The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation.
Study Design
- Studientyp
- Review
- Population
- Autoimmune disease patients (RA, MS, T1D, UC, psoriasis)
- Intervention
- The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation. None
- Vergleichsgruppe
- None
- Primärer Endpunkt
- Anti-inflammatory/immunomodulatory effects
- Wirkungsrichtung
- Positive
- Verzerrungsrisiko
- Unclear
Abstract
Inflammation is an integral part of autoimmune diseases, which are caused by dysregulation of the immune system. This dysregulation involves an imbalance between pro-inflammatory versus anti-inflammatory mediators. These mediators include various cytokines and chemokines; defined subsets of T helper/T regulatory cells, M1/M2 macrophages, activating/tolerogenic dendritic cells, and antibody-producing/regulatory B cells. Despite the availability of many anti-inflammatory/immunomodulatory drugs, the severe adverse reactions associated with their long-term use and often their high costs are impediments in effectively controlling the disease process. Accordingly, suitable alternatives are being sought for these conventional drugs. Natural products offer promising adjuncts/alternatives in this regard. The availability of specific compounds isolated from dietary/medicinal plant extracts have permitted rigorous studies on their disease-modulating activities and the mechanisms involved therein. Here, we describe the basic characteristics, mechanisms of action, and preventive/therapeutic applications of 5 well-characterized natural product compounds (Resveratrol, Curcumin, Boswellic acids, Epigallocatechin-3-gallate, and Triptolide). These compounds have been tested extensively in animal models of autoimmunity as well as in limited clinical trials in patients having the corresponding diseases. We have focused our description on predominantly T cell-mediated diseases, such as rheumatoid arthritis, multiple sclerosis, Type 1 diabetes, ulcerative colitis, and psoriasis.
Zusammenfassung
5 well-characterized natural product compounds (Resveratrol, Curcumin, Boswellic acids, Epigallocatechin-3-gallate, and Triptolide) are described, tested extensively in animal models of autoimmunity as well as in limited clinical trials in patients having the corresponding diseases.
Full Text
International Journal of
Molecular Sciences
Review
The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation
Kamal D. Moudgil 1,2,* and Shivaprasad H. Venkatesha 1,3
- 1 Department of Microbiology and Immunology, University of Maryland School of Medicine, Baltimore, MD 21201, USA
- 2 Baltimore VA Medical Center, Baltimore, MD 21201, USA
- 3 Vita Therapeutics, Baltimore, MD 21201, USA
* Correspondence: [email protected]
Citation: Moudgil, K.D.; Venkatesha, S.H. The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation. Int. J.
Mol. Sci. 2023, 24, 95. https:// doi.org/10.3390/ijms24010095
Academic Editors: Dong-Sung Lee and Wonmin Ko
Copyright: © 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https:// creativecommons.org/licenses/by/ 4.0/).
Abstract: Inflammation is an integral part of autoimmune diseases, which are caused by dysregulation of the immune system. This dysregulation involves an imbalance between pro-inflammatory versus anti-inflammatory mediators. These mediators include various cytokines and chemokines; defined subsets of T helper/T regulatory cells, M1/M2 macrophages, activating/tolerogenic dendritic cells, and antibody-producing/regulatory B cells. Despite the availability of many antiinflammatory/immunomodulatory drugs, the severe adverse reactions associated with their longterm use and often their high costs are impediments in effectively controlling the disease process. Accordingly, suitable alternatives are being sought for these conventional drugs. Natural products offer promising adjuncts/alternatives in this regard. The availability of specific compounds isolated from dietary/medicinal plant extracts have permitted rigorous studies on their disease-modulating activities and the mechanisms involved therein. Here, we describe the basic characteristics, mechanisms of action, and preventive/therapeutic applications of 5 well-characterized natural product compounds (Resveratrol, Curcumin, Boswellic acids, Epigallocatechin-3-gallate, and Triptolide). These compounds have been tested extensively in animal models of autoimmunity as well as in limited clinical trials in patients having the corresponding diseases. We have focused our description on predominantly T cell-mediated diseases, such as rheumatoid arthritis, multiple sclerosis, Type 1 diabetes, ulcerative colitis, and psoriasis.
Keywords: autoimmunity; boswellic acids; colitis; curcumin; diabetes; EGCG; multiple sclerosis; natural products; psoriasis; resveratrol; rheumatoid arthritis; triptolide
1. Introduction
Chronic inflammation is an integral component of autoimmune diseases, which are caused by dysregulation of the immune system, such that the host mounts an immune response to body’s own component proteins and/or cells [1–5]. A variety of antiinflammatory and immunomodulatory drugs are available for these disorders [6–10]. However, prolonged use of these drugs, which is necessary to keep persistent autoimmune reactivity in check, is frequently associated with severe adverse reactions and unavoidable high expense for the drugs. Newer drugs, such as biologics, may cause unintended systemic immune suppression, which increases the risk of severe infections and possibly, certain malignancies [6,7,10]. Furthermore, a proportion of autoimmune patients, for example, about 30–40% in case of rheumatoid arthritis (RA), fail to respond well to these drugs [11]. For these reasons, increasing proportion of patients with autoimmune diseases are resorting to the use of natural products [12–14]. Most popular among these products are plant-derived natural products, which have been part of various traditional systems of medicine across the world. Traditional Chinese medicine (TCM), Ayurvedic medicine, and indigenous African medicine are examples of such systems [11,13–19].
Int. J. Mol. Sci. 2023, 24, 95. https://doi.org/10.3390/ijms24010095 https://www.mdpi.com/journal/ijms
Accordingly, a variety of plant extracts and their bioactive components have extensively been studied for their anti-inflammatory and immunomodulatory activities, and for their applications in the treatment of autoimmune diseases [12–14]. These natural products are first tested in various animal models of autoimmunity and subsequently in limited clinical trials in patients with those diseases. In this article, we have focused our discussion to 5 purified compounds derived from plant extracts. These include, Resveratrol [19–26], Curcumin [12,27–31], Boswellic acids [15,16,32,33], Epigallocatechin-3-gallate (EGCG) [34–40], and Triptolide [18,41–46].
We have described below the effects of these natural products on inflammation in predominantly T cell-driven autoimmune diseases: rheumatoid arthritis (RA), multiple sclerosis (MS), type 1 diabetes (T1D), ulcerative colitis (UC; colitis), and psoriasis. For space constraints, limited number of studies for each natural product compound are discussed below.
2. Autoimmune Diseases Result from a Breakdown of Self-Tolerance Leading to the Generation of Immune Response to Self-Antigens
Autoimmune diseases are manifestations of a series of events that involve an interplay between genetic and environmental factors, leading to a breakdown of self-tolerance and activation of potentially self-reactive T cells [4,5,47–49]. In healthy individuals, such T cells exist in the mature T cell repertoire, but their activation is kept in check by a variety of peripheral tolerance mechanisms [50]. These mechanisms include, T cell ignorance, lack of co-stimulation, activity of different types of regulatory T cells, and others [51,52]. A failure of these mechanisms results in uncontrolled activation of T cells directed against various self-antigens implicated in different autoimmune diseases [53]. These activated T cells then stimulate macrophages and/or provide help to the B cells, leading to propagation of anti-self immune responses. A variety of cytokines, chemokines and biochemical mediators of inflammation released by these immune cells trigger cascades of inflammatory pathways, tissue damage and dysfunction, and clinical manifestations [53,54]. Autoimmune diseases can either be organ-specific or systemic. Rheumatoid arthritis (RA), multiple sclerosis (MS), type 1 diabetes (T1D), ulcerative colitis (colitis) (a component of inflammatory bowel disease or IBD), and psoriasis are examples of major human autoimmune diseases [55–60] (Table 1). The organs most affected in these diseases include the synovial joints in RA, the central nervous system (CNS) in MS, the pancreatic β-islets in T1D, colon in UC, and skin in psoriasis [57,58,60–62]. In addition, there may be varying levels of effects on other organs, as commonly observed in RA.
