mu Lv
SchoolofPharmacy,JiangxiUniversityof TraditionalChineseMedicine,Nanchang, China
Correspondence Ling‐yunZhong,SchoolofPharmacy,Jiangxi UniversityofTraditionalChineseMedicine, Nanchang,China. Email:[email protected]
Funding information NationalNaturalScienceFoundationof China,Grant/AwardNumber:81260643, 81460605and81660663
Abstract
Objective: Patientswithgastrointestinaldisorderscommonlysufferfrompoortreatmentoutcomesandadverseeffectsoftraditionalpharmacologicaltherapy.Herbal medicineisafavorablealternativeduetothelowriskofsideeffects.Thisstudywas performedtoexploretheantiemeticeffectsandtheimprovementeffectongastrointestinalfunctionofcomponentsofthreegingerjuiceexcipients. Methods: Thecompositionswereanalyzedbyliquidchromatographmassspectrometer(LC‐MS),especiallythegingerolsofdriedgingerjuice(DGJ),freshgingerjuice (FGJ),andfreshgingerboiledjuice(FGBJ).Furthermore,therespectivegastrointestinaleffectsonratmodelswithfunctionaldyspepsia(FD)werecompared. Results: The6‐keto‐PGF1α levelsintheserumofthetreatedgroupsweresignificantlyreduced(p < 0.05),ascomparedwiththecontrolgroup.Comparedwiththe cisplatingroup,therewasanapparentreductioninkaolinintakeforDGJ,FGJ,and FGBJ(p < 0.01; p < 0.01; p < 0.05).Theintestinalpropulsiverateoftheratsinthe treatedgroupwassignificantlyhigherthanthatinthecontrolgroup(p < 0.05).Ginger juicessignificantlyimprovedgastrointestinalfunctioninrats.EightcommoncomponentswerefoundinDGJ,FGJ,andFGBJ,amongwhich6‐paradol,10‐gingerol,and 12‐shogaolledtoinhibitedgastricmucosaldamagefunctioneffectaccordingtothe Pearsoncorrelationanalysis.Only6‐shogaolwasfoundtohaveapositivecorrelation withgastrointestinalfunctioneffectthroughPearsoncorrelationanalysis. Conclusion: Gingerjuiceshouldberecommendedforthemedicinalmaterialsusedin thetreatmentofconcurrentsymptomsofFD.
K E Y W O R D S
driedgingerjuice,freshgingerboiledjuice,freshgingerjuice,gastrointestinaleffects,gingerol
1 | INTRODUCTION
GingerrhizomeofZingiber officinale Roscoe,awidelyusedherbalmedicine,ispungentandwarmaccordingtotraditionalChinesemedicine
(TCM)theory.Thechemicalcompositionofbiologicalpropertiesof gingerprimarilyincludesnonvolatilepungentcompounds,6‐,8‐,10‐ gingerolsand6‐,8‐,10‐shogaols(Ho&Chang,2018).Gingerhasbeen knowntopreventorarrestnauseaandvomiting,diastoleandprotect
ThisisanopenaccessarticleunderthetermsoftheCreativeCommonsAttributionLicense,whichpermitsuse,distributionandreproductioninanymedium, providedtheoriginalworkisproperlycited. ©2019TheAuthors.Food Science & NutritionpublishedbyWileyPeriodicals,Inc.
Food Sci Nutr. 2019;7:2205–2213. www.foodscience-nutrition.com | 2205
thecoronaryartery,inhibitthecontractionofsmallintestineandmuch more(Chatturong,Kajsongkram,Tunsophon,Chanasong,&Chootip, 2018;Thamlikitkuletal.,2017;Wuetal.,2018).InTCM,gingerisregularlyusedasaMinisterialdrugtopotentiatetheMonarchdruginthe treatmentofthemaindiseaseortotreattheconcurrentsymptoms.
