Descripción
Participant enrollment and randomization flow for the double-blind trial evaluating α-galactosidase in children with gas-related symptoms is depicted.
More Figures from This Paper
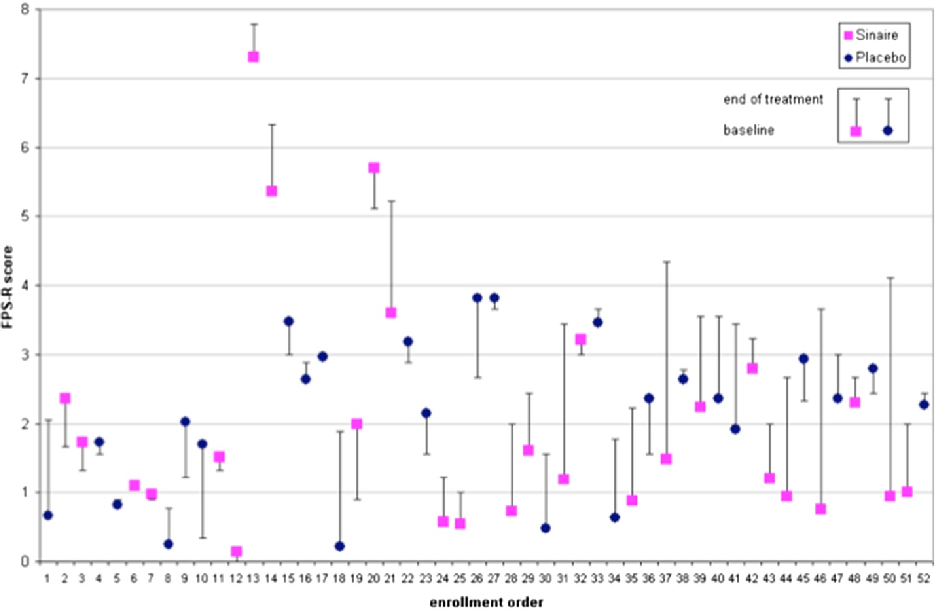
Figure 2
Frequency of gas-related episodes is compared between children receiving α-galactosidase and placebo over the treatment period, indicating changes in symptom burden.
chart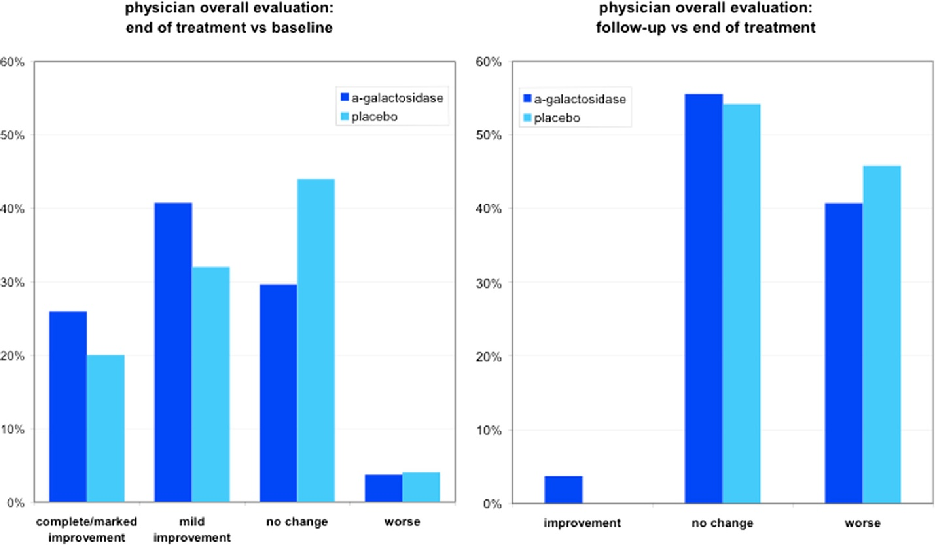
Figure 3
Overall symptom severity scores for gas-related complaints in children are tracked across the study timeline, comparing the α-galactosidase and placebo arms.
chartFigure 1
Flowchart
430 × 224px
· 29,6 KB
Source Paper
Efficacy and tolerability of α-galactosidase in treating gas-related symptoms in children: a randomized, double-blind, placebo controlled trial.Cite This Figure
 > Source: Giovanni Di Nardo et al. "Efficacy and tolerability of α-galactosidase in treating gas-related symptoms in." *BMC gastroenterology*, 2013. PMID: [24063420](https://pubmed.ncbi.nlm.nih.gov/24063420/)
<figure> <img src="https://pdfs.citedhealth.com/figures/24063420/54.png" alt="Participant enrollment and randomization flow for the double-blind trial evaluating α-galactosidase in children with gas-related symptoms is depicted." /> <figcaption>Figure 1. Participant enrollment and randomization flow for the double-blind trial evaluating α-galactosidase in children with gas-related symptoms is depicted.<br> Source: Giovanni Di Nardo et al. "Efficacy and tolerability of α-galactosidase in treating gas-related symptoms in." <em>BMC gastroenterology</em>, 2013. PMID: <a href="https://pubmed.ncbi.nlm.nih.gov/24063420/">24063420</a></figcaption> </figure>