Table 1. Salient features of the major T cell-mediated human autoimmune diseases.
Name Main Organs Affected *
Genetic Susceptibility
Self-Antigens Targeted
Drugs Used References
(e.g., HLA allele) (representative) (examples)
Rheumatoid arthritis (RA)
Synovial joints
DRB1 * 01/* 04/
CII, Hsp65 NSAIDs, DMARDs
[55]
* 10
Multiple sclerosis (MS)
Central nervous system
DRB1 * 1501 MBP, PLP IFN-β,
[63,64]
Glatiramer, Fingolimod
Type 1 Diabetes
Pancreatic β-islets
DRB1 * 0301/
Insulin, GAD65 Insulin [65]
* 0401
(T1D) Ulcerative colitis (UC)
Colon HLA ADCY7
Gut microbial antigens
Anti-TNF [66,67]
Psoriasis Skin HLA-Cw6 (PSORS1)
Cathelicidin LL-37 ADAMTSL5
Anti-TNF, MTX
[68]
* other organs may also be affected to varying extent in different diseases. HLA = human leukocyte antigen, CII- type II collagen, Hsp65 = heat-shock protein 65, MBP = myelin-basic protein, PLP = proteolipid protein, GAD = glutamic acid decarboxylase, NSAIDs= non-steroidal anti-inflammatory drugs, DMARDs = diseasemodifying anti-rheumatic drugs, IFN-beta= interferon-β; ADCY7 = adenylate cyclase 7 gene; LL-37 = cathelicidin peptide; ADAMTSL5 = melanocyte antigen, a disintegrin and metalloprotease domain containing thrombospondin type 1 motif-like 5; MTX = methotrexate; TNFα= tumor-necrosis factor alpha.
3. The Pathogenesis of Autoimmunity Involves an Interplay between Genetic andEnvironmental Factors
The precise etiology of autoimmune diseases is not yet fully known. It is clear that there is genetic susceptibility to several autoimmune diseases [49,65]. The major histocompatibility complex (MHC) locus is one of the important genetic factors in this regard (Table 1). There also is evidence for environmental factors, among which microbial infections are considered to be of high significance [5,65]. Microbes can contribute to the activation of self-reactive T cells via different mechanisms, including upregulation of co-stimulation in antigen-presenting cells (APCs), enhanced processing of certain antigenic determinants under inflammatory conditions, and antigenic mimicry (e.g., conformational/sequence resemblance) between microbial and host antigenic determinants (molecular mimicry) [55,56,59,63,69]. The pathogenic T cells in autoimmune diseases target subsets of self-antigens. Multiple self-antigens have been implicated in each of the abovementioned autoimmune diseases (RA, MS, T1D, colitis and psoriasis) [57,58,60–62,66–68,70] (Table 1).
4. Autoimmunity Is Characterized by an Imbalance of Cytokines and T Cell Subsets,and Aberrant Antibody Responses
Naïve T cells that are activated by a particular self-antigen can acquire different phenotypes depending on the local milieu, particularly the type of cytokines present. Accordingly, naïve T cells can differentiate into distinct T helper (Th) type, such as Th1 or Th17 cells, which can mediate pathogenic effector functions [53,71,72]. Under another set of conditions, a naïve T cell can develop into Th2 or T regulatory cells (Treg), which can control the activity of pathogenic Th cells [53,64]. The Th cells secrete prototypic cytokines and express a distinct transcription factor [72]. Among the cytokines, interferon-γ (IFN-γ) and IL-17 are shown to promote inflammation, whereas IL-4 and IL-10 inhibit inflammation [52]. An imbalance between pro-inflammatory and anti-inflammatory cytokines, as well as that between pathogenic (Th1/Th17) and protective (Th2/Treg, respectively) T cells have been reported in various autoimmune diseases [64,73,74]. A similar imbalance can occur in macrophage subsets, such as pro-inflammatory M1 macrophages versus anti-inflammatory M2 macrophages; in dendritic cells (DCs), such as activating DCs versus tolerogenic DCs;
and in B cells, such as antibody-producing B cells versus regulatory B cells that secrete IL-10 (known as B10 cells) (Figure 1). Accordingly, a deliberate re-setting of the balance between these cytokines (e.g., IFN-γ/IL-4 balance) and immune cell subsets (e.g., Th17/Treg balance) is one of the approaches under development for the treatment of these diseases [75]. This principle is also being exploited by using natural products compounds (Figure 1).
containing citrulline. Post-translational modification of antigen has also been implicated in the pathogenesis of T1D [77].
Figure 1. Immunomodulation by natural product compounds involves an altered balance between subsets of diverse immune cell types. The corresponding pathogenic (pro-inflammatory) (Right panel) versus protective (anti-inflammatory/immunomodulatory) (Left panel) immune cell subsets are shown: Th1 vs. Th2, Th17 vs. Treg, M1 vs. M2, activating vs. tolerogenic DC, and antibodyproducing vs. regulatory B cells, respectively. (Abbreviations (Top to bottom)): Th = T helper cell; Treg = T regulatory cells; M1 = M1-type macrophage; M2 = M2-type macrophage; DC = dendritic cells (Right panel, activating DC; Left panel, tolerizing DC); B10 = B cells secreting IL-10 (a regulatory B cell subset); B cell = activated B cell for antibody production.
A dysregulation of cytokines and Th cells in turn can influence B cell activation and the type of antibody isotype produced. Thus, both cellular and humoral arms of the immune response can be affected in this manner. For simplicity, autoimmune diseases may be classified as T cell-mediated or antibody-mediated, but in reality, the disease manifestation involves the contribution of both arms of the immune system to different extents. For example, T1D is characterized by anti-insulin antibodies. In RA, rheumatoid factors (RFs) have long served as a disease marker, but their pathogenic significance is not fully validated. However, there is a correlation between smoking and RA, which involves anti-citrullinated protein antibodies (ACPAs). Here, smoking has been invoked in inducing citrullination, a type of post-translational modification, of certain self-antigens in which an arginine residue is converted into citrulline [76]. This renders that protein immunogenic because the host is tolerant to the native protein, but not to the same protein containing citrulline. Post-translational modification of antigen has also been implicated in the pathogenesis of T1D [77].
5. Animal Models Have Contributed Significantly to Understanding of the Pathogen-esis and Treatment of Human Autoimmune Diseases
Understanding of the disease process in autoimmunity has been aided significantly by studies in animal models (Table 2). These models have also been the mainstay of testing of new drug candidates. There is no single animal model that can fully replicate all clinical features of a particular human autoimmune disease. Different rodent models are available for these diseases [57,58,60–62,68,78–80] (Table 2). Of these, some are experimentally induced, while others are spontaneous in origin. These include, for example, adjuvant-induced arthritis (AA) and collagen-induced arthritis (CIA) for RA; experimental autoimmune encephalomyelitis (EAE) for MS; non-obese diabetic (NOD) mice for T1D, chemical, spontaneous or T cell transfer models of colitis for ulcerative colitis (UC), and Imiquimodinduced psoriasis [57,58,60–62,68,78–80] (Table 2). Predominantly T cell-driven effector mechanisms are operative in these human autoimmune diseases.