Gingerolsandshogaolsareactiveconstituentsofginger,which aredifficulttopurifyduetoitsgreatvarietyandstructuralsimilarity.Thus,itisnecessarytodeveloparapidanalysisandidentificationmethodforgingeranditssubtypes.Analysismethodshave been developed, including nuclear magnetic resonance (NMR), high‐performanceliquidchromatography(HPLC),andultrahigh‐performanceliquidchromatography–massspectrometry(UHPLC‐MS) (Ghasemzadeh, Jaafar, Baghdadi, & Tayebi‐Meigooni, 2018; Li et al.,2018;Park&Jung,2016;Raietal.,2015).However,theHPLC technologyneedstobeappliedincombinationwiththestandard forqualitativeanalysisofunknowncompounds.GC‐MShasbeen widelyappliedingingeranalysis,butgingerolishardtobedetected becauseofitslowvolatility.HPLC‐MScouldintegratethestrong separation ability of liquid chromatography with high sensitivity andhighselectivity,providingstructuralinformation.Inparticular, high‐resolutiontime‐of‐flightmassspectrometrycanprovideaccuratemolecularweightinformationanddeducetheformulaofthe specificcompound.Park&Jungseparatedandquantified8major gingerolsandshogaolsfromgingerusingtheultrahigh‐performance liquid‐phasechromatography–electrosprayionization–tandemmass spectrometry(HPLC‐ESI‐MS/MS)technology(Park&Jung,2016).
NumerousTCMscanpreventgastricmucosaldamage,vomiting, andgastrointestinalmovement.Theinhibitioneffectsofaqueous extractfromArtemisiacapillariswereinvestigatedintermsofROS andNF‐kBofacutegastricmucosalinjuryinducedbyethanol(Yeo, Hwang,Kim,Youn,&Lee,2018).Strikingly,increased6‐keto‐PGF1α waspreviouslydemonstratedtoreducethedegreeofgastricmucosalinjury(Kouetal.,2018).Thegingerextractcanpreventthe picabehaviorofratsinducedbycisplatin(Huetal.,2016).Tangweian decoction,aChineseherbalmedicine,cansignificantlypromoteintestinalpropulsionrateindiabeticmice(Tianetal.,2017).
BasedontheChinesePharmacopoeia(2015version),thereare twosubtypesofginger,namelyfreshgingeranddriedginger.Both formscanbeusedtoobtaingingerjuice.AccordingtotheNational ProcessingStandardofTraditionalChineseMedicine—1988,ginger includesfreshgingerjuice(FGJ),freshgingerboiledjuice(FGBJ), and dried ginger juice (DGJ). However, no clear distinction was identifiedamongthegingerjuices.Thesegingerjuicesareoften interchangeablewhenpreparingaTCMformulation.Inthecurrent study, we aimed to analyze the nonvolatile components in FGJ, FGBJ,andDGJbyLC‐MS.Aftertheratswereadministeredwith gingerjuices,theethanol‐inducedgastricmucosaldamageindex andthelevelsofIL‐8,TNFα,6‐keto‐PGF1α,cisplatin‐inducedemesis,andtheintestinalpropulsionrateweredeterminedtoevaluate the effect on gastroenteric motivity. The chemical compositions of these three ginger juices were compared to determine which oneisthemosteffectiveinthepreparationofChinesetraditional medicine.
2 | MATERIALS AND METHODS
2.1 | Materials
A liquid chromatography coupled with a mass selective detector wasemployedtoanalyzethenonvolatilecomponents.Inaddition, ahigh‐speedrefrigeratedcentrifugeandfreeinstrumentwerealso used.Allinvolvedchemicalswereofanalyticalgrade,includinganhydrousethanol,petroleumether,andchloroform,whichwereseparatelypurchasedfromXilongChemicalCo.,Ltd,andTianjinDamao ChemicalReagentFactory.Nutritionalsemi‐pasteswerealsosupplied.Kaolinwaspreparedaspreviouslydescribed.Briefly,kaolin andarabicgumweremixedataratioof99:1,followedbytheadditionofdistilledwater(volume)toformathickpaste.Thepaste wasfurthercutintopiecesresemblingregularratchowpellets.After beingcut,thepelletsweredriedatroomtemperaturefor48hr(Han etal.,2014).
Freshforms(FoshanCity,GuangdongProvince)anddriedforms of ginger(QianweiCity,SichuangProvince)werepurchasedfrom the Tian Qi Hall medicine material crude slices limited company (ZhangshuCity,JiangxiProvince,China).
Theaforementioneddriedgingerwaspreparedforusebycuttingintopieces(0.3cm×0.3cm×0.3cm),followedbybeingbaked at55°Cfor18hr.
MaleSpragueDawley(SD)rats(weighing180and220g)were purchasedfromJi'nanPengYueExperimentalAnimalBreedingCo., Ltd(CertificateNo.SCXK(Lu)2014‐0007).Additionally,adultmale WistarratsandSpragueDawleyrats(ShanDong,CertificateNo. SCXK20130001),weighingbetween200and230g,wereincluded inthecurrentstudy.Allanimalexperimentswerecarriedoutin accordancewithguidelinesevaluatedandapprovedbytheEthics CommitteeofJiangxiUniversityofTraditionalChineseMedicine.
2.1.1 | Preparation of herbal decoctions
Fresh ginger juice
Firstly, 150g of fresh ginger was washed and squeezed as juice. Afterfilteringthejuice,theobtainedresidueswereresuspendedin theappropriateamountofwaterandsqueezedagain.Subsequently, theobtainedfiltrateswerecombined.
Fresh ginger boiled juice and dried ginger juice
Next,wesoaked150goffreshgingerand50gofdriedginger,respectively,in1Lwaterfor1hr.Afterboilingfor30minandfiltration,theobtainedresidueswereresuspendedinwaterandboiled foranother30min.Thefiltratewascombinedandconcentratedto 150ml.Thedosageratiooffreshanddriedgingeris3:1.
Sample preparation
Sampleswerepreparedbydissolving10mlofgingerjuicein20ml ofmethanol.Next,thesamplesunderwentultrasonicfor10minto breakthecells.Thesolutionswerefiltered,andthefiltratewasfurtherstrainedthrougha0.22‐μmMilliporefilter.
- 2.2 | Methods
- 2.2.1 | LC‐MS analytical condition
- 2.2.2 | Grouping and administration Ethanol‐induced gastric mucosal damage Theratswererandomlydividedintofivegroups:thenormalgroup, the model group, the FGJ group, the FGBJ group, and the DGJ group.FGJ,FGBJ,andDGJwereadministeredtoratsonceaday foradurationof7daysexcludingtheratsinthenormalandmodel groups,whichweregiven0.9%physiologicalsalineinsteadofFGJ, FGBJ,orDGJ.Alltheratsexceptthoseinthenormalgroupwere given 1ml absolute ethanol 1hr after the last administration of drugs.Subsequently,allanimalswereanesthetized.Next,thestomachswereremovedandopenedalongthelongcurvaturetoobserve thelesionsmacroscopically1hrafterbeinginjectedwith6mlof 4%paraformaldehyde.Scoringstandardwasasfollows:Intactgastricmucosawasscoredas0;point‐shapedbleeding,point‐shaped erosion, and strip‐shaped lesion of no more than 1mm were all scored as 1; strip‐shaped lesion with more than 1mm was doublyscored.Totalscoresofeveryratwereconsideredasthelesion index. Inhibition ratio of lesion formation (%)=(A−B)/A×100% (A,Bwerethelesionindicesofthemodelgroupanddrug‐treated group, respectively). Gastric tissues in the same location were fixed with 4% formalin, embedded with paraffin, and stained by HE.Inaddition,bloodsampleswereobtainedfromtheabdominal aorta.IL‐8,TNFα,and6‐keto‐PGF1αinplasmawereexaminedby radioimmunoassay.
Cisplatin‐induced emesis
After acclimatization to the laboratory environment for 3days, all animals were randomly divided into five groups, namely control, cisplatin,FGJ,FGBJ,andDGJgroups.Themiceinthecontrolgroup wereadministeredwithperitonealinjectionsandgastriclavagewith normalsaline.Theanimalsinthecisplatingroupreceivedperitoneal injectionsofcisplatin(5mg/kg)andgastriclavagewithnormalsaline,whileanimalsinthethalidomideandgranisetrongroupsreceived peritonealinjectionsofcisplatin(5mg/kg)andgastriclavagewith,respectively,FGJ(10ml/kg),FGBJ(10ml/kg),orDGJ(10ml/kg).