5. Animal Models Have Contributed Significantly to Understanding of the Pathogenesis and Treatment of Human Autoimmune Diseases
Understanding of the disease process in autoimmunity has been aided significantly by studies in animal models (Table 2). These models have also been the mainstay of testing of new drug candidates. There is no single animal model that can fully replicate all clinical features of a particular human autoimmune disease. Different rodent models are available for these diseases [57,58,60–62,68,78–80] (Table 2). Of these, some are experimentally induced, while others are spontaneous in origin. These include, for example,
Table 2. Animal models of the major T cell-mediated human autoimmune diseases.
Strain of Rat/Mouse Mode of Induction References Lewis rats Mtb (H37Ra) [78]
DBA/1 mice Type II collagen (CII) [35,79]
adjuvant-induced arthritis (AA) and collagen-induced arthritis (CIA) for RA; experimental autoimmune encephalomyelitis (EAE) for MS; non-obese diabetic (NOD) mice for T1D, chemical, spontaneous or T cell transfer models of colitis for ulcerative colitis (UC), and Imiquimod-induced psoriasis [57,58,60–62,68,78–80] (Table 2). Predominantly T cell-driven effector mechanisms are operative in these human autoimmune diseases.
Table 2. Animal models of the major T cell-mediated human autoimmune diseases.
Disease Model Strain of Rat/Mouse Mode of Induction References
Adjuvant-induced Lewis rats Mtb (H37Ra) [78] arthritis (AA)
Collagen-induced DBA/1 mice Type II collagen (CII) [35,79] arthritis (CIA) HLA.DR4 mice CII
Lewis rats CII
Experimental autoimmune Lewis rats MBP, MBP peptide [22,61,62] encephalomyelitis (EAE) SJL mice PLP, PLP peptide
(SWR x SJL) F1 mice PLP, PLP peptide C57BL/6 mice MOG, MOG peptide
Type 1 diabetes (T1D) C57BL/6 mice STZ [27,57] NOD mice Spontaneous
Colitis C57BL/6, BALB/c mice DSS [20,27] (Ulcerative colitis; UC) C57BL/6, BALB/c mice TNBS
IL-10-deficient mice Spontaneous RAG-deficient mice CD4+ CD45RBhi transfer
Psoriasis BALB/c Imiquimod [80]
Mtb—heat-killed Mycobacterium tuberculosis H37Ra, CII- type II collagen, MBP—Myelin basic protein, PLP—Proteolipid protein, MOG—Myelin oligodendrocyte protein, DSS—Dextran sodium sulphate, STZStreptozotocin, TNBS—2,4,6-trinitrobenzene sulfonic acid, RAG—recombinase activating gene.
6. The Use of Plant-Derived Natural Product Compounds for the Treatment of Autoimmune Diseases
The progression of initial autoimmune reactivity to subsequent clinical disease involves inflammation driven by multiple cellular and molecular mediators [53,54,81]. The role of the T cells, macrophages and B cells is briefly discussed above. Various biochemical (e.g., leukotrienes) and immunological (e.g., cytokines) mediators released by these cells, along with others (e.g., complement components), lead to autoimmune inflammation. When uncontrolled, such inflammation can lead to tissue damage and dysfunction. Thus, the control of inflammation is one of the main tenets of treatment of autoimmunity. In this regard, we have described below 5 representative natural product compounds that have extensively been studied for controlling inflammation and resetting the imbalanced immune system in autoimmune diseases. These include, Resveratrol [19–26], Curcumin [12,27–31], Boswellic acids [15,16,32,33], Epigallocatechin-3-gallate (EGCG) [34–40], and Triptolide [18,41–46]. A summary of these is presented below (Table 3), which also lists the biochemical molecules and pathways targeted by these 5 natural product compounds. The biochemical pathways themselves are outlined in Figure 2. We have focused mostly on the effects of natural product compounds on inflammation in predominantly T cell-driven autoimmune diseases. For space constraints, limited number of studies for each natural product compound are described below.
Int. J. Mol. Sci. 2023, 24, 95 = signal transducer and activator of transcription; SIRT1 = Sirtuin 1; SOCS = suppressor of cytokine6 of 32 signaling; TFIIH = transcription factor IIH; VEGF = vascular endothelial growth factor; XPB = XPB
Figure 2. A summary of the main biochemical pathways involved in autoimmune inflammation that are targeted by natural product compounds. Representative mediators of inflammation following modulation of gene expression are shown. (Abbreviations: AKT = an oncogene, also known as protein kinase B or PKB; AMPK = adenosine monophosphate-activated protein kinase; COX; cyclooxygenase, DAG, diacyl glycerol; ERK, extracellular signal-regulated kinase; IKK, IκB kinase; IL = interleukin; IP3, inositol triphosphate; JAK, janus kinase; JNK, c-Jun N-terminal kinase; LO (or LOX), lipoxygenase; MAPK, mitogen-activated protein kinase; MCP-1 = monocyte chemoattractant protein; MMP = matrix metalloproteinase; mTOR, mammalian target of rapamycin; MyD88, myeloid differentiation primary response gene 88; NEMO, NF-κB essential modulator; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B cells; Nrf-2, nuclear factor erythroid 2-related factor 2; PDGF = platelet-derived growth factor; PI3K, phosphatidylinositol-3-kinase; PIP, phosphatidylinositol bisphosphate; PLA2, phospholipase A2; PLCγ, phospholipase C gamma; RANTES =
Figure 2. A summary of the main biochemical pathways involved in autoimmune inflammation that are targeted by natural product compounds. Representative mediators of inflammation following modulation of gene expression are shown. (Abbreviations: AKT = an oncogene, also known as protein kinase B or PKB; AMPK = adenosine monophosphate-activated protein kinase; COX; cyclooxygenase, DAG, diacyl glycerol; ERK, extracellular signal-regulated kinase; IKK, IκB kinase; IL = interleukin; IP3, inositol triphosphate; JAK, janus kinase; JNK, c-Jun N-terminal kinase; LO (or LOX), lipoxygenase; MAPK, mitogen-activated protein kinase; MCP-1 = monocyte chemoattractant protein; MMP = matrix metalloproteinase; mTOR, mammalian target of rapamycin; MyD88, myeloid differentiation primary response gene 88; NEMO, NF-κB essential modulator; NF-κB, nuclear factor kappa-light-chainenhancer of activated B cells; Nrf-2, nuclear factor erythroid 2-related factor 2; PDGF = plateletderived growth factor; PI3K, phosphatidylinositol-3-kinase; PIP, phosphatidylinositol bisphosphate; PLA2, phospholipase A2; PLCγ, phospholipase C gamma; RANTES = Regulated upon Activation, Normal T Cell Expressed and Presumably Secreted; Ras = Ras oncogene; ROS = reactive oxygen species; STAT, signal transducer and activator of transcription; SIRT1 = Sirtuin 1; TRIF, TIR-domaincontaining adapter-inducing interferon-β; and VEGF = vascular endothelial growth factor).
Table 3. Select natural product compounds from plants: their origin, basic chemical characteristics, and targeted molecules and pathways for the treatment of autoimmune diseases.
Product Plant Sources Chemistry # Targets/Pathways $,* References Resveratrol Vitis vinifera, Stilbene AMPK, COX-2, MAPK [19–26]
Polygonum C14H12O3 NF-kB, Nrf2, cuspidatum MW 228.24 PI3K/Akt/mTOR,
SIRT-1, STAT3, AhR Wnt/β-catenin, PPARγ,
Curcumin Curcuma longa Diarylheptanoid NF-kB, COX-2, AMPK [12,27–31] (beta-diketone) STAT3, Nrf2, HO-1 C21H20O6 MAPK, JNK MW 368.4 ROS metabolic pathway
Table 3. Cont.
Product Plant Sources Chemistry # Targets/Pathways $,* References
Boswellic Boswellia Pentacyclic 5-LO, Akt, COX-2 [15,16,32,33] Acids serrata terpenoids MAPK, NF-kB, Nrf2 (e.g., AKBA) C32H48O5 STAT3, ERK1/2
MW 512.7 MMP9, VEGF
EGCG Camellia sinensis Catechin COX-2, NF-kB, AMPK [34–40] C22H18O11 AP-1, MMP 2 & 9, MW 458.4 HIF-1a, iNOS
PI3K/Akt/mTOR, STAT3, MAPK
Triptolide Tripterygium Diterpenoid XPB/TFIIH [18,41–46] wilfordii Hook F. triepoxide NF-kB, MAPK (TwHF) C20H24O6 IL-6/STAT3, SOCS3
MW 360.4
# MW = molecular weight. $ Targets in bold face font = upregulated; Targets in regular font = downregulated (this direction of change is representative and based on common anti-inflammatory activity of these natural product compounds, but subtle differences can be observed under different contexts of the dose, timing and underlying pathology involved). * Abbreviations: AhR = aryl hydrocarbon receptor; AKBA = acetyl-11-keto-betaboswellic acid; AMPK = adenosine monophosphate-activated protein kinase; Akt = an oncogene, also known as protein kinase B or PKB; AP-1 = activator protein-1; COX = cyclooxygenase; EGCG = epigallocatechin-3gallate; ERK = extracellular signal-regulated kinase; HIF= hypoxia-inducible factor; HO-1 = heme oxygenase-1; IKK = IκB kinase; iNOS = inducible nitric oxide synthase; JAK, janus kinase; JNK = c-Jun N-terminal kinase; LO (or LOX), = lipoxygenase; MAPK= mitogen-activated protein kinase; MMP = matrix metalloproteinase; mTOR = mammalian target of rapamycin; NF-κB = nuclear factor kappa-light-chain-enhancer of activated B cells; Nrf-2 = nuclear factor erythroid 2-related factor 2; PI3K = phosphatidylinositol-3-kinase; PPARγ = peroxisome proliferator activated receptor γ; ROS = reactive oxygen species; STAT = signal transducer and activator of transcription; SIRT1 = Sirtuin 1; SOCS = suppressor of cytokine signaling; TFIIH = transcription factor IIH; VEGF = vascular endothelial growth factor; XPB = XPB subunit of TFIIH.
7. Resveratrol
- 7.1. Immune Cells/Response
- 7.1.1. T Cell Activity and T Cell Subsets
Resveratrol is effective in controlling inflammation by targeting the T cells, by altering T cell differentiation, and by inhibiting the release of pro-inflammatory cytokines and other (biochemical) mediators of inflammation [82]. These outcomes are attributed to its effects on sirtuin-1, adenosine monophosphate-activated protein kinase (AMPK; energy sensor in cells), and nuclear factor-κB (NF-kB) (Table 3) [82,83]. Resveratrol activates sirtuin-1 (a deacetylase) and inhibits acetylation of its target protein p65/relA, which then inhibits NF-kB activity, resulting in reduced production of mediators of inflammation. Similarly, resveratrol targets AMPK, which is an activator of sirtuin-1 via regulating the cellular levels of nicotinamide adenine dinucleotide (NAD+). Another target of sirtuin-1 is peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), which is activated via deacetylation [82]. Interestingly, AMPK is also a target of other natural products, such as curcumin [84], EGCG [85], triptolide [86] and Soy isoflavones [87], demonstrating one of the key anti-inflammatory mechanisms of action of several plant-derived natural compounds.
Th17 is a prominent T cell subset targeted by resveratrol. Resveratrol is an agonist of sirtuin-1 that can cause deacetylation of STAT3 and inhibit its migration into the nucleus to interrupt activation of retinoid orphan receptor gamma t (RORγt) and IL-17A (henceforth, referred to as IL-17) production [21]. This leads to altered balance between Th17 vs. Treg. Another mode of action of resveratrol involves modulation of ligand-induced activation of aryl hydrocarbon receptor (AhR), which in turn suppresses Th17 activity [21]. RORγt also has inhibitory effect on Th1 differentiation and thereby, resveratrol also alters Th1/Th2 balance. Its inhibitory effects on both the pro-inflammatory T cell subsets mentioned above tilt the balance in favor of anti-inflammatory (Th2) and immunoregulatory (Treg) activities. The re-setting of the Th17/Treg cell balance by resveratrol was also evident from a study on immune cells of a patient with a different autoimmune disease, immune thrombocytopenic
purpura (ITP) [88]. The observed effects of Resveratrol were attributed primarily to its inhibitory effect on AhR. This was further validated by reversal of the effect of resveratrol by an agonistic ligand for AhR, 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) [88].
Resveratrol was found to be safe and well-tolerated when administered to healthy male individuals [89]. There was an increase in γδ T cells and Treg in circulation as well as antioxidant activity of plasma when compared with control subjects [89]. However, there was a reduction in pro-inflammatory cytokines TNF-α and MCP-1.
- 7.1.2. Macrophages and Dendritic Cells (DC)
- 7.1.3. B Cells, Plasma Cells and Autoantibodies
Resveratrol has been shown to have significant effects on the B cells, plasma cells and autoantibody production, all of which play a role in the pathogenesis of antibody/immune complex-mediated diseases, such as lupus [82]. Resveratrol treatment affords protection against lupus, including lupus nephritis, in part via activation of sirtuin-1 and upregulation of inhibitory Fc receptor (FcγRIIB) [82]. The former controls B cell proliferation and autoantibody production, while the latter reduces the activation and number of B cells/ plasma cells in the spleen and bone marrow as a result of apoptosis [82].
- 7.2. Rheumatoid Arthritis (RA)
- 7.2.1. RA Patients
- 7.2.2. Animal Models of RA
A study in the murine CIA model showed that resveratrol can inhibit clinical arthritis as well as bone loss [97]. Both prophylactic (before disease induction) and therapeutic (after disease onset) regimens of resveratrol administration showed anti-arthritic effects. In parallel, significant effects on the Th17 cell response and anti-collagen antibody levels were observed, with both showing reduced levels/activity compared with controls [97]. In another study in the same model, resveratrol effectively suppressed the development of arthritis in mice [98]. It was associated with inhibition of T cell activation and production of
cytokines, which were attributed to resveratrol-induced activation of Sirt1, but reduction of c-Jun acetylation and its migration into the nucleus [98]. Resveratrol treatment of rats with CIA resulted in reduction of paw inflammation, bone loss, cartilage damage and antibody biomarkers (RF and ACPA) [99]. However, there was an increase in the anti-inflammatory cytokine, IL-4.
Resveratrol treatment of arthritic rats having adjuvant arthritis (AA) caused a reduction in paw swelling and biochemical mediators of inflammation, including COX-2 in synovial tissue and prostaglandin E2 (PGE2) level in serum of rats [100]. Prophylactic aspect of resveratrol action against arthritis development was tested in an antigen-induced arthritis (AIA) model [101], and beneficial effects were observed when assessed from paw swelling (clinical effect) along with histology and immuno-histochemistry of paws (reduced synovial hyperplasia and markers of inflammation) [101].
- 7.3. Multiple Sclerosis/EAE
- 7.4. Type 1 Diabetes (T1D)
- 7.4.1. Diabetes Patients A pilot study on thirteen T1D patients given resveratrol for two months showed beneficial effects as measured by a panel of markers that document the anti-diabetic and antioxidant effects of this natural product [107]. However, there was not much effect on the levels of insulin and markers of inflammation and functions of liver and kidney. These are encouraging results for further exploration of anti-diabetic effects of resveratrol in a large, controlled study.