Gastrointestinal propulsion
Miceweredividedinto4groups,namelythenormal,FGJ,FGBJand DGJgroups.Themicewerefastenedfor12hrpriortotheexperiment.Fifteenminutesafterthelastadministrationofdrugs,themice weregivensemi‐solidnutritioninavolumeof1ml.Theanimalswere thensacrificedbycervicaldislocation20minafterthemeal,andthe wholegastrointestinaltractwasremoved.Theentirelengthofthe intestineswasstretchedoutonpaper,andthedistancetheinkhad travelledfromthepyloruswasexpressedasthepercentageofthe totallengthofsmallintestinebetweenpylorusandcecum.Atotalof 10animalswereusedforeachdoseortreatment,respectively.
2.3 | Statistical analysis
Allexperimentswereperformedatleastsixtimes.Theresultswere presentedasmeanvalue±standarderror(SE).Statisticalanalyses wereperformedusingtheSPSS19softwarewithindependent‐samplesttest(ref).Avalueofp < 0.05wasconsideredtobestatistically significant.Pearsoncorrelationanalysiswasappliedfortherelative analysisofcommoningredientsinallgingerjuicesandtheirpharmacodynamicsindicesusingtheSPSS19software.
3 | RESULTS
3.1 | Analysis for chemical compositions of FGJ,FGBJ, and DGJ
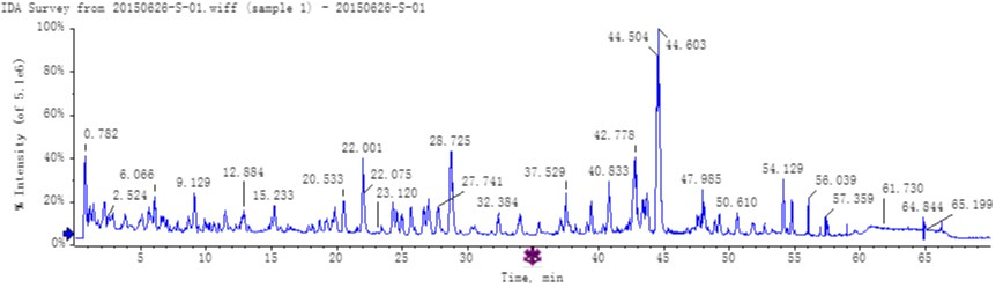
Typicaltotalionchromatograms(TICs)ofthenonvolatilecomponent fractionextractedfromFGJ,FGBJ,andDGJareshowninFigure1. AlthoughtheTICswerecomplicated,mostchromatographicpeaks werenotedtobewellseparated.PositiveionmodetotalionchromatogramswereanalyzedaccordingtoPeakviewTM1.7.viamethods oftargetscreeningandnontargetscreening.Thecommonchemical compositions of FGJ, FGBJ, and DGJ were initially identified (Table1)(Denniff,Macleod,&Whiting,1981;Hu,Guo,etal.,2011; Hu,Rayner,etal.,2011;Jiang,Sólyom,Timmermann,&Gang,2010; Kikuzaki,Kawasaki,&Nakatani,1994;Zhan,Wang,Xu,&Yin,2008; Zhang,Gan,&Hong,2005).
3.2 | Ethanol‐induced gastric mucosal damage
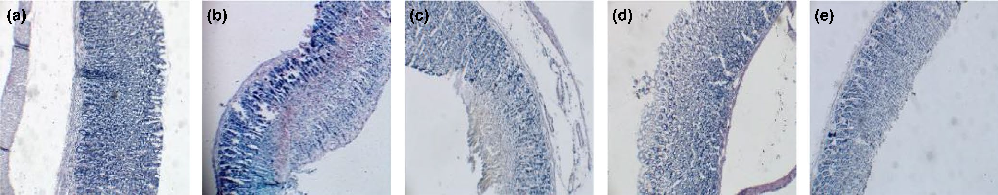
Evidentlargebandlikehemorrhagicerosionsintheglandularstomachwerenotedinthemodegroup(Figures2and3,Table2).In the FGJ, FGBJ, and DGJ groups, gastric mucosal damages were preventedinadose‐dependentmanner,whencomparedwiththe modelgroup.
Comparedwiththecontrolgroup,thegastricmucosaofrats inthemodelgroupwasfoundtobesignificantlydamaged.Inaddition, shedding, bleeding, and infiltration of inflammatory cells were apparent in the mucosa. Relative to the model group, the gastric mucosa was observed to be lighter and presented with lessinflammatorycellinfiltrationinthegingerboiledjuicegroup and ginger juice group, while slightly alleviated gastric mucosal
- (a)
- (b)
- (c)
- FIGURE 1 LC‐MSTIC(FGJ‐A,FGBJ‐B,DGJ‐C)
lesionandinflammatorycellinfiltrationwerenotedintheginger boiled juice group. However, the gastric was severely damaged inthedriedgingerboiledjuicegroupandgingerjuicegroup.The aforementionedresultsindicatethatgingerjuicecaninhibitthe inflammationofgastricmucosaandcanfurthermorepromotethe repairingprocessofgastricmucosa.
The ulcer index (UI) of the groups following administration wasfoundtobesignificantlylowerthanthatinthemodelgroup (p < 0.01).ComparedwiththeFGBJgroup,asignificantreduction intheratewasrevealedintheFGJandDGJgroups(p < 0.05).The significantreductionssuggestthatadministrationofgingerjuiceled todecreasedintestinalpropulsiverate(p < 0.05)(Tables2and3).
AsshowninTable3,themodelgroupshowednosignificantdifferencesrelativetothecontrolgroupinthecontentofIL‐8(p > 0.05).
ThecontentofTNFα waselevated,buttherewerenosignificant differences within any groups. And the content of 6‐keto‐PGF1α inserumsignificantlyincreasedcomparedwiththatinthecontrol group (p < 0.05).Thisdemonstratesthattheacuteinjurystimulates thebody'sinflammatoryresponse.Thecontentof6‐keto‐PGF1α in serumofginger‐treatedgroupwassignificantlydecreasedcompared withthemodelgroup(p < 0.01,p < 0.05).Furthermore,thecontent ofFGJgroupwaslowerthantheFGBJgroup,inwhichithasbasicallyreturnedtonormallevels.
3.3 | Cisplatin‐induced emesis
Twenty‐fourhoursafteradministeringperitonealinjectionsofcisplatin, kaolin intake was shown to be significantly increased in
- TABLE 1 PossiblecommonchemicalcompositionofthesamplesbyLC‐MS
N Name TR/min Formula MS Error (ppm)
Area% DGJ FGJ FGBJ
- 1 4‐shogaol 27.01 C15H20O3 249.1484[M+H]+ −0.5 1.75 0.89 1.83
- 2 6‐gingerdiol 40.84 C17H28O4 317.1717[M+Na]+ −1.9 1.62 1.60 5.27
- 3 6‐gingerol 44.56 C17H26O4 295.1896[M+H]+ −1.9 7.54 19.59 13.83 317.1717[M+Na]+ −2.7
- 4 6‐shogaol 44.57 C17H24O3 277.1792[M+H]+ −2.2 0.90 0.57 1.24
- 5 methyl‐6‐gingerol 50.61 C18H28O4 331.1875[M+Na]+ −1.3 0.82 1.61 1.91
- 6 3‐or5‐acetoxy‐6‐gingerdiol 51.81 C19H30O5 361.1982[M+Na]+ −1.1 0.63 0.65 0.89
- 7 diacetoxy‐4‐gingerdiol 52.69 C19H28O6 375.1771[M+Na]+ −1.9 0.29 0.21 0.45
- 8 8‐shogaol 54.13 C19H28O3 305.2105[M+H]+ −2.2 1.59 4.01 0
- 9 8‐gingerol 54.13 C19H30O4 345.2033[M+Na]+ −1 1.59 5.34 2.65
- 10 methyl‐3‐or
- 11 10-gingerdione 55.67 C21H32O4 371.2184[M+Na]+ −2.5 0.08 0 0
- 12 6‐paradol 56.1 C17H26O3 301.1771[M+H]+ −1 0 4.27 0
- 13 1‐dehydro‐6‐gingerdione 56.33 C17H22O4 313.1408[M+Na]+ −0.9 0 0.12 0.52
- 14 acetoxy‐8‐gingerol 56.94 C21H32O5 387.2124[M+Na]+ −4.7 0.13 3.82 0
- 15 10‐gingerol 56.96 C21H34O4 373.234[M+Na]+ −2.5 0.13 3.82 0
- 16 acetoxy‐10‐gingerol 58.99 C23H36O5 415.2441[M+Na]+ −3.3 0 1.50 0
- 17 12‐shogaol 59.12 C23H36O3 361.2727[M+H]+ −3 0 0.87 0
- 18 10‐shogaol 59.65 C21H32O3 355.2228[M+Na]+ −4.4 0.14 0.72 0
- 19 1‐dehydro‐10‐gingerdione 60.51 C21H30O4 347.2204[M+H]+ −3.8 0 1.15 0 369.2019[M+Na]+ −4.6
Note.DGJ:driedgingerjuice;FGJ:freshgingerjuice;FGBJ:freshgingerboiledjuice;LC‐MS:LiquidChromatograph‐MassSpectrometer.