- 7.4.2. Animal Models of T1D
A study in streptozotocin (STZ)-induced diabetes in rats showed that resveratrol afforded protection against T1D development (prophylactic effect) [108]. This effect involved
inhibition of apoptosis of pancreatic beta islet cells and suppressed activation of caspase 3 and poly (ADP-ribose) polymerase (PARP). In the STZ model in mice, resveratrol ameliorated the metabolic (biochemical and oxidative stress) aberrations as well as dysregulated immune cell infiltration into pancreatic islets [109]. In another study, resveratrol treatment of diabetic mice reduced the infiltration of macrophages into pancreatic islets as well as the expression of CXCL16/ NF-kB p65 in the islets and spleen compared with untreated mice [109]. One of the complications of T1D involves the reproductive system, specifically reduced spermatogenesis and poor quality of sperms, which affects male fertility. Resveratrol improved both these aspects relating to sperm generation and function by inhibiting sperm DNA damage in rats [110,111].
Resveratrol was also effective in protection against development of T1D in non-obese diabetic (NOD) mouse model [112]. This effect correlated with reduced migration of Th17 cells and macrophages from the peripheral lymph nodes into pancreatic islets. Chemokine (C-C motif) receptor (CCR6) expressed on the surface of both these cell types was one of the targets of resveratrol action [112].
From the foregoing description, it might appear that Th17 and IL-17 are always pathogenic and IL-10 is a typical immunomodulatory cytokine. However, certain microbial components (e.g., Bacillus Calmette-Guérin (BCG) and complete Freund’s adjuvant (CFA)) that can induce IL-17 response may also be protective against inflammation, as elaborated in animal models of T1D [81,113]. In this case, the T cells producing IL-17 and IL-10 (Treg17) show regulatory effects in T1D. Similarly, IL-10 has also been shown to have a pathogenic role under certain disease conditions [114]. Therefore, the outcome of the activity of certain T cell subsets and cytokines can differ in various disease conditions, depending on the timing and level of immune response.
- 7.5. Colitis
- 7.6. Psoriasis
- 7.7. Safety and Adverse Effects
The use of resveratrol to control inflammation and disease pathology in various autoimmune diseases, both animal models and patients/volunteers, was generally found to be safe and well-tolerated [89,122,123]. Although uncommon, in some cases, the side effects observed include fatigue, headache, dizziness, gastrointestinal symptoms (e.g.,
nausea, indigestion, abdominal pain, or vomiting), skin rash or fever. However, there was no evidence of elevated enzymes in blood or other features of hepatotoxicity associated with the use of resveratrol.
8. Curcumin
- 8.1. Inflammation/Immune Response
- 8.1.1. T Cell Activity and T Cell Subsets
- 8.1.2. Cytokines
- 8.1.3. Dendritic Cells (DCs)
Curcumin reduced the production of pro-inflammatory cytokines IL-12 and IL-23 by DCs and the Th17 response via its action on Nrf2-sensitive heme oxygenase 1 and STAT3 phosphorylation [137]. (Nrf2 is a transcription factor that plays an important role in antioxidant pathways.) Furthermore, unlike the immune-activating function of DCs, tolerogenic DCs manifest anti-inflammatory and immunomodulatory activities, which suppress T celland cytokine-driven immune responses. Curcumin facilitates the generation of tolerogenic DCs in response to certain antigens, which otherwise would have induced an immune response [138]. Curcumin mediates this effect via reducing the expression of co-stimulatory and adhesion molecules on DCs and inhibiting their antigen presentation and cellular migration functions [138].
- 8.2. Rheumatoid Arthritis (RA)
- 8.2.1. RA Patients
Pilot clinical studies in RA patients have revealed beneficial effects of curcumin on clinical symptoms, including joint swelling, pain and discomfort [139–141]. These include randomized and controlled studies. A combination of mechanisms relating to curcumin’s anti-inflammatory, antioxidant, and immunomodulatory activities contribute to this antiarthritic effects. Several of these mechanisms are also discussed below in the context of curcumin’s effects in animal models of RA.
- 8.2.2. Animal Models
- 8.3. Ankylosing Spondylitis (AS)
- 8.4. Multiple Sclerosis/EAE
- 8.5. T1D
Several studies in murine/rat models of STZ-induced T1D [158–161] and diabetes in NOD mice [162] have shown the anti-diabetic effects of curcumin. Additional information about the protective effect of curcumin was obtained by using insulinoma cells lines, such as MIN6 [163]. In animal models, curcumin reduced blood glucose levels, improved lipid profile, reduced lipid peroxidation, and improved pancreatic beta cell functions. Taken together, various mechanisms unveiled in these studies demonstrate the anti-inflammatory, antioxidant, and immunomodulatory activities of curcumin [158–162]. For example, cur-
cumin reduced pro-inflammatory cytokines such as TNF-α; decreased ROS; inhibited NF-kB; and decreased Th1 differentiation and activation status of DCs. In cell line-based assays, curcumin reduced apoptosis of beta islet cells by reducing oxidative stress and endoplasmic reticulum (ER) stress [163]. In the animal models, besides its beneficial effects against general metabolic aspects of T1D, curcumin also can provide protection against various complications associated with T1D, such as cardiomyopathy [164], nephropathy [165], and skeletal muscle atrophy [161]. Taken together, these studies have set the stage for clinical testing of curcumin in the treatment of human T1D and its complications.
- 8.6. Colitis
- 8.7. Psoriasis
- 8.7.1. Psoriasis Patients
- 8.7.2. Animal Models
Studies in imiquimod-induced murine model of psoriasis have validated the beneficial effects of curcumin in reducing the severity of skin lesions and associated symptoms [171–174]. Taken together, this protective effect was attributed to reduction of pro-inflammatory cytokines IL-1β and IL-6 [171]; inhibition of IL-6/STAT3 signaling pathway [173]; and suppression of NLRP3 expression, reduction in inflammation caused by cytokines IL-18 and IL-22, and inhibition of IL-22-induced STAT3 activation [174].
- 8.8. Safety and Adverse Effects
- 9. Boswellic Acids
- 9.1. Inflammation/Immune Response
Boswellic acids and its parent herb have been shown to be beneficial in several chronic inflammatory/autoimmune diseases, such as RA, ulcerative colitis, and diabetes [32,177,178]. Boswellic acids, a mixture isolated from gum resin of boswellia serrata, include four major pentacyclic triterpene acids: beta-boswellic acid, 3-acteyl-betaboswellic acid, 11-keto-beta-boswellic acid (KBA), and acetyl-11-keto-beta-boswellic acid (AKBA) [179]. Among these, KBA and AKBA are widely studied for their biological and medicinal effects [33]. Boswellic acids affect the immune system in multiple ways. For example, boswellic acids inhibit pro-inflammatory cytokines (e.g., TNF-α, IL-1β, IL-2, IL-4, IL-6 and IFN-γ), increase phagocytosis of macrophages, alter antibody production, and inhibit the classical complement pathway [177,178]. The mode of action of boswellic acids differs from that of NSAIDs. Inhibition of 5-lipooxygenase (5-LO or 5-LOX) leading to reduced production of leukotrienes comprises a major activity of boswellic acids, whereas inhibition of prostaglandin synthesis is rather a minor effect (Table 3, Figure 2) [180,181]. Another prominent mode of action of boswellic acids involves inhibition of NF-kB activation [32,182].