- FIGURE 2 Protectionofgingerjuicesongastricmucosalinjuryinducedbyethanolinrats.(a)Normalgroup;(b)Modelgroup;(c)FGBJ group;(d)FGJgroup;(e)DGJgroup
- FIGURE 3 Gastrictissues.(a)Normalgroup;(b)Modelgroup;(c)FGBJgroup;(d)FGJgroup;(e)DGJgroup
- TABLE 2 Effectofethylacetateextractfromgingerjuiceon gastriclesionsinducedbyethanolinrats
Groups Number UI Normal 6 – Model 6 64.17±6.49 FGBJ 6 48.83±3.31* FGJ 6 27.33±2.80*,† DGJ 6 26.17±3.60*,†
Note.DGJ:driedgingerjuice;FGJ:freshgingerjuice;FGBJ:freshginger boiledjuice;UI:ulcerindex.
*p < 0.01versusmodelgroup;†p < 0.05versusFGBJgroup.
TABLE 3 EffectofgingerjuicesonIL‐8,TNFα,and6‐keto‐PGF1α activityofgastricmucosadamagedbyethanolinrats
Groups IL‐8 (pg/ml) TNFα (pg/ml) 6‐keto‐PGF1α (pg/ml) Normal 47.21±6.73 57.70±7.77 82.42±7.34 Model 47.01±2.34 63.90±6.42 95.67±7.95* FGJ 46.27±5.22 55.75±8.14 81.89±5.16‡ FGBJ 46.40±5.05 61.35±5.87 83.58±10.318† DGJ 47.11±2.27 67.04±6.27 84.55±8.00†
Note.DGJ:driedgingerjuice;FGJ:freshgingerjuice;FGBJ:freshginger boiledjuice.
*p < 0.01versusnormalgroup;†p < 0.05versusmodelgroup;‡p < 0.01 versusmodelgroup.
thecisplatinandadministeredgroups(p < 0.01,vs.controlgroup; Table4).However,thedifferenceinkaolinintakebetweentheFGJ groupandDGJgroupreflectednostatisticallysignificantdifference. After24hr,graduallydecreasingkaolinintakewasobservedinthe administeredandcisplatingroups.
3.4 | Gastrointestinal propulsion
TheresultsshowedthatFGBJ,FGJ,andDGJgroupsweresignificantlydifferentfromthenormalgroup(p < 0.01).Comparedwith theFGBJgroup,theFGJgroupwassignificantlyreduced(p < 0.05). Boiledgingerjuiceexhibitedthestrongesteffectongastrointestinal propulsion,followedbygingerboiledjuiceandgingerjuice.Among these,FGBJpresentedwiththestrongesteffectonintestinepropulsion(asshowninTable5).
accountedfor10%,45%,and45%,respectively,forthetotalscores ofthegastricmucosaldamagefunction.Kaolinintakeandintestinal propulsiverateaccountedfor50%and50%,respectively,forthe totalscoresofthegastrointestinalfunction.
Theingredients,including4‐shogaol,6‐gingerol,6‐shogaol,8‐ gingerol,6‐paradol,1‐dehydro‐6‐gingerdione,10‐gingerol,and12‐ shogaol,wereanalyzedaccordingly(Table6).Theresultsrevealed that6‐shogaol,6‐paradol,10‐gingerol,and12‐shogaolshowedsignificantpositivecorrelationwithgastrointestinalfunctionandinhibitedgastricmucosaldamagefunction(p < 0.05)(Table7).
4 | DISCUSSION
3.5 | Correlation analysis of common components and efficacy index
TheDGJgroupwiththelowestUIwasindexedas100points.The FGJ group with the lowest concentration of TNFα in serum was taken as 100 points. The FGJ group with the lowest concentrationof6‐keto‐PGF1αinserumwastakenas100points.TheFGBJ groupwiththelowestkaolinintakewasalsoindexedas100points. TheFGBJgroupwiththehighestintestinalpropulsiveratewasindexedas100points.Inaddition,theaveragevaluesoftheother groupsweresequentiallycalculated.UI,TNFα,and6‐keto‐PGF1α
TABLE 4 Changesinkaolinintake
Kaolin 24 hr 48 hr
Groups
Normal 0.1731±0.028 0.1556±0.058 Cisplatin 3.66±0.75 0.54±0.18 FGJ 2.86±0.68* 0.50±0.14 FGBJ 1.58±0.22* 0.40±0.29 DGJ 2.23±0.78* 0.77±0.21
Note.DGJ:driedgingerjuice;FGJ:freshgingerjuice;FGBJ:freshginger boiledjuice.
*p < 0.01versuscisplatingroup.
Thechiefpungentcompoundofgingerisgingerol.Gingerols,unstableinheat,arepronetodehydrationreactionandproducingthe correspondingshogaols(Jungetal.,2017),whichexertantioxidant, anti‐inflammatory,antiallergic,anticancer,andantimicrobialeffects (Semwal,Semwal,Combrinck,&Viljoen,2015).
Interestingly,thegastrointestinaleffectisoneofitsmajorfunctions.Inthecurrentstudy,weanalyzedgingerolsbyLC‐MSandtheir gastrointestinaleffects.Thegingerolschangedingingerjuiceswhatevermaybetheircomposition,andthegastrointestinaleffectsof gingerjuiceswereextremelydifferent.Atotalof8gingerolswere foundinDGJ,FGJ,andFGBJ,amongwhichthree,namely6‐paradol,10‐gingerol,and12‐shogaol,sharedapositivecorrelationwith
TABLE 5 Intestinalpropulsiverateofgroups
Intestinal propulsive distance (cm)
Intestinal propulsive rat%
Group n
Normal 10 15.19±1.09 28.48±2.48 FGBJ 10 24.59±2.18 52.01±3.18* FGJ 10 23.25±1.98 48.15±3.50* DGJ 10 26.07±2.54 50.66±4.14*
Note.DGJ:driedgingerjuice;FGJ:freshgingerjuice;FGBJ:freshginger boiledjuice.
*p < 0.01versusnormalgroup.
- TABLE 6 Commoningredients(relativecontents)X1 to X8andgastrointestinalcomprehensivevalueY
- TABLE 7 Pearsoncorrelationanalysis
X1 X2 X3 X4 X5 X6 X7 X8 Y1 Pearson −0.983 0.905 −0.801 0.986 0.994 0.994 −0.191 0.990 1
Y Relevance 0.059 0.140 0.204 0.054 0.035 0.035 0.439 0.044 1 Significant * * * N 3 3 3 3 3 3 3 3
X1 X2 X3 X4 X5 X6 X7 X8 Y2 Pearson 0.955 −0.600 0.988 −0.793 −0.929 −0.929 0.628 −0.940 1
Y Relevance 0.096 0.295 0.049 0.209 0.120 0.120 0.284 0.111 1 Significant * N 3 3 3 3 3 3 3 3
*Correlationissignificantatthe0.05level(1‐tailed).
theinhibitoryeffectsongastricmucosaldamage,onthebasisofa Pearsoncorrelationanalysis.Notably,only6‐shogaolpresentedwith a positive correlation with the gastrointestinal effect. Among the fourcompounds,6‐paradoland12‐shogaolwerepresentinFGJonly. 10‐gingerolwasfoundinDGJandFGJ.6‐shogaolwasfoundinDGJ, FGJ,andFGBJ.Importantly,thecontentof6‐shogaolineachginger juicesfromhightolowwasFGBJ,DGJ,andFGJ.The6‐shogaolof boiledgingerjuiceishigherthanthatofunboiledgingerjuice,itmay beattributedtothatgingerolinboiledgingerjuiceistransformedinto shogaolafterheating,andthecontentofgingerolsinfreshgingermay behigherthanthatinfriedginger.