T Cell Activity/T Cell Subsets
Boswellic acids inhibited the production of pro-inflammatory IL-2 and IFN-γ (both Th1-related cytokines) but upregulated the production of anti-inflammatory/ immunomodulatory IL-4 and IL-10 (both Th2-related cytokines) by murine splenic T cells [183]. This Th1 to Th2 switch contributes to the anti-inflammatory and anti-arthritic effects of boswellic acids. In addition, AKBA has been shown to inhibit both Th17 differentiation and the production of cytokine IL-17 by these cells [184]. Two of the mechanisms relating to Th17 differentiation were identified as the targets in this process: inhibition of IL-1β signaling and STAT3 phosphorylation [184].
- 9.2. Rheumatoid Arthritis
- 9.2.1. RA Patients
Boswellic acids have anti-inflammatory properties and have shown anti-arthritic activity in pilot clinical trials in RA patients [180,185]. Their use was associated with mild side effects. Similarly, boswellic acids have been used to relieve pain in patients with osteoarthritis [182], which is known to have a component of inflammation, although of much lower level than in RA. Furthermore, boswellic acids combined with glucosamine showed synergistic effect, suggesting that similar drug combinations can be explored for clinical applications in arthritis patients and possibly other chronic inflammatory disorders [179,186].
- 9.2.2. Animal Models of RA
Boswellic acids are known to mediate anti-inflammatory activity when administered systemically. Their similar efficacy after topical application has also been reported in various rodent models of inflammation, such as arachidonic acid and croton oil-induced
ear edema and carrageenan-induced paw edema models in mice, and adjuvant-induced arthritis model in rats [179,186].
Boswellia extract containing boswellic acids was tested for anti-inflammatory/antiarthritic activity in different assay systems, including RAW 264.7 macrophages, human peripheral blood mononuclear cells stimulated with bacterial lipopolysaccharide (LPS), and the rat CIA model [187]. The following mediators were found to be reduced in these assays [187]: TNF-α, IL-6, nitric oxide, and COX-2 secretion; phosphorylated-NFκB (P65); collagenase, elastase, hyaluronidase, and matrix-degrading enzymes; reactive oxygen species (ROS); circulating anti-collagen antibodies; C-reactive protein (CRP) and prostaglandin E2 (PGE2); and cartilage oligomeric matrix protein (COMP). However, increased levels of hyaluronan were found in the synovial fluid.
Boswellic acids were shown to possess anti-inflammatory activity against AA in rats [188]. Poor solubility and bioavailability of boswellic acids has become a limitation in its use for arthritis and other inflammatory conditions [189]. Attempts are being made to develop novel formulations to enhance their therapeutic index. Micellar formulation has shown improved efficacy over regular preparation of boswellic acids, when tested in the rat AA model [144]. Another study provided insight into increased lysosomal stability following administration of boswellic acids to rats with AA [190,191]. Boswellic acids also revealed their anti-arthritic activity in bovine serum albumin (BSA)-induced arthritis in rabbits, when administered orally or injected into knee joints.
- 9.3. Type 1 Diabetes
- 9.4. Multiple Sclerosis/EAE
- 9.5. Colitis
- 9.5.1. Colitis Patients
Patients with colitis (UC) were treated with Boswellia serrata gum resin and then various disease-related parameters were assessed [197]. These included, stool examination, rectal biopsy histology and scan microscopy, and blood chemistry and cell counts [197]. All these parameters improved following above treatment. Remission was noted in 82% of treated patients [197]. In another study by the same group, a similar treatment of colitis patients resulted in improvement of clinical symptoms as well as test results in 18 of 20 patients [198]. Of the 20 patients, 14 had remission from the disease symptoms.
- 9.5.2. Animal Models
A drug delivery system targeted to the colon was developed and tested for the delivery of boswellic acids in a rat model of colitis [199]. One of the objectives of this approach was to limit the action of this natural product to the colon, without any collateral unwanted effects on other gastrointestinal components. This approach resulted in increased bioavailability of boswellic acids in the colon, while sparing other organs such as the stomach, pancreas and liver. Another study revealed the effect of a semi-synthetic AKBA on downregulation of the expression of P-selectin in the colonic tissue, as well as inhibition of P-selectin-mediated cellular interactions with endothelial cells of venules in colonic lesions [200]. Intravital microscopy was used to study these cellular interactions.
- 9.6. Psoriasis
- 9.7. Safety and Adverse Effects
The use of AKBA or other common boswellic acids has been found to be generally safe [180,185]. Adverse effects were rarely observed in clinical trials or animal model studies [203,204]. However, in some cases, gastrointestinal symptoms (e.g., nausea, diarrhea or abdominal pain) have been reported, but these were mild and manageable.
10. EGCG
- 10.1. Rheumatoid Arthritis
Green tea polyphenols (GTP), when administered orally to mice, resulted in reduced incidence as well as severity of CIA [35]. This was associated with reduced expression levels of COX-2, TNF-α, IFN-γ, and anti-collagen antibodies, but increased activity of neural endopeptidase in the joints of mice. Subsequent studies in the CIA model further validated the disease-protective effects of EGCG. In one such study, EGCG treatment of obese CIA mice showed its effectiveness in suppressing arthritis progression [205]. It correlated with reduced Th17 combined with increased Treg, and inhibition of STAT3. In another study, the treatment of CIA mice with EGCG was shown to ameliorate progression of clinical arthritis [206]. This outcome correlated with altered Th17/Treg balance in favor of the latter, along with suppression of osteoclastogenesis [206]. These effects involved EGCGinduced upregulation of the activity of phosphorylated-ERK, Nrf2, and HO-1, coupled with downregulation of STAT3 activation. Another aspect of anti-inflammatory activity of EGCG involves activation of AMPK, which can then activate SIRT1 [85]. This action of EGCG is similar to that of resveratrol and curcumin described above.
The testing of EGCG in RA synovial fibroblasts in culture as well as rat adjuvantinduced arthritis (AA) model have unveiled novel aspects of action of this natural product. Exposure to EGCG of IL-1β-treated RA synovial fibroblasts resulted in inhibition of several chemokines, including RANTES, epithelial-derived neutrophil-activating peptide 78 (ENA78), GROα, MCP-1 [34]. Also reduced was MMP2 activity. These outcomes were attributed to inhibitory effects of EGCG on PKCδ phosphorylation and NF-kB activation. In another study, EGCG treatment of rats with AA caused suppression of clinical disease coupled with reduction of IL-6 in serum and joints [207]. Using RA synovial fibroblasts, EGCG was shown to inhibit IL-1β-induced IL-6 production and increased soluble gp130 (sgp130) production. The latter effect was attributed to trans-signaling via induction of alternative
splicing of gp130 mRNA [207]. Increased sgp130 synthesis was proposed to inhibit IL6/soluble IL-6R-induced MMP2 activity in RA synovial fibroblasts and joint tissue of rats. In a different, prophylactic study in the rat AA model, green tea extract containing EGCG was shown to afford protection against arthritis development [208]. This treatment reduced proinflammatory cytokine IL-17 but increased immunomodulatory cytokine IL-10 production by the draining lymph node cells [208]. In addition, the level of antibodies to mycobacterial heat-shock protein 65 (Bhsp65), a disease (arthritis)-related antigen, was reduced.
The testing of EGCG in interleukin (IL)-1 receptor antagonist knockout (IL-1RaKO) arthritis model showed its inhibitory effect against clinical arthritis, bone damage, and osteoclast activity [209]. In addition, EGCG decreased the levels/activity of pro-inflammatory cytokines, oxidative stress proteins, p-STAT3, mTOR and HIF-1α [209]. Furthermore, the effects on mTOR and HIF-1α correlated with altered Th17/Treg balance.