Prostaglandinsarerecognizedasdefensiverepairfactorsofthe gastricmucosa.6‐keto‐PGF1αisastablemetaboliteofPG,andthe contentof6‐keto‐PGF1αcanbeusedtoreflectthecontentofPG inplasma.Thecurrentstudyfoundthatallgingerjuicescansignificantlyreducethecontentof6‐keto‐PGF1αinserum.Furthermore, 6‐paradol,10‐gingerol,and12‐shogaolwerenotdetectedinFGBJ, whichwasconsistentwiththeUIresultsofgastricmucosalinjury inducedbyethanol,revealingFGJandDGJhavesignificanttherapeuticeffectscomparedwithFGBJ.After24hr,kaolinintakeinthe FGBJ,DGJ,andFGJgroupswasgraduallydecreasedandshowedno statisticallysignificantdifference,suggestingthatallkindsofgingers canpreventvomitingeffectively.Inaddition,allgingerscanpromote intestinepropulsion,andFGBJexhibitsthestrongesteffect.
6‐Shogaoland6‐paradolarecharacterizedbyantimicrobialactivity(Galal,2008),whichmaybehelpfulforinhibitingbacterialinfectionintheprocessofgastricmucosalinjury.Additionally,6‐shogaol and 10‐gingerol have effective antioxidant and anti‐inflammatory properties(Dugasanietal.,2010),whichcanpreventoxygenfree radicalsfromactingonsulfenyltodegenerateproteinsandinactivate
enzymes, thus alleviating gastric mucosal damage. Moreover, the aforementioned antioxidant and anti‐inflammatory activities can ameliorategastrointestinalinjurybysuppressingtheinflammatory cascadereaction(Zhang,Ma,Gao,Sun,&Zhang,2017).Currently, there are almost no specific pharmacological researches on 12‐ shogaol.However,accordingtotheresultofthecurrentstudy,we inferthatitspharmacologicaleffectsarerelatedtositeslocalized inthedigestivetractandgastrointestinalinjurytoacertainextent. Besides,6‐shagoalmaybethemostcloselyrelatedsubstancetogastrointestinalfunction.
In conclusion, our findings demonstrate that when the ginger juiceFGJorDGJcouldbeusedfortheinhibitionofgastricmucosalinjury.Additionally,FGBJmaybethebestchoiceforpromoting gastrointestinalpropulsionandallkindsofgingersaresuitablefor preventing vomiting.
Overall,evidencehasbeenpresenteddemonstratingabasicun-
derstandingofdifferentgingerjuicesandtheirtherapeuticfeatures. Furtherstudiesshouldaimtoexplorethecorrelationsbetweenthe componentsofgingerjuicesandotherpharmacodynamicindicesto revealtheentireeffectsofgingerjuicesfortherapeuticregimens.
Onbehalfof,andhavingobtainedpermissionfromalltheauthors, theauthordeclaresthat:
- • Thestudy'sprotocolsandprocedureswereethicallyreviewedand approvedbyEthicsCommitteeonLaboratoryAnimalsofJiangxi UniversityofTraditionalChineseMedicine.Wecompliancewith LaboratoryAnimal‐Guidelineforethicalreviewofanimalwelfare andLaboratoryAnimal‐GeneralrequirementsforanimalexperimentofStateStandardofthePeople'sRepublicofChina.
- • Humantestingisunnecessaryinourstudy
CONFLICT OF INTEREST Theauthorsdeclarethattheyhavenoconflictofinterest.
ORCID Heng‐li Tong https://orcid.org/0000‐0002‐0456‐0000
Figures
Used In Evidence Reviews
Similar Papers
Food & function · 2013
A review of the gastroprotective effects of ginger (Zingiber officinale Roscoe).
World journal of gastroenterology · 2011
Effect of ginger on gastric motility and symptoms of functional dyspepsia.
Journal of ethnopharmacology · 2019
The role of gut microbiota for the activity of medicinal plants traditionally used in the European Union for gastrointestinal disorders.
Evidence-based complementary and alternative medicine : eCAM · 2015
The Effect of Ginger (Zingiber officinalis) and Artichoke (Cynara cardunculus) Extract Supplementation on Functional Dyspepsia: A Randomised, Double-Blind, and Placebo-Controlled Clinical Trial.
Nutrients · 2018
Herbs and Spices in the Treatment of Functional Gastrointestinal Disorders: A Review of Clinical Trials.
Methods and findings in experimental and clinical pharmacology · 2001