A limited number of pilot clinical studies have indicated protection by EGCG against cardiovascular and rheumatic complications in RA [210,211], but larger clinical trials are needed to validate these claims.
- 10.2. Sjogren’s Syndrome (SS)
SS is characterized by immune cell infiltrates into the salivary and lacrimal glands, leading to their destruction and dysfunction. In a study in the NOD mouse model of human SS, EGCG treatment reduced the levels of autoantibodies as well as immune cell infiltrates into the submandibular glands [212]. EGCG also ameliorated TNF-α-induced cytotoxicity of acinar cells, which involved p38 MAPK phosphorylation-based activation.
- 10.3. Multiple Sclerosis/EAE
The protective effect of EGCG against EAE in B6 model was shown to involve the production as well as cell signaling events related to cytokines IL-2, IL-15, and IL-7, which share the physiological function of regulating the survival, expansion, and differentiation of T cells [213]. EGCG also inhibited the expression of receptors for these 3 cytokines. The effect of EGCG on other cytokines and T cell subsets in EAE was revealed in additional studies. For example, in one study, EGCG was effective in inhibiting the progression of EAE by reducing the production of IFN-γ and IL-17 by CD4+ T cells and inhibiting the differentiation of these two pathogenic T cell subsets [214]. It involved modulation of the T cell subset-specific transcription factors, T-bet and RORγt, respectively, along with that of the STAT pathway [214]. EGCG also suppressed the activity of antigen-presenting cells by reducing their costimulatory function mediated via CD80 and CD86. In another study, EGCG treatment of EAE mice afforded protection against clinical disease and immune pathology [215]. These beneficial effects involved reduction in CD4+ T cell activation and proliferation, along with suppression of both pro-inflammatory cytokine production and the frequency of Th1 and Th17 cells [215]. However, there was an increase in Treg cells. The impact of EGCG on macrophages was unveiled in another study where EGCG treatment was shown to reduce macrophage infiltration in the CNS and the spleen, which was associated with reduced disease severity, [216]. Furthermore, EGCG was able to alter M1/M2 ratio in favor of the latter, when tested in vitro, which was attributed to NF-kB inhibition and glycolysis.
- 10.4. T1D
The anti-diabetic effects of EGCG were revealed through in vitro and in vivo testing. In vitro, EGCG treatment prevented cytokine-driven destruction of RINm5F cells (an insulinoma cell line) and restored insulin secretion [217,218]. Reduction in the activity of nitric oxide synthase (NOS), apparently via NF-kB inhibition; suppression of the cytokineinduced generation of reactive oxygen species (ROS); and modulation of the mitochondrial pathway were involved in this protective effect of EGCG. In vivo, EGCG treatment of STZinduced T1D [217] reduced the blood glucose level, downregulated iNOS, and reduced the islet cell mass [218]. In the NOD mouse model, EGCG treatment resulted in delayed
onset of diabetes, but without any effect on insulitis per se [219]. The level of insulin and glycosylated hemoglobin in plasma was reduced, whereas the level of immunomodulatory cytokine IL-10 was increased [219].
- 10.5. Colitis
- 10.6. Psoriasis
- 10.7. Safety and Adverse Effects
EGCG is a component of green tea, which is one of the most widely consumed beverages in the world. The use of EGCG in studies in animal models and patients/volunteers has revealed that overall, it has a good safety profile. However, in some cases with underlying diseases such as diabetes and colitis, EGCG may display unexpected adverse reactions [231,232]. For example, EGCG was shown to induce nephrotoxicity or cardiotoxicity in animal models of diabetes and renal toxicity in an animal model of colitis [231,232]. In addition, hepatotoxicity has also been linked with EGCG use in some cases.
11. Triptolide
Triptolide mediates its effects on multiple cell types via inhibiting the XPB subunit of the transcription factor TFIIH core complex, which inhibits RNA polymerase II-mediated transcription and repair [233,234]. Specific targets related to inflammation that are identified include, NF-kB, IL-6/STAT3/SOCS3 signaling pathway, MAPK, and AMPK [86,234,235] (Table 3, Figure 2).
- 11.1. Immune Response
Triptolide has immune-inhibitory effects, which are mediated via its effects on the T cells, DCs, and B cells [41,236,237]. In one study, the efficacy of triptolide in suppressing the proliferation and cytokine production (e.g., IL-2 and IFN-γ) of human T cells was compared with that of a well-known immunosuppressant drug, FK506 [236]. Triptolide was found to be superior to FK506 for inhibiting T cell proliferation and IFN-γ production, but comparable to FK506 for IL-2 production. In another study using human monocyte derived DCs, it was shown that triptolide inhibits the maturation and migration of DCs [41]. This was inferred from the in vitro testing of the phenotype of DCs (including cytokine production and chemotaxis in response to defined chemokines), as well as in vivo testing in mice of the migration of Langerhans cells from the skin to the regional lymph nodes [41].
- 11.2. Rheumatoid Arthritis
- 11.2.1. RA Patients/RA Synovial Fibroblasts
Synovial fibroblasts play a vital role in arthritis pathogenesis, and novel approaches are being tried to suppress their activity and migration in an effort to control the disease process. In a study on synovial fibroblasts from RA patients (RA-FLS), triptolide was shown to inhibit the viability and migration of these cells, and to reduce the production of pro-inflammatory cytokines, but to increase anti-inflammatory cytokine levels [238]. Also inhibited was the activation of JAK-STAT pathway. In another study, the profiles of gene expression and cellular signaling pathways were examined in the synovial tissues of patients with RA and healthy controls [239]. Among several gene groups, CD2 (a T cell surface antigen) was identified and shown to be the target of a triptolide derivative.
Angiogenesis is an integral component of the pathogenesis of RA. TwHf (the parent herb for triptolide) has been shown to inhibit arthritis severity and progression of disease in RA patients and animal models [240]. It inhibits angiogenesis, highlighting one of the several mechanisms of its anti-arthritic activity.
- 11.2.2. Clinical Trials on TwHF in RA Patients
- 11.2.3. Animal Models of RA
Using the rat CIA model, it was shown that triptolide can inhibit clinical arthritis and reduce the level of pro-inflammatory cytokine IFN-γ in the peripheral lymphoid
tissue [246]. However, the level of immunoregulatory cytokine TGF-β in the periphery was enhanced. The above alterations correlated with reduced numbers of CD4 and CD8 T cells in Peyer’s patches, the site of immune tolerance, and reduced CD4 numbers in the periphery [246].
Triptolide affects a variety of innate immune cells, which influence various downstream and inter-connected immune pathways in arthritis and other autoimmune diseases [247]. A study based on gene expression and bioinformatics analysis pointed to triggering receptors expressed on myeloid cells (TREM)-1 signaling pathway as a major target [248]. Also reduced were DNAX-associated protein (DAP)12, JAK2, and STAT3. The levels of pro-inflammatory cytokines TNF-α, IL-1β and IL-6 were also reduced [248]. Above changes induced by triptolide were observed in both in vitro and in vivo (in ankle tissue of arthritic rats with CIA) model systems.
In a study in the rat AA model, it was shown that triptolide treatment resulted in inhibition of the disease [249]. The effects of triptolide were also tested on autophagy and cytokine production in the tissues (e.g., synovial tissue, thymus and spleen) [249]. Autophagy plays an important role in a variety of physiological and pathological processes, including RA. The key genes involved in promoting autophagy were suppressed by triptolide, whereas certain pro-inflammatory cytokines tested were found to be reduced.
- 11.3. Multiple Sclerosis/EAE
- 11.4. T1D
- 11.5. Colitis
Several studies in two different models of colitis have unveiled the beneficial effects of triptolide. The two models used were DSS-induced colitis [253–257] and spontaneous colitis in IL-10-deficient mice [258–260]. Investigations based on the DSS-colitis model showed that triptolide mediates its disease-protective effects via diverse mechanisms [253–257]. These mechanisms include, inhibition of IL-1β in the colonic tissue [257]; suppression of IL-
- 6 expression in the mucosa [256]; recovery from disordered composition of gut microbiota and establishing an optimal balance of Bacteroides vs. Firmicutes [255]; reduced ROS production, M1 polarization, and immune cell infiltration into the mucosa, which involved effect of triptolide on Nrf2/HO-1 signaling as well as PDE4B/Akt/NF-kB pathway [254]; and inhibition of Th1 and Th17 differentiation, which involved targeting of JAK-STAT pathway by a triptolide derivative, ZT01 [253]. Another set of studies on triptolide based on IL-10-deficiency-colitis model indicated the following mechanisms in its beneficial effects [258–260]: the targeting IL-17 and inhibition of IL-6/STAT3 pathway [258]; reduction of pro-inflammatory cytokines (e.g., IL-12, IL-23, TNFα, IFN-γ) in colonic tissue, which involved NF-kB inhibition [259]; and inhibition of immune cell infiltration into the mucosa, along with reduced TNFα, IFN-γ, and TNF receptor 2, and NF-kB activity [260].
- 11.6. Psoriasis
- 11.7. Safety and Adverse Effects
Despite the beneficial effects of triptolide in arthritis and other autoimmune diseases, its clinical application has been impeded by concerns regarding its side effects [43,86,241–245,264]. The adverse effects were observed in animal models and patients/volunteers. These pertained to gastrointestinal tract, liver, kidney, heart and reproductive organs [43,86,241–245,264,265]. Furthermore, adverse effect on the reproductive system in both males and females is one of the primary concerns. In clinical trials, in some cases, reduced sperm count and functionality in men, and effects on menstruation in women were observed.
12. Concluding Remarks
Natural products derived from diverse dietary and medicinal plants are a promising resource for potential therapeutic compounds for the management of chronic inflammatory and autoimmune diseases. The foregoing discussion highlighted mechanisms of action and applications of five such representative compounds in animal models of autoimmunity. In many parts of the world, several natural product extracts are already being used by practitioners of traditional medicine. Similarly, some of the isolated/purified compounds are consumed as over-the-counter food supplements by healthy individuals worldwide for improving immunity and health maintenance. It is hoped that following well-controlled clinical trials in patients with specific autoimmune diseases of interest, one or more of these compounds with desirable traits would be usable globally as therapeutic agents in near future. Probably, these compounds might initially be used as adjuncts to conventional medications before they are considered as alternative choices for certain drugs and diseases.
These decisions might differ for different populations based on their prior knowledge and/or experience in the use of natural products for treatment purposes. Nevertheless, natural product compounds offer a vast opportunity for the development of promising new drugs for the treatment of autoimmune diseases.
Author Contributions: Conceptualization, K.D.M. and S.H.V.; writing—original draft preparation, K.D.M.; writing—review and editing, K.D.M. and S.H.V.; and visualization, S.H.V. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Institutional Review Board Statement: Ethical review and approval were waived for this study due to the nature of this study, which is a review of published original studies conducted by a large number of investigators.
Informed Consent Statement: Not applicable. Data Availability Statement: Not applicable.
Acknowledgments: We thank Steven Dudics and David Langan, our past laboratory members and now colleagues, for helpful discussions on natural product compounds during their own studies, which were helpful in writing this review article.
Conflicts of Interest: The authors declare no conflict of interest.
Figures
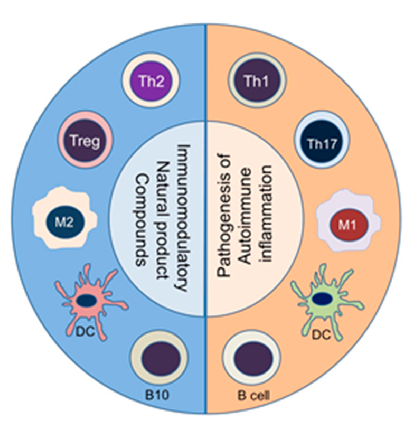
Figure 6
Natural product compounds shift the balance between pathogenic pro-inflammatory immune cells (right panel) and protective anti-inflammatory/immunomodulatory cell subsets (left panel). This diagram illustrates how diverse natural products can rebalance immune cell populations in autoimmune disease.
diagram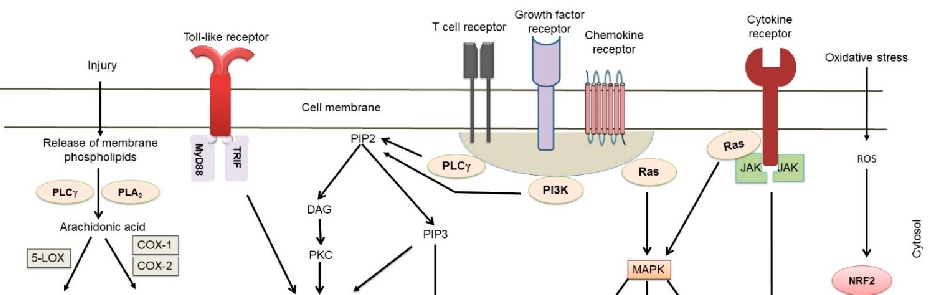
Figure 7
In vivo models of autoimmune diseases have demonstrated therapeutic efficacy of various natural compounds. This figure presents preclinical evidence from animal models of rheumatoid arthritis, multiple sclerosis, or inflammatory bowel disease treated with natural products.
chart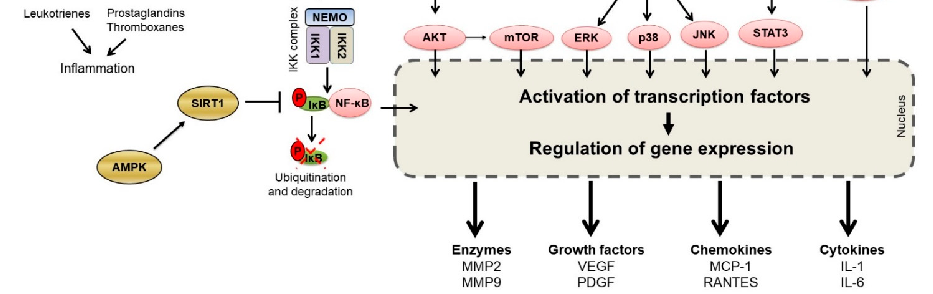
Figure 8
Clinical translation of natural anti-inflammatory products requires understanding of bioavailability and dose-response relationships. This figure discusses the therapeutic potential and limitations of natural product-based interventions for autoimmune conditions.
diagramUsed In Evidence Reviews
Similar Papers
The AAPS journal · 2013
Therapeutic roles of curcumin: lessons learned from clinical trials.
Digestive diseases and sciences · 2005
Curcumin therapy in inflammatory bowel disease: a pilot study.
Molecular nutrition & food research · 2013
Multitargeting by turmeric, the golden spice: From kitchen to clinic.
Gastroenterology · 2017
Diet as a Trigger or Therapy for Inflammatory Bowel Diseases.
Frontiers in endocrinology · 2023
Oxidative stress, hormones, and effects of natural antioxidants on intestinal inflammation in inflammatory bowel disease.
Journal of nanobiotechnology · 2022